ISSN: 0973-7510
E-ISSN: 2581-690X
https://dx.doi.org/10.22207/JPAM.13.1.64 | © The Author(s). 2019
Cancer is a major cause of death worldwide and angiogenesis is critical in cancer progression. Development of new blood vessels and nutrition of tumor cells are heavily dependent on angiogenesis. Angiogenesis is the formation of new blood vessels from existing vasculature. There are several “on” and “off” switches that regulate the process. Decreasing or inhibiting angiogenesis can be therapeutic in cancer and other diseases. Thus, angiogenesis is an important process that occurs both during health and disease. This study was conducted to investigate the anti-angiogenic activity of Carica papaya leaf. The anti-angiogenic activity of papaya leaf was evaluated using docking behaviour of known bioactive compounds of leaf as ligands with angiogenic receptors VEGFR1 and VEGFR2 along with their putative binding sites using Swiss Dock Web server (In silico) and further based on docking results leaf aqueous extract was used for implantation in chorioallantoic membrane (CAM) egg yolk angiogenesis model (in vivo). Docking studies and binding free energy calculations revealed that among known bioactive compounds of leaf Ascorbic acid, quercetin, riboflavin and lycopene have maximum free energy as compared to other investigated ligands. Similarly, CAM assay also showed the inhibitory effect of the Carica papaya leaf with respect their reduction in length, size and junctions of blood capillaries compared to untreated egg yolk. The results showed that Ascorbic Acid, Quercetin, Riboflavin and, Lycopene (leaf compounds) can attenuate angiogenesis in pathological conditions and can be potent in drug discovery as well as medical science.
Carica papaya, Angiogenesis, Medical Science, Cancer, Pathological, Phytochemical.
Angiogenesis is a multistep process. In normal physiology, angiogenesis has a role in embryogenesis, the female reproductive cycle, and wound healing and bone formation2. Angiogenesis is controlled by number of growth factors and inhibitors. Well known angiogenic (stimulatory) growth factors include basic Fibroblast Growth Factor (bFGF), Vascular Endothelial Growth Factor (VEGF), Granulocyte Colony-Stimulating Factor (G-CSF), Interleukin-8 (IL-8) and Transforming Growth Factors alpha and beta (TGF-a and TGF-b) 8. When these factors are over expressed leads to diseases for example in Cancer-when solid tumor can’t grow beyond a limited size but VEGF over expression leads to Metastasis of Cancer6,15,25. Angiogenic inhibitors include Angiostatin, Interferons (alpha, beta and gamma), Endostatin, Interleukin-12 and retinoids. Many plants have been identified and used as medicinal herbs from prehistoric times12,19. Treatment with medicinal plants is considered very safe as there is no or minimal side effects18,19. Carica papaya is a giant herbaceous plant-resembling a tree but not woody. It is one of the 22 accepted species in the genus Carica of the family Caricaceae26,27. It is native to lowland tropical areas of Central and South America, but is now grown in tropical to subtropical regions throughout the world both commercially and in kitchen gardens19. This evergreen tree is commonly called as papaya with broadleaf that bears papaya fruits throughout the year. The leaves are large, 50-70 cm in diameter and spirally arranged confined to the top of the trunk with palmately lobed structure. Whole plant parts fruits, seeds, bark, pulp, peel of Carica papaya have different medicinal properties1,24. Further studies can be done on these parts to identify and isolate the most active phytochemical that attributing the medicinal properties of the plant. The aim of the present study is to evaluate anti angiogenic activity of the leaves of Carica papaya. In this study, reported constituents of Carica papaya leaf aqueous extract docked with angiogenic receptors using one of the molecular docking methods. Based on preferred orientation and docking results of some compounds leaf aqueous extract was selected and used for in vivo CAM model assay. To analyze the mechanism underlying normal and pathological angi-ogenesis, numerous in vivo angiogenic assays have been stabilized employing different species of laboratory animals, including (mouse, rat and rabbit) and fish. In this study, we focussed on major models of angiogenesis in the chick embryo. In the chick embryo, the chorioallantois membrane is formed between the days 4 and 5 of development, when the outer mesodermal layer of the allantois fuses with the mesodermal lining of the chorion and a network of blood vessels is gradually formed between the two layers. The central portion of the CAM is fully developed by the day 8 to 1021. At this time, it becomes capable of sustaining tissue grafts, while the outskirts of the CAM are still developing and expanding until the CAM fully envelops the embryo at the day 12 of incubation5. The CAM consists of three germ layers, ectoderm, mesoderm and endoderm. After developing the CAM model, we assessed the potential role of Carica papaya(s) active ingredients as anti- angiogenic factors by which it can treat or prevent disease(s) like Cancer.
Fertilized chicken eggs taken from nearby poultry, and fresh green papaya leaves, randomly collected from Yadav Nursery, Geeta Colony, East Delhi.
Chemical Requirements
Phosphate buffer saline (7.4 pH), Ethanol, Distilled water.
Aqueous extract preparation
The leaves were washed, cut into small pieces and crushed finely into grinder. The sample was filtered, an aqueous ratio of 1:10 ml was prepared and stored into refrigerator at 4°C in an airtight container prior to use for analysis.
Docking studies
Docking is frequently used to predict the binding orientation of small molecule drug candidates to their protein targets in order to in turn predict the affinity and activity of the small molecules22. Docking studies were carried out on the crystal structure of the angiogenic receptors VEGFR1 and VEGFR2 retrieved from the Protein Data Bank (PDB) using Swiss Dock web server under protein ligand interaction with known bioactive components of Carica papaya as ligands whose structures were determined from Zinc database. Most negative Gibb’s free energy interaction pose was chosen and leaf aqueous extract was selected for implantation in CAM model assay.
Egg yolk angiogenesis (CAM) assay5
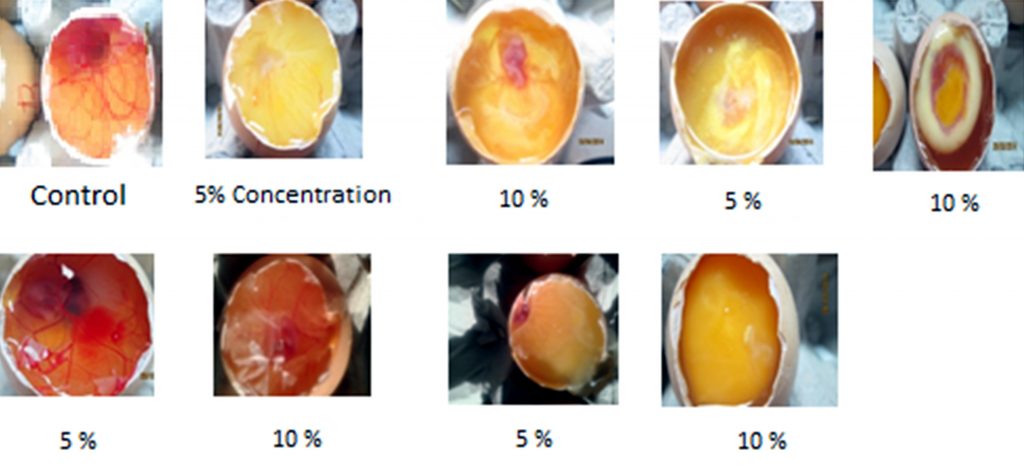
Fertilized leg horns chicken eggs were taken from nearby poultry. To check the viabilities of embryos, eggs were observed over a self-made lamp and kept in a humidified incubator at 37°C for 8 days. After the 8th day of incubation small window was cut in each egg and the plant extract was implanted at different concentration through the implanted disc in the experimental eggs while the control was implanted with normal saline under sterile conditions. Windows were sealed with tape and incubated for 3 days. After the period of incubation windows were reopen. Images were taken using Kodak digital camera and quantification of angiogenesis was done by observing the number of vessels and their branching. Experimental groups were compared with control groups.
Reported studies of Carica papaya leaves showed that aqueous extract contains great portion of steroids, quinones, tannins and alkaloids3. Compounds known in leaf aqueous extract are tocopherol, ascorbic acid, carpaine, quercetin, deoxyquercetin, lycopene, folic acid, riboflavin and cystine. Among all the compounds reported few compounds showed docking interaction with angiogenic receptors (VEGFR1 and VEGFR2).
Docking results
Negative sign signifies that binding between ligands (Ascorbic acid, Ouercetin, Riboflavin, and Lycopene) and receptors (VEGFR1 and VEGFR2) is going to occur at normal temperature and pressure (i.e. 293K and 1atm) and this reaction is thermodynamically spontaneous. It can be inferenced from the table 1, that Ascorbic acid binds most efficiently with VEGFR1 as compared to others. And, Lycopene binds with VEGFR2, there by inhibiting their functions (Fig. 1).
Table (1):
(Docking Results)
Compound |
Gibb’s free energy with VEGFR1 (kcal/mol) |
Gibb’s free energy with VEGFR2 (kcal/mol) |
|---|---|---|
Ascorbic acid |
-10.56 |
-7.37 |
Quercetin |
-7.36 |
-7.97 |
Riboflavin |
-6.99 |
-7.50 |
Lycopene |
-10.04 |
-8.18 |
CAM results
On the basis of docking results compounds with most negative interaction energy i.e. ascorbic acid, quercetin, riboflavin and lycopene were studied and leaf aqueous extract was used for implantation in CAM model assay to determine the anti-angiogenic property of Carica papaya leaves. Anti-angiogenic behaviour of leaf extract varies depending on the concentration. In control egg there was the development of number of blood vessels as the embryonic growth took place (Fig. 2). Implantation of the experimental egg with papaya leaves extract at different concentration inhibits the formation of blood vessels at different extend. Two concentrations of papaya leaves extract 5% and 10% were used. It was observed that there was less number of blood vessels in egg with 5% concentration as compare to control and the number of vessels formation inhibits as the concentration of extract was increased (Fig. 2). On the basis of these results it can be conclude that papaya leaves may be beneficial in various health conditions where formation of blood vessels leads to disease conditions.
Papaya is not only the delicious fruit, but it is loaded with vitamins, phytochemicals and several mineral compounds which help to treat health problems. The bioactivity of plant is attributed to phytochemical constituents. Reported studies on young leaves of Carica papaya revealed the presence of pharma-cologically active phytocompounds, alkaloids, phenolics, flavonoids and amino acids. From this study we can concluded that Carica papaya leaf extract may promotes immune system that may be due to the presence of Quercetin, Ascorbic acid, Riboflavin and lycopene as these compounds showed their potent binding with angiogenic receptors during molecular docking process, also the anti-angiogenic property of leaf extract reveals that Carica papaya may be used as a potent cancer fighter in the field of oncology as well as pharmacy. Also, it is reported that lycopene inhibits angiogenesis at achievable concen-trations in vivo and it can be regarded as a promising anti-angiogenic compound10. It may be due to this property of lycopene that it can be used as medicinal compound in future aspects. Ascorbic acid also found to be as anti oxidant in various chronic diseases4.
Based on reported studies and docking behaviour of some compounds we may concluded that anti angiogenic activity of carica papaya leaves in CAM model assay can be due to the presence of all these four compounds. As all plants Carica papaya leaves are rich in compounds of different properties. Further studies need to be conducted to determine anti-angiogenic pathways followed by leaf extract or juice. However, it can be concluded that administration of Carica papaya leaf juice is safe and does induce anti angiogenic process during disease condition like Cancer. It may play valuable role in the management of various diseases in the future.
The author would like to thank her guide Dr. Nidhi Srivastava for giving her guidance, support and cooperation to publish this paper.
The authors declare that they have no conflict of interest.
- Akhila. S, Vijayalakshmi N.G. Phytochemical studies on Carica papaya leaf juice. International journal of pharmaceutical sciences and research, 2015; 6(2): 880-883.
- Augustin HG, Kozian DH, Johnson RC. Differentiation of endothelial cells: analysis of constitutive and activated endothelial cell phenotypes. Bioessays, 1994; 16: 901-906.
- Ayoola PB, Adeyeye A. Phytochemical and nutrient evaluation of Carica Papaya leaves. IJRRAS, 2010; 5(3).
- Barrita JL, Sanchez MD. Anti oxidant role of ascorbic acid and his protective effects on chronic diseases. Intech open science, 2013.
- Beckers M, Gladis VM, Hamann W, Schmutzler W, Zwadlo KG. The use of chorioallantoic membrane of the chick embryo as test for anti inflammatory activity. Inflamm Res., 1997; 46: S29-S30.
- Carmeliet P. Angiogenesis in health and disease. Nature medicine, 2003; 9(6):653-660.
- Castejon MG, Casado AR. Dietary phytochemicals and their potential effects on obesity: A review. Pharmacological research, 2011; 64: 438-455.
- Chiou V.L, Kohn E.C, Davarpanah N, Lee JM. Novel therapeutic strategies for angiogenesis inhibition in recurrent ovarian cancer. Current angiogenesis, 2014; 3(4): 179-192.
- Craft PS, Harris AL. Clinical prognostic significance of tumor angiogenesis. Ann oncol., 1994; 5: 305-311.
- Denekamp J. Vascular endothelium as the vulnerable elements in tumors. Acta radiol oncol., 1984; 23: 217-225.
- Elgass S, Cooper AJ, Chopra M. Lycopene inhibits angiogenesis in human umbilical vein endothelial cells and rat aortic rings. The British journal of nutrition, 2011.
- Fajrin A, Tunjung WA. The flavonoids content in leaves and fruits of papaya (Carica papaya L.) Var. California and var. Gandul. KnE life sciences, 2015; 2: 154-158.
- Fauziya S, Krishnamurthy R. Papaya (Carica papaya): Source material for anti cancer. CIBTech journal of pharmaceutical science, 2013; 2(1): 25-34.
- Ferrara N, Kerbel RS. Angiogenesis as a therapeutic target. Nature, 2005; 438: 967-974.
- Hanahan D, Weinberg RA. Hallmarks of cancer: The next generation, 2011; 144: 646-674.
- Jain SK. Medicinal plant India, land and land people. National book trust of India. 1968:1-3.
- Jeong SJ, Koh W, Lee EO, Lee HJ, Bae H et al. Antiangiogenic phytochemicals and medicinal herbs. Phytotherapy research, 2010.
- Kadioglu O, Seo EJ, Efferth T. Targeting angio-genesis by phytochemicals. Medicinal and aromatic plants, 2013; 2(5).
- Krishna KL, Paridhavi M, Patel JA. Review on nutritional, medicinal and pharmacological properties of papaya (Carica papaya Linn.). Natural product radiance, 2008; 7(4): 364-373.
- Liekens S, Clercq ED, Neyts J. Angiogenesis: regulators and clinical applications. Biochemical Pharmacology, 2001: 253-270.
- Mohr T, Desser L. Plant proteolytic enzyme papain abrogates angiogenic activation of human umbilical vein endothelial cells in vitro. BMC complementary and alternative medicine, 2013; 13: 231.
- Narayanaswamy R, Shymatak M, Chatterjee S, Wai LK, Arumugam M. Inhibition of angiogenesis and nitric oxide synthase, by embelin and vilangin using in vitro, in vivo & in silico studies. Advanced pharmaceutical bulletin, 2014; 4(2): 543-548.
- Oklu R, Walker TG, Wicky S, Hesketh R. Angiogenesis and current antiangiogenic strategies for the treatment of cancer. Emerging technologies, 2010; 21: 1791-1805.
- Ruggiero L, Sogno I, Foccaccetti C, Bartolini D, Magnani E, et al. Effects of diet derived molecules on the tumor microenvironment. Current angiogenesis, 2012; 1(3): 206-214.
- Verheul HM, Pinedo HM. Inhibition of angiogenesis in cancer patients. Expert opin emerg drugs, 2005; 10: 403-412.
- Vyas SJ, Khatri TT, Ram VR, Dave PN, Joshi HS. Biochemical constituent in leaf of Carica papaya: ethno medicinal plant of Kachchh region. International letters of natural sciences, 2014; 12: 16-20.
- Yogiraj V, Goyal PK, Chauhan CS, Goyal A, Vyas B. Carica papaya Linn: An overview. International journal of herbal medicine, 2014; 2(5): 01-08.
© The Author(s) 2019. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.