ISSN: 0973-7510
E-ISSN: 2581-690X
The pandemic on SARS-CoV-2 infection has adversely impacted mankind all over the globe and more importantly, amidst individuals having Type-2 diabetic mellitus (T2DM) as well as among those who produce SARS-CoV-2 intricacies. Humoral and T cell-mediated immunity are adaptive immunity has a pivotal role to play in removing pathogens, comprising SARS-CoV-2. The infected cells are being eliminated by the primarily cytotoxic CD8+ T cells as well as certain antibodies in opposition to SARS-CoV-2 among humoral immunological responses possess the capacity for neutralizing this virus or by eliminating the infected cells with the support of cytotoxic in order to manage the progression of the disease. This cross-sectional study was carried out between January 2022 to December 2022. The serum samples were used to analyze SARS-CoV-2 total Ab among individuals having and not having T2DM and several metabolic risk factors like hypertension using WANTAI SARS-CoV-2 Total Ab ELISA Kit. There were 354 individuals, of which 141 (39.8%) had T2DM and 213 (60.2%) were nondiabetic patients. T2DM showed reduced antibody levels (average 5 AU/ml) than those without diabetes (average 12 AU/ml). SARS-CoV2 total antibody levels are considerably lower in hypertension patients (8 AU/ml) over those who are normotensive (14 AU/ml). The present study implies that the continuous monitoring of the total antibody profile of SARS-CoV-2 that may be a practical strategy to help individuals with T2DM and hypertension to determine their need of precautionary doses for sustaining SARS-CoV-2 vaccines by producing immunity for protection against infections.
SARS-CoV-2 Vaccines, Diabetic, Non-diabetic, Comorbidities, SARS-CoV-2 Total Antibody Levels
The World Health Organization (WHO) estimates as 6.6 million populations have been deceased due to SARS-CoV-2 infection, which has affected 651 million people globally as of December 23, 2022.1 The pandemic on SARS-CoV-2 infection has adversely impacted mankind all over the globe up to a remarkable level, more importantly, amidst individuals having Type-2 diabetic mellitus (T2DM) as well as among those who have additional causative factors in order to produce SARS-CoV-2 intricacies.2 Case fatality rates might surge 10% as a result of prior medical complications including cardiovascular disease, T2DM, metabolic syndrome, and hypertension. Also, the development of metabolic inflammation, a condition of chronic, low-grade inflammation, and compromised innate/adaptive immunity may enhance the vulnerability of T2DM patients to the increasing difficulties of SARS-CoV-2 infections.3,4
To a strong sense of urgency, SARS-CoV-2 vaccinations have been created at a tremendous rate., BNT162b2 mRNA (Pfizer–BioNTech vaccine) which is the first mRNA vaccine, was authorized in the United Kingdom by the European Medicine Agency on November 02, 2020. Two vaccines in India include ChAdOx1-nCOV (CovishieldTM) and BBV-152 (CovaxinTM) have received approval under the Emergency Use Authorization, which began on January 16, 2021. ChAdOx1-nCOV is developed by Oxford University and AstraZeneca using a vaccine that is recombinant replication-deficient chimpanzee adenovirus-vectored which codes spike antigen of SARS-CoV-2 from human embryonic kidney 293 cells that are genetically engineered.5,6,7
BBV152 is a whole virion vaccine that is inactivated by ß-propiolactone and has an adjuvant made of SARS-CoV-2 proteins and Toll-like receptor 7/8. It was created cooperatively by Bharat Biotech through the Indian Council of Medical Research, Hyderabad. Particularly, immunological alterations among humans are caused as a result of acquired infections or humoral responses to vaccination.8 As a result of long-lived memory B cell generation, antibody-secreting cells (ASC) are also efficient in boosting recall responses when exposed to the same antigens again. Memory B cells take the lead in recall response for producing new antibodies for starting the reaction of the germinal center in manufacturing antibodies or production of newer ASC once the failure in the production of immunity occurred because of circulating antibodies.9
Experts suggest that immunological memories (antibodies and memory B cells) last around 6 months. Therefore, following the exposure of antigen, the chronic inflammation caused by obesity may reduce the activation of macrophages, production of innate cytokine, and decrease pro-inflammatory mechanisms. These modified, obesogenic conditions might slightly elucidate the existence of vaccine escape mechanisms and antiviral resistance in people with T2DM and/or obesity.10 Likewise, Obesity impairs the responses of B and T cell, particularly among persons with type 2 diabetes who are obese. Immune response dysregulation is also a further consequence of the adverse hormonal environment.11,12 Generally, due to the increased production of many pro-inflammatory cytokines/chemokines including TNF- a, IFN-g, IL-1b, IL-18, IL-12, RANTES, IL-6, MCP-1, and obese T2DM patients frequently have impaired innate/adaptive immunity. In fact, all of these bioactive inflammatory proteins have been linked to a higher risk of having SARS-CoV-2 problems.13,14
Therefore, in the present study, responses of humoral immunity were compared among individuals having and not having T2DM and several metabolic risk factors like hypertension and obesity. Also, in this article, we provide data demonstrating levels of SARS-COV-2-specific antibodies among communities having increased incidence of T2DM.
Study Cohort and Participants Recruitment
The Ethical Review Committee (2923/IEC/2021) of SRM Medical College Hospital and Research centre, Kattankulathur, Chengalpattu, reviewed and accepted. In order to participate in this trial, participants had to have at least three weeks passed since receiving the second dose of the SARS-CoV-2 vaccine. Self-reporting served as the basis for T2DM diagnosis. This cross-sectional study was carried out between January 2022 to December 2022. The serum samples were used to analyze SARS-CoV-2 total Ab among individuals having and not having T2DM and several metabolic risk factors like hypertension using WANTAI SARS-CoV-2 Total Ab ELISA Kit. Participants with their completed course of SARS-CoV-2 vaccine and were among the ages of 18 to 65 were included. Participants with autoimmune diseases, taking immunosuppressants, those with arthritis, and who had recently received the second dosage in regard to SARS-CoV-2 vaccination were not included. A form for informed permission and a sheet of patient information was acquired and subsequently approved. All participants in the research received an authorized questionnaire, which they fully filled out. A patient proforma sheet was used to collect information on each participant, including their age, gender, BMI, SARS-CoV-2 infection history, and any current illnesses (like diabetes along with hypertension).
Blood Sample – Collection and Processing
Following the completion of the consent form, an experienced phlebotomist took venous blood in a red top plain tube under aseptic conditions, then spun the blood after clot formation to separate serum. Following that, serum samples were aliquot and kept on -80°C till the tests run.
Measuring serum SARS-CoV 2 total antibodies levels specific to SARS-CoV-2
Enzyme-linked immunosorbent assay (ELISA) kit (WANTAI SARS-CoV-2 Total Ab ELISA Kit, Wantai SARS-CoV-2 Diagnostics, India) was used in accordance with the manufacturer’s instructions to determine specific antibodies in serum sample for SARS-CoV-2. For sample clarity and to remove any remaining cells or platelets, centrifugation of frozen serum was done at 10,000 g for five minutes at four degrees Celsius. Enzyme-linked immunosorbent assay (ELISA) was used in total antibodies detection involving IgG and IgM of SARS-CoV-2 qualitatively within the human serum. The polystyrene micro-well strips that were used in ELISA were called WANTAI SARS-CoV-2 Ab and were already coated by recombinant SARS-CoV-2 antigen. An antigen employed in the test was SARS-CoV-2 spike protein’s receptor-binding domain.
Addition of calibrators and samples
100 µl of Specimen, 50 µl of Positive calibrator, and 50 µl of Negative calibrator were added into the appropriate wells, except the Blank.
Incubation
For 30 minutes, plates were incubated at 37°C with the plate cover.
Washing
With diluted Wash Buffer, each well were washed five times and soaked for 30–60 seconds every time. Plate was then placed on a fresh towel or blotting paper followed by the last cycle for washing and tapping was done to take away leftover water.
Addition of HRP-Conjugate
HRP-Conjugate (100 µl) was added inside every well leaving out Blank.
Incubation
Incubated for 30 minutes, at 37°C with a plate cover in place.
Washing
The plate cover was removed and discarded the plate cover when the incubation period was over. With diluted Wash Buffer, each well were washed five times and soaked for 30–60 seconds every time. Plate then placed on a fresh towel or blotting paper followed by the last cycle for washing and tapping was done to take away leftover water.
Coloring
In each well, including the Blank, Chromogen Solution A (50 µl), Chromogen Solution B (50 µl) were added and stir delicately. Incubated for 15 minutes for 37°C and avoid light during incubation period. In Positive calibrator, reaction among the HRP-conjugate and the chromogen solutions for SARS-CoV-2 antibody-positive specimens, resulted in blue color.
Stopping Reaction
In every well Stop Solution (50 µl) were added and combined gently using a multichannel pipette or by manual method. SARS-CoV-2 antibody-positive specimens and positive calibrator shows well-developed intense yellow hue.
Calculating Absorbance
After the calibration with the Blank well of the plate reader, the absorbance was read at 450 nm. The reference wavelength of 600–650 nm has been set when using a dual filter instrument. The Cut-off value was determined and outcomes were assessed.
Population Factors
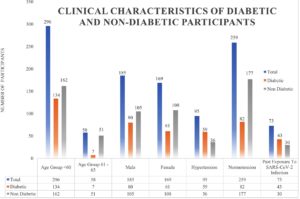
There were 354 individuals in the present study, of which 141 (39.8%) had T2DM and 213 (60.2%) were nondiabetic patients. Of these 354 people, 95 (26.1%) had hypertension and 259 (73.1%) had normotension. Among them, 169 (47.7%) females and 185 (52.3%) men and including 296 (83.6%) are ≤ 60 years of age, and 58 (16.4%) are among 61 to 65 years of age. In accordance with body mass index (BMI), 87(24.5%) individuals had BMI less than 25 kg/m2(normal weight), 164(46.3%) had a BMI between 25 and 30 kg/m2 (overweight), and 103 (29.2%) had higher BMI than 30 kg/m2 (obese). Before receiving immunizations, 73 (20.6%) of the 354 individuals had experience with SARS-CoV-2 infection and concomitant conditions. In which, 141 (39.8%) had type 2 diabetes, and 95 (26.9%) (Table and Figure).
Table:
Anti-SARS-CoV-2 serological findings and Clinical Characteristics among individuals stratified by diabetic and non-diabetic status
Variables |
Non-Diabetic (n=213) |
Diabetic (n=141) |
Overall (n=354) |
|---|---|---|---|
Age (in years) 18 – 60 years 61-65 |
162 (76%) 51 (24%) |
134 (95%) 7 (5%) |
296 (83.6%) 58 (16.4%) |
Gender Female Male |
108 (50.8%) 105 (49.2%) |
61 (43.2%) 80 (56.8%) |
169 (47.7%) 185 (52.3%) |
Hypertension Yes No |
36 (16.6%) 177 (83.4%) |
59 (41.8%) 82 (58.2%) |
95 (26.9%) 259 (73.1%) |
BMI Less than 25 Between 25 and 30 Greater than 30 |
62 (29.1%) 99 (46.4%) 52 (24.5%) |
25 (17.7%) 65 (46.0%) 51 (36.3%) |
87 (24.5%) 164 (46.3%) 103 (29.2%) |
Previous infection of SARS-CoV-2 Yes No |
30 (14%) 183 (86%) |
43 (30.4%) 98 (69.6%) |
73 (20.6%) 281 (79.4%) |
Total Antibody Titre (AU/ml) Mean (SD) Median (min, max) |
14.22 (4.06) 15.7 (0.7,22.4) |
9 (3.66) 10 (0.3,19.8) |
12.1 (4.66) 11.3 (0.3, 22.4) |
Total SARS-CoV-2 antibodies in those with or without type 2 diabetes
Depending on total SARS-CoV-2 antibodies analysis on the basis of T2DM condition, it was revealed that these individuals were not provoked high amounts of these antibodies. The total antibody level of SARS-CoV-2 was reactive totally with more than 9.5 AU/ml, that was greater than nine times the cutoff of the manufacturer’s suggested positive result. In contrast, when our cohort was divided into groups according to the presence of T2DM, those who have T2DM showed reduced SARS-CoV-2 total antibodies levels (average 5 AU/ml) than those without diabetes (average 12 AU/ml) based on T2DM conditions were included (Table).
Total SARS-CoV-2 antibodies among individuals with and without hypertension
The levels of SARS-CoV2 total antibody is significantly reduced in patients with hypertension (average 8 AU/ml) than in those without hypertension (average 14 AU/ml).
Total SARS-CoV-2 antibodies by age, gender and BMI
Males and females did not have substantially different levels of circulating SARS-CoV2 total antibodies. In general, SARS-CoV2 total antibodies levels were generally higher in females (15AU/ml) than in males (13AU/ml). From BMI results, regression analysis did not demonstrate a significant statistical difference in obesity status.
This recent work was done for estimating the SARS-CoV-2- total antibodies among 354 adults with both diabetic and non-diabetics, following two doses of BBV-152 (CovaxinTM) and ChAdOx1-nCOV (CovishieldTM). Of which 141 (39.8%) had T2DM and 213 (60.2%) were nondiabetic patients. Out of these 354 people, 95 (26.1%) had hypertension and 259 (73.1%) had normotension as same results were determined in the research work conducted by Hamad Ali et al which shows that 26.3% were found to have hypertension and 73.7% had normotensive.15 Then, it is analyzed in relation to diabetes status, gender, BMI, time after the second dosage, and hypertension. Regarding age, gender, and BMI there were no appreciable differences in the overall SARS-CoV-2 antibody levels in our research population as they were also associated with the study findings of Pellini R et al shows no significance regarding age, gender and BMI.16
The present study involves substantially reduced SARS-CoV-2- total antibodies levels among T2DM population that are comprehensible because several defects of the immune system like neutrophil function, and macrophage/ monocyte impairment, dysfunction of the complement system, defects in antigen presentation, as well as the decreased proliferation of lymphocyte were known to be induced by insulin resistance and hyperglycemia.17 Our findings indicate that high levels of SARS-CoV-2- total antibodies are almost nine times more than the seropositivity threshold (9.5 AU/ml,), were recorded following vaccination as same as Klinger J et al., which showed nearly 8 times of the threshold raise.18
Robust levels of SARS-CoV-2- total antibodies in the present cohort study after BBV-152 (CovaxinTM) and ChAdOx1-nCOV (CovishieldTM) in two doses show that all people have effective processing and presentation of antigens, suggested like different BBV-152 (CovaxinTM) and ChAdOx1-nCOV (CovishieldTM) vaccination studies in diverse cohorts.15,16 Inhibition of the ACE2 receptor interaction and the spike (S1) protein of SARS-CoV-2 has been observed to be strongly positively correlated with viral neutralization experiments.19,20
Additionally, we looked at whether having type 2 diabetes affected the effectiveness of the humoral immune response to the BBV-152 (CovaxinTM) and ChAdOx1-nCOV (CovishieldTM) vaccination.21 To that purpose, the information demonstrating elevated levels of SARS-CoV-2- total antibodies among T2DM cohort is reliable and the study showing that diabetic patients’ humoral immunity to SARS-CoV-2 were equivalent to those of non-diabetic patients in terms of antibody titers. However, these reports were in accordance with the results derived the study conducted by Ali H et al., Chen Y et al. and Alagheband Bahrami, A et al.22,23 The levels of SARS-CoV2 total antibody is significantly reduced in patients with hypertension (average 8 AU/ml) than in those without hypertension (average 14 AU/ml). However, these outcomes coordinated with a study report which is conducted by Pellini R et al., shows an average between 6-10 AU/ml.16
In a study conducted by Hamad Ali et al., it was reported that there is equal potency beyond >60 age showed reduced vaccination efficiency among persons with chronic comorbidities, including T2DM, although the T2DM population’s humoral immunity and our findings were concomitant with the present study results as they have demonstrated decreased efficacy towards vaccination.15 There is emerging evidence that individuals with T2DM may have insufficient immune responses to humoral immunity against infection, placing them at a high risk of contracting the illness again.24 It should be noted that due to their increased risk of infection and inadequate prognostic techniques, patients having type-2 diabetes might need vaccinations first.25 According to research on antibody titers conducted 7 days following the vaccination second dose, humoral immunity significantly influences gender, age but not hypertension.26,27 Study results based on the discrepancies between BBV-152 (CovaxinTM) and ChAdOx1-nCOV (CovishieldTM) report the influence of hypertension, gender, age, BMI, diabetic status and its comorbidities on immune responses may be related to variations in cohorts, after the second dosage.28,29
Following two doses of the BBV-152 (CovaxinTM) and ChAdOx1-nCOV (CovishieldTM) vaccination, our findings show that individuals with and without T2DM have strong SARSCoV-2-total antibody responses. Nevertheless, compared to non-diabetics, T2DM sample possessed substantially reduced antibody titers; however, antibody titers do not significantly affect age, hypertension, gender and BMI. It is significant to note that continuous monitoring of the total antibody profile of SARS-CoV-2 may be a practical strategy to help individuals with T2DM determine their unique needs for precautionary doses for the maintenance of immune responses among SARS-CoV-2 vaccines.
ACKNOWLEDGMENTS
The author would like to thank Dr. KV Leela, M.D., Professor and Head of Microbiology for her constant guidance.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
This study was approved by the Institutional Ethical Committee of SRM Medical College Hospital and Research Centre (2923/IEC/2021).
INFORMED CONSENT
Written informed consent was obtained from the participants before enrolling in the study.
- Hu B, Guo H, Zhou P, Shi ZL. Characteristics of SARS-CoV-2 and COVID-19. Nat ev Microbiol. 2021;19:141–154.
Crossref - Who coronavirus (COVID-19) dashboard. World Health Organization. https://covid19.who.int/. Accessed August 26, 2022.
- Yen FS, Wei JC, Chiu LT, Hsu CC, Hwu CM. Diabetes, hypertension, and cardiovascular disease development. J Transl Med. 2022;20(1):9.
Crossref - Berbudi A, Rahmadika N, Tjahjadi AI, Ruslami R. Type 2 Diabetes and its Impact on the Immune System. Curr Diabetes Rev. 2020;16(5):442-449.
Crossref - Venkatesan B, Vajravelu LK, Ravi S, Thulukanam J, Muthamilan OL. SARS-CoV-2 Non Responders – an Analysis of Non Responsiveness to SARS-CoV-2 Vaccines among Healthcare Workers in 2021. J Pure Appl Microbiol. 2022;16(2):1187-1191.
Crossref - Wondmkun YT. Obesity, Insulin Resistance, and Type 2 Diabetes: Associations and Therapeutic Implications. Diabetes Metab Syndr Obes. 2020;13:3611-3616.
Crossref - Venkatesan B, Vajravelu LK, Ravi S, Thulukanam J, Muthamilan OL. Therapeutic and Diagnostic Approaches by using Nanotechnology in SARS-CoV-2 Infections. J Pure Appl Microbiol. 2022;16(4):2324-2336.
Crossref - Zabetakis I, Lordan R, Norton C, Tsoupras A. COVID-19: The Inflammation Link and the Role of Nutrition in Potential Mitigation. Nutrients. 2020;12(5):1466.
Crossref - Zheng J, Deng Y, Zhao Z, et al. Characterization of SARS-CoV-2-specific humoral immunity and its potential applications and therapeutic prospects. Cell Mol Immunol. 2022;19(2):150-157.
Crossref - Zatterale F, Longo M, Naderi J, et al. Chronic Adipose Tissue Inflammation Linking Obesity to Insulin Resistance and Type 2 Diabetes. Front Physiol. 2020;10:1607.
Crossref - Singh AK, Phatak SR, Singh R, et al. Humoral antibody kinetics with ChAdOx1-nCOV (Covishield™) and BBV-152 (Covaxin™) vaccine among Indian Healthcare workers: A 6-month longitudinal cross-sectional Coronavirus Vaccine-induced antibody titre (COVAT) study. Diabetes Metab Syndr. 2022;16 (2):102424.
Crossref - Siegel CA, Melmed GY, McGovern DPB, et al. SARS-COV-2 vaccination for patients with inflammatory bowel diseases: Recommendations from an international consensus meeting. Gut. 2021;70 (4):635-640.
Crossref - Franke F, Kirchenbaum GA, Kuerten S, Lehmann PV. IL-21 in Conjunction with Anti-CD40 and IL-4 Constitutes a Potent Polyclonal B Cell Stimulator for Monitoring Antigen-Specific Memory B Cells. Cells. 2020;9(2):433.
Crossref - Dinleyici EC, Borrow R, Safadi MA, van Damme P, Munoz FM. Vaccines and routine immunization strategies during the COVID-19 pandemic. Human Vaccines ; Immunotherapeutics. 2020;17(2):400-407.
Crossref - Ali H, Alterki A, Sindhu S, et al. Robust Antibody Levels in Both Diabetic and Non-Diabetic Individuals After BNT162b2 mRNA COVID-19 Vaccination. Front Immunol. 2021;12:752233.
Crossref - Pellini R, Venuti A, Pimpinelli F, et al. Initial observations on age, gender, BMI and hypertension in antibody responses to SARS-CoV-2 BNT162b2 vaccine. E Clinical Medicine. 2021;36:100928.
Crossref - Yan Y, Pang Y, Lyu Z, et al. The COVID-19 Vaccines: Recent Development, Challenges and Prospects. Vaccines. 2021;9(4):349.
Crossref - Prasad N, Bansal SB, Yadav B, et al. Seroconversion Rate After SARS-CoV-2 Infection and Two Doses of Either ChAdOx1-nCOV COVISHIELD™ or BBV-152 COVAXIN™ Vaccination in Renal Allograft Recipients: An Experience of Two Public and Private Tertiary Care Center. Front Immunol. 2022;13:911738.
Crossref - Klingler J, Weiss S, Itri V, et al. Role of IgM and IgA Antibodies in the Neutralization of SARS-CoV-2. med Rxiv. 2020.
Crossref - Pratesi F, Caruso T, Testa D, et al. BNT162b2 mRNA SARS-CoV-2 Vaccine Elicits High Avidity and Neutralizing Antibodies in Healthcare Workers. Vaccines. 2021;9(6):672.
Crossref - Boroumand AB, Forouhi M, Karimi F, et al. Immunogenicity of COVID-19 vaccines in patients with diabetes mellitus: A systematic review. Front Immunol. 2022;13:940357.
Crossref - Chen Y, Klein SL, Garibaldi BT, et al. Aging in COVID-19: Vulnerability, immunity and intervention. Ageing Res Rev. 2021;65:101205.
Crossref - Bahrami AA, Azargoonjahromi A, Sadraei S, Aarabi A, Payandeh Z, Rajabibazi M. An overview of current drugs and prophylactic vaccines for coronavirus disease 2019 (COVID-19). Cell Mol Biol Lett. 2022;27:38.
Crossref - Daryabor G, Atashzar MR, Kabelitz D, Meri S, Kalantar K. The Effects of Type 2 Diabetes Mellitus on Organ Metabolism and the Immune System. Front Immunol. 2020;11:1582.
Crossref - Pal R, Bhadada SK, Misra A. COVID-19 vaccination in patients with diabetes mellitus: Current concepts, uncertainties and challenges. Diabetes Metab Syndr. 2021;15(2):505-508.
Crossref - Hwang YC, Lu RM, Su SC, et al. Monoclonal antibodies for COVID-19 therapy and SARS-CoV-2 detection. J Biomed Sci. 2022;29(1):1.
Crossref - Ledford H. How obesity could create problems for a COVID vaccine. Nature. 2020;586(7830):488-489.
Crossref - Singh AK, Phatak SR, Singh R, et al. Antibody response after first and second-dose of ChAdOx1-nCOV (CovishieldTM®) and BBV-152 (CovaxinTM®) among health care workers in India: The final results of cross-sectional coronavirus vaccine-induced antibody titre (COVAT) study. Vaccine. 2021;39(44):6492-6509.
Crossref - Sette A, Crotty S. Immunological memory to SARS-CoV-2 infection and COVID-19 vaccines. Immunol Rev. 2022;310(1):27-46.
Crossref
© The Author(s) 2023. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.