ISSN: 0973-7510
E-ISSN: 2581-690X
The vaginal microbiome plays a vital role in maintaining reproductive health. Vaginal pH serves as a simple yet reliable clinical marker reflecting microbial balance. A healthy vaginal environment is typically dominated by Lactobacillus species, which help maintain an acidic pH that inhibits colonization by pathogenic organisms. An increase in vaginal pH disrupts this balance and is associated with leukorrhea, infertility, preterm birth, and increased susceptibility to sexually transmitted infections (STIs). This study aimed to determine the association between vaginal pH and microbial community composition in women attending a tertiary healthcare center. A total of 50 vaginal swab samples were analysed using standard culture and biochemical methods, and vaginal pH was categorized as low (≤4.5), mid (4.6-5.5), or high (>5.5). Elevated pH values were frequently associated with pathogenic organisms such as Escherichia coli, Klebsiella pneumoniae, and Gardnerella vaginalis, whereas low pH corresponded to predominance of Lactobacillus species. Candida albicans was detected across a wide pH range. These findings demonstrate that vaginal pH is closely linked to microbial profiles, where high pH reflects dysbiosis and low pH indicates a healthy vaginal ecosystem. Routine monitoring of vaginal pH may therefore serve as a useful tool in guiding diagnostic and therapeutic decisions in gynecological practice.
Vaginal pH, Microbiome, Microbial Communities, Vaginal Health, Bacteria
The vaginal microbiome is a highly dynamic ecosystem that plays a critical role in maintaining female reproductive health.1,2 It is primarily dominated by Lactobacillus species, which maintain a low vaginal pH through lactic acid production, creating an environment hostile to pathogenic microorganisms.3,4 Alterations in this balance, often characterized by an increase in vaginal pH, can disrupt microbial communities, predisposing women to bacterial vaginosis (BV), vulvovaginal candidiasis (VVC), and other reproductive tract infection.5-7
Recent studies suggest that dysbiosis of the vaginal microbiome is associated not only with infections but also with adverse reproductive outcomes such as infertility, spontaneous preterm birth, and increased susceptibility to sexually transmitted infections (STIs).8-12 Even minor fluctuations in vaginal pH can trigger microbial shifts, serving as a predictive marker for BV or VVC.13-15 These findings highlight the importance of routine monitoring of vaginal pH as a low-cost, non-invasive clinical parameter.16 Globally, the prevalence of BV remains high, affecting up to 30% of reproductive-age women, with regional variations influenced by demographic, cultural, and geographic factors.11,17 In India, the burden of reproductive tract infections, particularly BV and VVC, is significant and remains underreported due to inadequate screening practices.18 Understanding the relationship between vaginal pH and microbial composition in symptomatic women is crucial for early detection and prevention of these conditions. The composition of the vaginal microbiome is shaped by host factors such as antibiotic exposure, hormonal changes, sexual activity, and immune responses.19-21 Lactobacillus species, especially L. crispatus and L. jensenii, are strongly associated with reproductive health as they produce lactic acid, bacteriocins, and hydrogen peroxide, which inhibit pathogen overgrowth.22,23 Pathogenic species like Gardnerella vaginalis, Atopobium vaginae, and enteric bacteria such as E. coli thrive in alkaline environments.1,6,24 These pathogens can form biofilms and persist even after treatment, leading to recurrent infections.7,8,12 There is growing literature supporting the role of pH-targeted therapies such as lactic acid gels and probiotics in restoring vaginal microbiome balance.25-27 Clinical trials have demonstrated that lowering vaginal pH can suppress pathogenic growth and reduce recurrence rates of BV.27,28
However, data from Northeast India, particularly Assam, are sparse, and regional studies are essential to understand population specific pattern.18 The present study aims to analyse the correlation between vaginal pH levels and microbial prevalence in symptomatic women attending a tertiary care hospital in Assam. This study addresses a significant gap in regional data and provides insights that could guide diagnostic and preventive strategies.
This study was conducted after obtaining approval from the Institutional Ethics Committee of Gauhati Medical College & Hospital (Approval No: MC/190/2007/PT-II/MARCH.2023/9). All procedures involving human participants were performed in accordance with the ethical standards of the institutional research committee and relevant national guidelines. Written informed consent was obtained from all participants prior to sample collection. Participants were informed about the purpose of the study, procedures involved, potential risks and benefits, and their right to withdraw at any stage. Confidentiality and anonymity of patient data were strictly maintained throughout the study.
Data Source
Data were obtained from patients visiting the gynaecology and obstetrics department of Gauhati Medical College and Hospital containing laboratory and clinical records of female patients presenting with symptoms such as vaginal discharge, itching, and lower abdominal pain. Variables included presenting symptoms, pregnancy status, colour of the discharge, Microbial growth status, biochemical test results, colony characteristics, odour, measured vaginal pH, and identified microbial species along with antibiotic sensitivity test.
Sample inclusion criteria
Samples were included if they had complete data for vaginal pH and microbial identification. Entries with missing or unclear microbial data or pH values were excluded. A total of 50 valid samples were analysed.
Microbial identification
Vaginal swab samples were collected aseptically from the posterior fornix and lateral vaginal walls using sterile cotton swabs. Each swab was immediately placed into sterile transport tubes and transported to the microbiology laboratory within two hours of collection. Vaginal pH was measured directly from vaginal secretions using sterile pH indicator strips (range 3.8-7.0), which were applied to the vaginal wall prior to swab collection.
For microbial isolation, swabs were inoculated onto MacConkey agar and nutrient agar for bacterial culture and Sabouraud dextrose agar for fungal culture. Bacterial cultures were incubated aerobically at 37 °C for 24-48 hrs, while fungal cultures were incubated at 28 °C for up to 72 hrs. Isolates were identified based on colony morphology, hemolytic pattern, pigmentation, odour, Gram staining, and standard biochemical tests including catalase, indole, citrate utilization, and glucose fermentation. Identification was performed according to Bergey’s Manual of Systematic Bacteriology.
Vaginal pH categorization
Samples were categorized based on vaginal pH into three groups:
- Low pH (≤4.5): Typically, indicative of a healthy vaginal environment
- Mid-range pH (4.6-5.5): Transitional or early dysbiotic state
- High pH (>5.5): Suggestive of dysbiosis and potential infection
Data analysis
Descriptive and inferential statistical methods were used to analyse the relationship between microbial communities and vaginal pH. Frequency distributions, cross-tabulations, and trend analyses were employed to interpret patterns. Pearson correlation coefficients and chi-square tests were used to evaluate the strength of association between pH levels and microbial prevalence.
Microbial distribution
The distribution of microorganisms isolated from the samples is summarized in
Table 1. Both pathogenic and commensal organisms were detected. Escherichia coli was the most frequently isolated organism (24%), followed by Klebsiella pneumoniae (16%), Lactobacillus spp. (16%), Candida albicans (14%), Staphylococcus aureus (12%), Gardnerella vaginalis (10%), and Streptococcus spp. (8%). The detection of Lactobacillus spp. (Table 1) in 16% of samples indicates that only a minority of women retained a microbiome consistent with vaginal health, while the majority showed evidence of microbial imbalance or infection.
Table (1):
Frequency of Isolated Microbial Species
Microbial Species |
No. of Samples |
% of Total Samples |
|---|---|---|
Escherichia coli |
12 |
24% |
Klebsiella pneumoniae |
8 |
16% |
Candida albicans |
7 |
14% |
Staphylococcus aureus |
6 |
12% |
Gardnerella vaginalis |
5 |
10% |
Streptococcus spp. |
4 |
8% |
Lactobacillus (Normal Flora) |
8 |
16% |
Table (2):
Vaginal pH of the isolated Microbial Species
Microbial Species |
Vaginal pH (Mean ± Standard Deviation) |
Above Normal Limit (>4.5) |
|---|---|---|
Escherichia coli |
6.9 ±0.3 |
Yes |
Klebsiella pneumoniae |
6.8 ± 0.2 |
Yes |
Candida albicans |
6.4 ± 0.2 |
Yes |
Staphylococcus aureus |
6.5 ± 0.2 |
Yes |
Gardnerella vaginalis |
7.0 ± 0.3 |
Yes |
Streptococcus spp. |
6.3 ± 0.2 |
Yes |
Lactobacillus (Normal Flora) |
4.0 ± 0.1 |
No |
Mean vaginal pH associated with isolated microbial species
Vaginal pH patterns across microbial groups
A strong and consistent trend was observed between vaginal pH levels and microbial composition (Table 2). Samples dominated by Lactobacillus spp. exhibited a mean pH of 4.0 ± 0.1, which falls well within the normal acidic range of a healthy vagina. None of these samples had pH values exceeding 4.5, confirming that Lactobacillus dominance is tightly linked with maintenance of vaginal acidity. In contrast, samples harbouring pathogenic organisms showed substantially elevated pH values. Gardnerella vaginalis showed the highest mean pH (7.0 ± 0.3), consistent with its established role in bacterial vaginosis and biofilm-associated alkalinization of the vaginal environment.
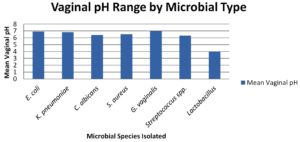
Escherichia coli and Klebsiella pneumoniae, both enteric pathogens, were detected primarily in highly alkaline environments with mean pH values of 6.9 ± 0.3 and 6.8 ± 0.2, respectively (Figure).
Figure. Vaginal pH Range by Microbial Type. Bar chart showing means pH values for each microbial group. Higher pH values are observed in samples with E. coli, Klebsiella pneumoniae, and Gardnerella vaginalis, whereas Lactobacillus-dominated samples show consistently lower pH
Staphylococcus aureus and Streptococcus spp. were also associated with elevated pH values (6.5 ± 0.2 and 6.3 ± 0.2, respectively), indicating that these organisms preferentially colonize disrupted vaginal ecosystems.
Candida albicans, however, demonstrated a broader pH distribution. Although its mean pH was elevated (6.4 ± 0.2), it was also detected in some samples with relatively lower pH, suggesting that fungal overgrowth is influenced not only by pH but also by host immunity, antibiotic use, and hormonal status. Overall, the data reveal a clear biological gradient:
As vaginal pH increases, protective Lactobacillus declines and pathogenic bacteria become dominant.
Relationship between pH elevation and dysbiosis
When samples were grouped according to pH category, nearly all specimens with pH >5.5 showed growth of potentially pathogenic bacteria or fungi, whereas samples with pH ≤4.5 were dominated exclusively by Lactobacillus spp. or showed no pathogenic growth.
This pattern demonstrates that vaginal alkalinization is strongly associated with dysbiosis, even though multiple organisms can coexist within this high-pH environment.
Statistical interpretation
A chi-square test comparing microbial presence with elevated vaginal pH (>4.5) yielded c2 = 0.000, p = 1.000, indicating that when analysed as a binary variable, pH elevation alone did not significantly discriminate between individual microbial species. This likely reflects the overlapping ecological niches of pathogens in dysbiotic vaginal environments, where multiple organisms thrive simultaneously at high pH.
Pearson correlation analysis between mean vaginal pH and microbial groups revealed a moderate negative correlation (r = -0.678), suggesting a trend toward decreasing Lactobacillus dominance with increasing pH; however, this association did not reach statistical significance (p = 0.094), possibly due to the limited sample size.
The present study shows the importance of vaginal pH as a fundamental modulator of microbial community composition in symptomatic women. Boskey et al.1 and Hillier et al.2 showed that vaginal pH and hydrogen peroxide producing Lactobacillus are key determinants of microbial balance. Ravel et al. 3 and Brotman4 confirmed that vaginal microbiota composition in reproductive-age women is largely shaped by Lactobacillus dominance, while Ma et al.5 emphasized that elevated pH disrupts this protective flora and promotes dysbiosis. Verstraelen and Swidsinski6 described how biofilm formation contributes to bacterial vaginosis epidemiology and treatment challenges. Borges et al.7 further linked probiotics to restoration of vaginal health, and Machado and Cerca8 demonstrated that Gardnerella vaginalis biofilms persist in higher pH, explaining recurrent infections.
Mitra et al.9 associated vaginal microbial imbalance with HPV-related cervical disease. Tachedjian et al.10 highlighted evolutionary uniqueness of Lactobacillus dominance, while van de Wijgert and Jespers11 reviewed global health impacts of vaginal dysbiosis. Tachedjian et al.12 then identified lactic acid as a key molecule in maintaining vaginal health. Ma et al.13 confirmed that acidic vaginal environments support protective microbial ecosystems in adolescents. Srinivasan and Fredricks14 provided an extensive overview of microbial dynamics, while Wilson et al.15 used computational models to show how small pH shifts can trigger transitions to BV or VVC.
Sobel16 originally described vulvovaginal candidiasis, noting its multifactorial nature. Amabeb and Anumb17 emphasized the physiological role of Lactobacillus in maintaining vaginal health, while Coudray and Madhivanan18 summarized bacterial vaginosis epidemiology in India. Muzny et al.19 updated the conceptual model of BV pathogenesis, Ceccarani et al.20 linked microbiome and metabolome changes during genital infections, and Nunn et al.21 reviewed the impact of vaginal pH on microbial community structure.
Santiago et al.22 showed that E. coli adheres more readily to epithelial cells at high pH, while France et al.23 reviewed diversity in vaginal microbiota across demographics. Fettweis et al.24 further connected dysbiosis and preterm birth risk. Souza et al.25 presented compelling evidence that elevated pH and depletion of Lactobacillus spp. are linked with infertility and early pregnancy loss. Sousa et al.26 demonstrated that lactic acid therapy can disrupt Gardnerella biofilms and restore balance. Armstrong-Buisseret et al.27 compared lactic acid gel with metronidazole, showing similar long-term cure rates, while Tidbury et al.28 confirmed better tolerability of lactic acid gels compared to antibiotics.
In summary, these studies collectively validate that vaginal pH acts as a central regulator of microbial community dynamics. Elevated pH favors pathogens like E. coli, Klebsiella, and Gardnerella, while low pH supports Lactobacillus-dominated health. In agreement with existing literature, our findings demonstrate vaginal pH as a non invasive, cost-effective screening tool to guide diagnostics, monitor risk, and inform therapeutic strategies. Our findings support the use of vaginal pH as a non-invasive, cost-effective screening tool to guide diagnostics, monitor risk, and inform therapeutic strategies.
Vaginal pH is a vital, non-invasive marker of reproductive tract health. This study demonstrates a statistically significant correlation between pH values and microbial prevalence. Elevated pH values are associated with potentially pathogenic communities, while lower pH levels correspond to healthy flora. These insights are valuable in clinical diagnostics and support the implementation of routine pH monitoring in gynecological assessments. Future studies should incorporate molecular diagnostics and longitudinal sampling to better understand the dynamics of microbiota shifts.
The study’s limitations include the relatively small sample size and reliance on culture-based methods. Future research should incorporate metagenomic sequencing and longitudinal sampling to delineate microbial dynamics with precision.
ACKNOWLEDGMENTS
The authors acknowledge the facilities and technical support provided by the Department of Microbiology, Gauhati Medical College, which made this work possible. Finally, heartfelt appreciation is extended to all patients who consented to participate in the study.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
The study was approved by the Institutional Ethics Committee, Gauhati Medical College & Hospital (Approval No: MC/190/2007/PT-II/MARCH.2023/9).
INFORMED CONSENT
Written informed consent was obtained from the participants before enrolling in the study.
- Boskey ER, Cone RA, Whaley KJ, Moench TR. Origins of vaginal acidity: high D/L lactate ratio is consistent with bacteria being the primary source. Hum Reprod. 2001;16(9):1809-1813.
- Hillier SL, Krohn MA, Rabe LK, Klebanoff SJ, Eschenbach DA. The normal vaginal flora, H₂O₂-producing lactobacilli, and bacterial vaginosis in pregnant women. Clin Infect Dis. 1993;16(Suppl 4):S273–S281.
Crossref - Ravel J, Gajer P, Abdo Z, et al. Vaginal microbiome of reproductive-age women. Proc Natl Acad Sci U S A. 2011;108(Suppl 1):4680-4687.
Crossref - Brotman RM. Vaginal microbiome and sexually transmitted infections: an epidemiologic perspective. J Clin Invest. 2011;121(12):4610-4617.
Crossref - Ma B, Forney LJ, Ravel J. Vaginal microbiome: rethinking health and disease. Annu Rev Microbiol. 2012;66:371-389.
Crossref - Verstraelen H, Swidsinski A. The biofilm in bacterial vaginosis: implications for epidemiology, diagnosis and treatment: 2018 update. Curr Opin Infect Dis. 2018;32(1):38-42.
Crossref - Borges S, Silva J, Teixeira P. The role of lactobacilli and probiotics in maintaining vaginal health. Arch Gynecol Obstet. 2014;289(3):479-489.
Crossref - Mitra A, MacIntyre DA, Lee YS, et al. The vaginal microbiota, human papillomavirus infection and cervical intraepithelial neoplasia: what do we know and where are we going next?. Microbiome. 2016;4(1):58.
Crossref - Mitra A, MacIntyre DA, Lee YS, et al. The vaginal microbiota, HPV infection and cervical intraepithelial neoplasia. Microbiome. 2016;4(1):58.
Crossref - Tachedjian G, Aldunate M, Bradshaw CS, Cone RA. The role of lactic acid production by probiotic Lactobacillus species in vaginal health. Res Microbiol. 2017;168(9-10):782-792.
Crossref - van de Wijgert JHHM, Jespers V. The global health impact of vaginal dysbiosis. Res Microbiol. 2017;168(9-10):859-864.
Crossref - van de Wijgert JHHM, Jespers V. The global health impact of vaginal dysbiosis. Res Microbiol. 2017;168(9-10):859-864.
Crossref - Srinivasan S, Fredricks DN. The human vaginal bacterial biota and bacterial vaginosis. Interdiscip Perspect Infect Dis. 2008;2008:750479.
Crossref - Srinivasan A, Fredricks DN. The vaginal microbiome in health and disease. Nat Rev Microbiol. 2023;21(1):33-45.
Crossref - Lee CY, Diegel J, France MT, Ravel J, Arnold KB. Evaluation of vaginal microbiome equilibrium states identifies microbial parameters linked to resilience after menses and antibiotic therapy. PLoS Computational Biology. 2023;19(8):e1011295.
Crossref - Sobel JD. Vulvovaginal candidosis. Lancet. 1997;369(9577):1961-1971.
Crossref - Amabebe E, Anumba DOC. The vaginal microenvironment: the physiologic role of Lactobacilli. Front Med. 2018;5:181.
Crossref - Coudray MS, Madhivanan P. Bacterial vaginosis—a brief synopsis of the literature. Eur J Obstet Gynecol Reprod Biol. 2020;245:143-148.
Crossref - Muzny CA, Taylor CM, Swords WE, et al. An updated conceptual model on the pathogenesis of bacterial vaginosis. J Infect Dis. 2019;220(9):1399-1405.
Crossref - Ceccarani C, Foschi C, Parolin C, et al. Diversity of vaginal microbiome and metabolome during genital infections. Sci Rep. 2019;9(1):14095.
Crossref - Onderdonk AB, Delaney ML, Fichorova RN. The human microbiome during bacterial vaginosis. Clin Microbiol Rev. 2016;29(2):223-238.
Crossref - Abou Chacra L, Fenollar F, Diop K. Bacterial Vaginosis: What Do We Currently Know? Front Cell Infect Microbiol. 2021;11:672429.
Crossref - Condori‑Catachura S, Ahannach S, Ticlla M, Kenfack J, Livo E, Anukam KC, Pinedo‑Cancino V, Collado MC, Dominguez‑Bello MG, Miller C, Merten S, Donders GG, Gehrmann T, Lebeer S. Diversity in women and their vaginal microbiota. Trends in Microbiology. 2025;33(11):1163-1172.
Crossref - Fettweis JM, Serrano MG, Brooks JP, et al. The vaginal microbiome and preterm birth. Nat Med. 2021;25(6):1012-1021.
Crossref - Souza SV, Monteiro PB, Moura GA de, Santos NO, Fontanezi CTB, Gomes IA, Teixeira CA. Vaginal microbioma and the presence of Lactobacillus spp. as interferences in female fertility: A review system. Jornal Brasileiro de Reprodução Assistida. 2023;27(3):496–506.
Crossref - O’Hanlon DE, Moench TR, Cone RA. Vaginal pH and microbicidal lactic acid when lactobacilli dominate the microbiota. J Infect Dis. 2013;207(11):1729-1737.
Crossref - Armstrong-Buisseret L, Brittain C, Anstey Watkins J, et al. Intravaginal lactic acid gel versus oral metronidazole for recurrent bacterial vaginosis: the VITA randomized controlled trial. BMC Womens Health. 2023;23:241.
Crossref - Tidbury F, Brülhart G, Müller G, Pavicic E, Weidlinger S, Eichner G, von Wolff M, Stute P. Effectiveness and tolerability of lactic acid vaginal gel compared to oral metronidazole in the treatment of acute symptomatic bacterial vaginosis: a multicenter, randomized‑controlled, head‑to‑head pilot study. BMC Women’s Health. 2025;25(1):7.
Crossref
© The Author(s) 2026. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.