ISSN: 0973-7510
E-ISSN: 2581-690X
Obligate anaerobes, which are part of normal intestinal flora are now gaining pathogenic potential by becoming more virulent and causing moderate to severe abdominal infections. Moreover, there is delay in initiation of appropriate antimicrobial therapy. The study aimed to describe and analyse 23 years data on anaerobic intra – abdominal infections in regards to the distribution and antimicrobial susceptibility patterns of the obligate anaerobes which were isolated from various intra – abdominal infections. The demographic and microbiological data was retrieved from the microbiology departmental registers. Total number of cases/specimen were 1124. Bacteroides fragilis group (238) (56%) and Peptostreptococcus sp (109) (25%) amounted to the majority of the isolates. Rare anaerobes like Clostridium sporogenes, Propionibacterium sp, Clostridium bifermentans and Fusobacterium varium were also isolated. Majority of mixed anaerobic infections were contributed by Bacteroides fragilis group and Peptostreptococcus sp (99) out of 102 mixed anaerobic infections). Chronic alcoholism was the most common predisposing condition (p value <0.05). Among the antimicrobials which were used by the clinicians for treating the infection, only Metronidazole was tested for its susceptibility pattern. One isolate was resistant to metronidazole (Diameter of inhibition zone was 6 mm). As they are fastidious they usually go unnoticed. Hence, this descriptive study intends to bring light on the large number of various obligate anaerobes and the potential diseases that they can cause and also the need for their antibiotic susceptibility testing to look for antimicrobial resistance among the isolates.
Anaerobes, Bacteroides Fragilis Group, Peptostreptococcus sp, Metronidazole
Intra-abdominal infections encompasses a plethora of pathological conditions that involve lesions of all the intra-abdominal organs. They range from inflammation of a single organ to peritonitis which can be primary, secondary or tertiary. They also include intraperitoneal, retroperitoneal and parenchymal abscesses.1 In uncomplicated intra-abdominal infections the infectious process only involves a single organ and does not proceed to peritoneum. In complicated intra-abdominal infections, the infectious process proceeds beyond the organ, and causes either localized peritonitis or diffuse peritonitis and are associated with morbidity and poor prognosis.1,2 Although the facultative anaerobes like E.coli, Klebsiella and other members of Enterobacteriaceae are the most common organisms to be implicated in intra-abdominal infections, obligate anaerobes also have the potential to cause intra abdominal infections. Obligate anaerobes, which are part of normal intestinal flora are now gaining pathogenic potential by becoming more virulent and causing moderate to severe abdominal infections. The obligate anaerobes were largely neglected as their mere incapability of causing serious abdominal infections. Infections due to anaerobic isolates can sometimes be missed because of the special measures required for their transportation (anaerobic transportation). These obligate anaerobes (like Bacteroides fragilis group, Peptostreptococcus sp) unlike the sturdy gram negatives and gram positives, are very fastidious and do not grow on common bacteriological media; hence, difficult to isolate them and hence, there is delayed diagnosis. Hence, successful isolation of these microorganisms in the microbiology laboratory requires incubation in anaerobic atmosphere, the use of specialized culture media, and prolonged incubation of culture. Moreover, there is delay in initiation of appropriate antimicrobial therapy. Despite the establishment and distribution of treatment guidelines,3,4 there is still lack of standardization in empiric management, specifically for certain indications where antibiotic therapy is most common that is for obligate anaerobes.5-7 In recent years, higher resistance rates of these microorganisms to some antimicrobial agents have been observed. Hence it is essential to know when an anaerobic infection is vital in order to use appropriate microbiologic methods to identify the bacteria and to select the correct treatment.8 In this regard, this study was aimed to describe and analyse the distribution of various obligate anaerobes causing varied anaerobic intra abdominal infections, and also to document any antimicrobial resistance among the obligate anaerobes.
A hospital-based retrospective analytical cross-sectional study was conducted in the Microbiology laboratory, at Jawaharlal Institute of Postgraduate Medical Education and Research, which is a tertiary care centre and a teaching hospital, situated in Puducherry (Union Territory), Tamil Nadu, South India. The study process analysed 1124 intra – abdominal infection cases received over a period of 23 years and 8 months (January 1994 – September 2017). The demographic and microbiological data was retrieved from the microbiology departmental registers maintained as a database in the Microbiology Department of the institute. The demographic data included age, gender and occupation of the patient. The microbiological data included the specimen received, the method of isolation and the isolated obligate anaerobes from various specimens. Attempt was also made to retrieve medical records of the concerned patients to perform a case review. Only past 10 years records (2007 to 2017) were retrieved from the medical record section. The clinical information (like antibiotic usage and risk factors) of the patients were retrieved from the medical records section.
Methods for processing of specimens
Pus and serosanguinous fluid was the specimen of choice. The specimen was collected in the syringe (intraoperative collection), and was immediately transferred into RCM (Robertson Cooked Meat) broth. The broth with the sample was sent to the microbiology laboratory for processing. Once, the broth with the sample is reached, it is incubated for 48 hours at 37°C in the incubator.
Flow chart for processing
RCM (Robertson Cooked Meat) broth with sample (DAY 1)
↓
After 48 hours of incubation
↓
RCM Smear was prepared from the broth and subjected to Gram staining (DAY 3)
↓
In the Gram stain of RCM, the type of organisms (Gram positive or Gram negative) seen were noted down
↓
RCM
↓
ANAEROBIC CULTURE was done on the following media:
PEA agar (Phenyl Ethyl agar)
Neomycin agar
Blood agar
Anaerobic culture was read on DAY 5, i.e. in 48 hours
On Day 5 the anaerobic culture plates were read and colonies with different morphologies were marked and subjected to aerotolerance (done on Blood agar)
DAY 5: ANAEROBIC CULTURE (Aerotolerance)
24 Hours 48 Hours  O2+ O2 –
O2+ O2 –
Day6 Day7
If there is growth in anaerobic incubation (O2-) on day 7 and no growth in the corresponding aerobic incubation (O2+), the isolate is confirmed as an obligate anaerobe, and gram staining, 3% KOH, bile esculin and sensitivity are done for the concerned isolate. The biochemical reactions and sensitivity plates are read on day 9 (after 48 hours of incubation).
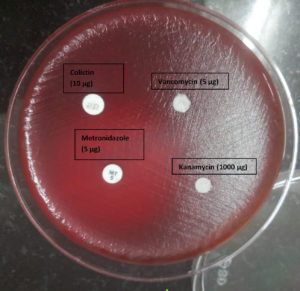
Presumptive identification (Table 3) was performed with antibiotic discs and spot tests (Wadsworth method)9; for example, at the genus level (Bacteroides fragilis group) (Figure 1).
Antimicrobial susceptibility testing for obligate anaerobes was started only in 2016. Metronidazole was the only antimicrobial which was tested. It was tested only against Bacteroides fragilis group according to standard CLSI guidelines. As the antimicrobial susceptibility testing for obligate anaerobes was started only in 2016, and among the antibiotics like beta-lactamase resistant penicillin, cephalosporin, clindamycin, macrolide, metronidazole and tetracycline, metronidazole is the most active antibiotic against Bacteroides fragilis group and it was the most common antibiotic preferred for treating anaerobic infections by the clinicians, hence, in this context, only metronidazole was tested against Bacteroides fragilis group. Moreover, there were studies documenting metronidazole resistance among the isolates of Bacteroides fragilis group.10-14 As regards the statistical analysis, continuous variables were analysed by mean and standard deviations, and percentages and proportions were used for categorical variables. Chi-square or Fisher’s exact tests were used to find significant association between predisposing factors and the clinical condition and also whether the isolated obligate anaerobe was significantly associated with the clinical condition. A univariate logistic regression analysis was also performed to document whether isolated obligate anaerobe was significantly (P < 0.05) associated with the clinical condition.
Total number of cases/specimens (A single specimen was received from a single patient) received over the period of 23 years and 8 months were 1124. Demographically, the majority of the patients were above the age of 40 (49%) (Table 1). The spectra of uncomplicated abdominal infections which were received were, Liver abscess (180), Appendicular abscess (224), Acute/chronic appendicitis (128), Necrotizing pancreatitis (74), Pancreatic abscess (64), Splenic abscess (68), Gut gangrene (56), and Perinephric abscess (54). Similarly, complicated abdominal infections were Intestinal perforations/ Perforation peritonitis (secondary peritonitis) (186) and Peritonitis (other than perforation peritonitis) (90). Appendicular abscess (224) and intestinal perforations/Perforation peritonitis (186) were the majority of the intra-abdominal infections (Table 2). Pus or serosanguinous fluid was the major specimen received in the anaerobic section. The specimens were processed anaerobically according to the SOP (Standard Operating Procedures). Out of 1124 cases only 326 (29%) cases were culture positive, whereas 798 cases were culture negative. These 326 cases yielded 428 anaerobic isolates. Bacteroides fragilis group (238) (56%) and Peptostreptococcus sp (109) (25%) amounted to the majority of the isolates. Rare anaerobes like Clostridium sporogenes, Propionibacterium sp, Clostridium bifermentans and Fusobacterium varium were also Isolated (Table 4 and Table 5). Out of 238, 107 (45%) isolates of Bacteroides fragilis group were isolated from appendicular abscess.
Table (1):
Age distribution.
Range of Age |
No. (out of 1124) |
Percentage |
|---|---|---|
5 – 10 years |
86 |
7.65 |
11 – 20 years |
144 |
13 |
21- 40 years |
348 |
31 |
40 years and above |
546 |
49 |
Table (2):
No. of cases.
Clinical conditions |
No. of cases |
|---|---|
Liver abscess |
180 |
Appendicular abscess |
224 |
Acute/chronic appendicitis |
128 |
Intestinal perforations/Perforation peritonitis (secondary peritonitis) |
186 |
Peritonitis (other than perforation peritonitis) |
90 |
Necrotizing pancreatitis |
74 |
Pancreatic abscess |
64 |
Splenic abscess |
68 |
Gut gangrene |
56 |
Perinephric abscess |
54 |
Total |
1124 |
Mixed anaerobic infections
Majority of mixed anaerobic infections were contributed by Bacteroides fragilis group and Peptostreptococcus sp ( 99 out of 102 mixed anaerobic infections). The rest three cases (all the three cases were of intestinal perforation) yielded Clostridium sporogenes, Bacteroides fragilis group and Peptostreptococcus sp in the first, Fusobacterium sp and Bacteroides fragilis group in second and Fusobacterium varium and Peptostreptococcus sp in the third.
Table (3):
Presumptive identification with antibiotic disks of main anaerobes. (Adapted from Wadsworth Anaerobic Bacteriology manual)18.
Microorganism or group |
Vancomycin (5 μg) |
Kanamycin (1000 μg) |
Colistin (10 μg) |
Penicillin (2 U) |
|---|---|---|---|---|
Gram-positive |
S |
V |
R |
V |
Gram-negative |
R |
R |
S |
|
B. fragilis group |
R |
R |
R |
R |
Other Bacteroides species |
R |
R |
V |
S |
Porphyromonas |
S |
R |
R |
|
Fusobacterium |
R |
S |
S |
|
Clostridium perfringens |
S |
S |
R |
|
Prevotella |
R |
V |
V |
|
Parabacteroides |
R |
R |
R |
S = sensitive; R = resistant; V = variable
Case review
We could retrieve only the last 10 years case records. A total of 656 intra-abdomen infection cases were received in last 10 years (2007 to 2017). The demographic information was compiled from the department registers (not from the case records). Out of 1124 cases, 728 (65%) were males and 396 (35%) were females. Occupation wise, majority of them were labourers (the occupation data was obtained only for 656 cases, as the data had to be retrieved from the case records). Predisposing or underlying conditions were present in 69 (out of 110 culture positive cases) (63%) instances. Chronic alcoholism, leading to gastric/duodenal ulcers and intestinal perforation (43%) was the most common predisposing condition (p value < 0.05). As regards the significant association of obligate anaerobe with the clinical condition, using univariate logistic regression analysis, Bacteroides fragilis group (45%) was found to be significantly associated with appendicular abscess (p value < 0.05), and Peptostreptococcus sp (47%) was significantly associated with intestinal perforation (p value < 0.05).
Other Predisposing conditions were, recent abdominal surgery in 35% of instances, malignancy in 15% of instances (leukemia), immune deficiency in 7% of instances. The antimicrobials used for treating the infection were beta-lactamase resistant penicillin, cephalosporin, clindamycin, macrolide, metronidazole and tetracycline.
Table (4):
Spectrum of anaerobic isolates.
Isolate |
No |
(%) |
|---|---|---|
Bacteroides fragilis group |
238 |
56 |
Peptostreptococcus sp |
109 |
25 |
Peptostreptococcus anaerobius |
56 |
13 |
Clostridium sporogenes |
4 |
0.9 |
Fusobacterium sp |
3 |
0.7 |
Obligate non sporing gram positive bacilli |
5 |
1.2 |
Clostridium perfringens |
7 |
1.6 |
Propionibacterium sp |
1 |
0.2 |
Clostridium bifermentans |
3 |
0.7 |
Microaerophilic streptococci |
1 |
0.2 |
Fusobacterium varium |
1 |
0.2 |
TOTAL |
428 |
Table (5):
Specimen wise distribution of isolates.
Isolate |
Clinical conditions |
No. of obligate anaerobes isolated from the specimen |
Percentage |
|---|---|---|---|
Bacteroides fragilis group |
1) Appendicular abscess |
107 |
45 |
2) Intestinal perforation |
82 |
44 |
|
3) Acute/chronic appendicitis |
32 |
25 |
|
4) Necrotizing pancreatitis |
8 |
11 |
|
5) Gut gangrene |
7 |
13 |
|
6) Splenic abscess |
2 |
3 |
|
Total |
238 |
||
Peptostreptococcus sp |
1) Intestinal perforation |
88 |
47 |
2) Appendicular abscess |
9 |
4 |
|
3) Gut gangrene |
10 |
18 |
|
4) Splenic abscess |
2 |
3 |
|
Total |
109 |
||
Peptostreptococcus anaerobius |
1) Intestinal perforation |
48 |
26 |
2) Appendicular abscess |
8 |
3.6 |
|
Total |
56 |
||
Clostridium sporogenes |
Intestinal perforations/ |
4 |
2 |
Perforation peritonitis |
|||
Fusobacterium sp |
Intestinal perforation |
3 |
2 |
(appendicular perforation) |
|||
Fusobacterium varium |
Peritonitis |
1 |
1 |
Obligate non sporing |
Intestinal perforations/ |
5 |
2.7 |
gram positive bacilli |
Perforation peritonitis |
||
Clostridium perfringens |
1) Gut gangrene |
4 |
7 |
2) Intestinal perforation |
2 |
8 |
|
3) Necrotising pancreatitis |
1 |
1.4 |
|
Total |
7 |
||
Propionibacterium sp |
Peritonitis |
1 |
1.1 |
Clostridium bifermentans |
Appendicular abscess |
1 |
0.4 |
Microerophilic streptococci |
Intestinal perforations |
1 |
0.5 |
Antimicrobial susceptibility testing
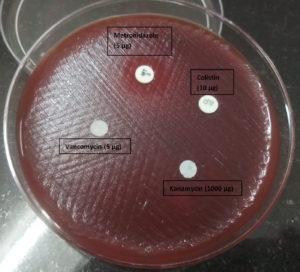
The antimicrobial susceptibility testing for the anaerobes was started only in 2016. Among the antimicrobials which were used by the clinicians for treating the infection, only Metronidazole was tested for its susceptibility pattern. It was tested only against Bacteroides fragilis group by Kirby – Bauer disk diffusion method according to standard CLSI Guidelines. From January 2016 to September 2017, 17 (out of 238) Bacteroides fragilis isolates were obtained from intra – abdominal infections. Among them only one isolate was resistant to metronidazole (Diameter of inhibition zone was 8 mm, Figure 1 and Figure 2).
Figure 1. Identification of Bacteroides fragilis group using discs Vancomycin (5 μg)-Resistant, Kanamycin (1000 μg) – Resistant and Colistin (10 μg) – Resistant.
Over a period of 23 years, 1124 intra – abdominal infection cases were received. Majority of the patients were males (65%), and majority were labourers by occupation. Appendicular abscess (224) and intestinal perforations/Perforation peritonitis (186) were the majority of the intra-abdominal infections, with pus or serosanguinous fluid being the major specimen received. Bacteroides fragilis group (238) (56%) and Peptostreptococcus sp (109) (25%) amounted to the majority of the isolates. Massimo Sartelli,10 also documented Bacteroides fragilis group to be the major anaerobic isolate.10 In a similar study done by Brook I and Frazier EH,11 the most frequently isolated anaerobes were Bacteroides spp. (B. fragilis group), Peptostreptococcus, Clostridium and Fusobacterium spp.11 Bacteroides fragilis emerged as the predominant isolate in a study by Shree N et al.12 Simillarly, Chia T C et al. in their study also documented Bacteroides fragilis as the major anaerobic isolate.13 Nicole Lopez et al.14 also documented Bacteroides spp. (B. fragilis group), and Clostridium as major anaerobic isolates. In our study, we had also isolated rare anaerobes like Fusobacterium varium, Clostridium bifermentans and Propionibacterium sp. Rare anaerobes like Fusobacterium spp was also isolated by Brook I and Frazier EH.11 The various studies have shown variety of anaerobes causing significant intra–abdominal infections.15-18 Majority of Bacteroides fragilis group (107) were isolated from appendicular abscess, followed by intestinal perforation (82) in our study. The above studies and the present study shows the importance of anaerobes in various intra-abdominal infections. Antibiotic resistance among anaerobic microorganisms has also increased in recent years.19-22
In the present study, only metronidazole was tested for its susceptibility pattern and was tested only against Bacteroides fragilis group. One isolate of Bacteroides fragilis group was resistant to metronidazole in the present study. In a study by Karlowsky JA et al.,23 regarding the Bacteroides fragilis group, resistance to penicillin was observed in 80–90% of isolates, while a higher proportion of the strains (20%) were also resistant to amoxicillin clavulanate.23 The overall resistance rate to carbapenems was very low.22,23 In another study, resistance to penicillin was observed to be around 80–90% of Bacteroides fragilis group. while a higher proportion of the strains (20%) were resistant to amoxicillin–clavulanate.24 The overall resistance rate to carbapenems was very low (<1%).24 But in another study, the authors have reported a high rate (64%) of carbapenem resistance among the Bacteroides fragilis group.25 As in the present study, antibiotic susceptibility for other antibiotics was not performed, the resistant patterns for other antibiotics could not be commented on. In another study, regarding Prevotella, resistance rate to penicillin was increasingly documented, and their resistance rate to clindamycin ranged from 11 to 40%.24,25 Their resistance rate to clindamycin ranged from 11 to 40%. Overall, no resistance to metronidazole was found in Prevotella isolates, although in some studies, this resistance has been detected.26,27 On the other hand, resistance to antibiotics has been found in very few isolates of Fusobacterium, except for penicillin due to beta lactamase production.27 Among Clostridium, C. difficile showed a high resistance rate to imipenem (90%) and clindamycin (40%) but no resistance to metronidazole.28 In a similar study, 4% metronidazole resistance was documented among the anaerobic bacteria (Bacteroides fragilis group).29 But they did not report any carbapenem (Imipenem and Meropenem) resistance among the anaerobic bacteria. In a recent study by Valdezate S et al., they documented Bacteroides fragilis strains which were resistant to meropenem.30 Among Clostridia, C. difficile showed a high resistance rate to imipenem (90%).31
In conclusion, as there is growing antimicrobial resistance among the obligate anaerobes, routine antimicrobial susceptibility testing for anaerobes has become essential and provides information regarding the antimicrobial resistance patterns and permits empirical therapies to be selected in accordance with local data on resistant strains.
ACKNOWLEDGMENTS
The authors would like to thank Department of Microbiology and Department of General Surgey for their immense support.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
This study was approved by Jipmer Ethics Committee, with reference number
JIP/IEC/2018/4653.
INFORMED CONSENT
Written informed consent was obtained from the participants before enrolling in the study.
- Menichetti F, Sganga G. Definition and Classification of Intra-abdominal Infections. J Chemother. 2009;21(Suppl 1):3-4.
Crossref - Marcus G, Levy S, Salhab G, et al. Intra-abdominal Infections: The Role of Anaerobes, Enterococci, Fungi, and Multidrug-Resistant Organisms. Open Forum Infect Dis. 2016:20;3(4):ofw232.
Crossref - Goldstein EJC. Intra-Abdominal Anaerobic Infections: Bacteriology and Therapeutic Potential of Newer Antimicrobial Carbapenem, Fluoroquinolone, and Desfluoroquinolone Therapeutic Agents. Clin Infect Dis. 2002;35(Suppl 1):S106–S111.
Crossref - Solomkin JS, Mazuski JE, Bradley JS, et al. Diagnosis and management of complicated intra-abdominal infection in adults and children: guidelines by the Surgical Infection Society and the Infectious Diseases Society of America. Clin Infect Dis. 2010;50(2):133-164.
Crossref - Tellor B, Skrupky LP, Symons W, et al. Inadequate source control and inappropriate antibiotics are key determinants of mortality in patients with intra-abdominal sepsis and associated bacteremia. Surg Infect. 2015;16(6):785-793.
Crossref - Flaherty SK, Weber RL, Chase M, et al. Septic shock and adequacy of early empiric antibiotics in the emergency department. J Emerg Med. 2014;47(5):601-607.
Crossref - Snydman DR. Empiric antibiotic selection strategies for healthcare-associated pneumonia, intra-abdominal infections, and catheter-associated bacteremia. J Hosp Med. 2012;7(Suppl 1):S2-12.
Crossref - Wacha H, Hau T, Dittmer R, Ohmann C: Risk factors associated with intraabdominal infections: a prospective multicentre study. Peritonitis Study Group. Langenbecks Arch Surg. 1999;384(1):24-32.
Crossref - Jousimies-Somer HR, Summanen PH, Citron DM, Baron EJ, Wexler HM, Finegold SM. Wadsworth anaerobic bacteriology manual (6th edn.), Star Publishing Company, Belmont, CA. 2002.
- M Sartelli. A focus on intra-abdominal infections. World J Emerg Surg. 2010;5:9.
Crossref - Brook I, Frazier EH. Aerobic and anaerobic microbiology in intr-abdominal infections associated with diverticulitis. J Med Microbiol. 2000;49(9):827-830.
Crossref - Shree N, Arora BS, Mohil RS, Kasana D, Biswal I. Bacterial profile and patterns of antimicrobial drug resistance in intra-abdominal infections: Current experience in a teaching hospital. Indian J Pathol Microbiol. 2013;56(4):388-392.
Crossref - Chia TC, Szu YL, Wei SY, Huei WC, Cheng CG, Chung JY. Peritonial dialysis peritonitis by anaerobic pathogens: Retrospective case series. BMC Nephrol. 2013;14:111.
Crossref - Lopez N, Kobayashi L, Coimbra R. A Comprehensive review of abdominal infections. World J Emerg Surg. 2011;6:7.
Crossref - Agarwal N, Saha S, Srivastava A, Chumber S, Dhar A, Garg S. Peritonitis: 10 years’ experience in a single surgical unit. Trop Gastroenterol. 2007;28(3):117-120.
- Hawser SP, Badal RE, Bouchillon SK, Hoban DJ; SMART India Working Group. Antibiotic Susceptibility of Intra Abdominal Infection isolates from Indian Hospitals during 2008. J Med Microbiol. 2010;59(Pt 9):1050-1054.
Crossref - Chen YH, Hsueh PR, Badal RE, et al. Antimicrobial susceptibility profiles of aerobic and facultative Gram-negative bacilli isolated from patients with intra-abdominal infections in the Asia-Pacific region according to currently established susceptibility interpretive criteria. J Infect. 2011;62(4):280-291.
Crossref - Lee Y, Park Y, Kim MS, et al. Antimicrobial susceptibility patterns for recent clinical isolates of anaerobic bacteria in South Korea. Antimicrob Agents Chemother. 2010;54(9):3993-3997.
Crossref - Gupta AS, Padmaja AK, Ajay CK. Clinico-microbiological profile of Bacteroides fragilis with focus on molecular detection of emerging resistance. Indian J Med Res. 2021:154(5):750-756.
Crossref - Ana P, Jozseph S, David P. Modulation of Iron Import and Metronidazole Resistance in Bacteroides fragilis Harboring a nimA Gene. Front Microbiol. 2022;13:898453
Crossref - Finegold SM. Anaerobic infections in humans: An overview. Anaerobe. 1995;1(1):3-9.
Crossref - Brook I, Wexler HM, Goldstein EJC. Antianaerobic antimicrobials: Spectrum and susceptibility testing. Clin Microbiol Rev. 2013:26(3):526-546.
Crossref - Karlowsky JA, Walkty AJ, Adam HJ, Baxter MR, Hoban DJ, Zhanel GG. Prevalence of antimicrobial resistance among clinical isolates of Bacteroides fragilis group in Canada in 2010-2011: CANWARD surveillance study. Antimicrob Agents Chemother. 2012;(56):1247.
Crossref - Cobo F. Antimicrobial Susceptibility and Clinical Findings of significant Anaerobic Bacteria in Spain. Anaerobe. 2019;(59):49-53
Crossref - Snydman DR, Jacobus NV, McDermott LA, et al. Update on resistance of Bacteroides fragilis group and related species with special attention to carbapenems 2006-2009. Anaerobe. 2011;17(4):147-151.
Crossref - Cobo F, Rodriguez-Granger J, Sampedro A, Navarro-Mari JM. Infected breast cyst due to Prevotella buccae resistant to metronidazole. Anaerobe. 2017;48:177-178.
Crossref - Shilnikova II, Dmitrieva NV. Evaluation of antibiotic susceptibility of Bacteroides, Prevotella and Fusobacterium species isolated from patients of the NN Blokhin cancer research center, Moscow, Russia. Anaerobe. 2015;31:15-18.
Crossref - Sartelli M, Chichom MA, Labricciosa FM, et al. The management of intra-abdominal infections from a global perspective: 2017 WSES guidelines for management of intra-abdominal infections. World J Emerg Surg. 2017;12:29.
Crossref - Fatma B, Fatih C, Nida O, Nezahat A. Identification and antimicrobial susceptibility testing of anaerobic bacteria isolated from clinical samples. J Bacteriol Mycol. 2020;8(1):29-32.
Crossref - Valdezate S, Cobo F, Monzon S, et al. Genomic background and phylogeny of cfiA-positive Bacteroides fragilis strains resistant to meropenem-EDTA. Antibiotics.2021;10(3):304.
Crossref - Cobo F. Antimicrobial Susceptibility and Clinical Findings of Anaerobic Bacteria. Antibiotics. 2022;11(3):351.
Crossref
© The Author(s) 2023. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.