ISSN: 0973-7510
E-ISSN: 2581-690X
Aromatic hydrocarbons contamination is widely prevalent in various parts of the world due to anthropogenic activities and leads to anaerobic conditions. As a result, most of its biodegradation is due to anaerobic microorganisms, and most specifically by anaerobic bacteria capable of using sulfates as final electron acceptor to degrade these compounds. Although there are reports on consortia of microorganisms that are involved in the anaerobic biodegradation of monoaromatic hydrocarbons (MAH) and polyaromatic hydrocarbons (PAH), only few reports are available using pure cultures. This paper describes an anaerobic, gram positive and spore forming bacterial strain (C1Fd), which was isolated and purified from aromatic compounds degrading consortium developed using bovine rumen fluid as inoculum. C1Fd was able to use MAH and PAH under anaerobic conditions and removed up 9.4 mM of MAH and 9.2 mM of PAH in less than 72 h. The strain was identified as Bacillus sp. and is phylogenetically related to the hydrocarbon degrading bacteria, Desulfotomaculum sp., isolated from a wastewater treatment plant.
Benzene, Naphthalene, Phenanthrene, Toluene.
Aromatic compounds are one of the major organic compounds that pollute soil, water and air and this is mainly attributed to oil and diesel spills (Becerril et al., 2008; Morlett et al., 2008a). Monoaromatic hydrocarbons (MAH) and polyaromatic hydrocarbons (PAH) are persistent compounds and remain in the ecosystem for a long time (EFSA, 2008), resulting in damage to the quality of ecosystem, agricultural crops, fishes, crustaceans and food (Agudo, 2010; Rubio et al., 2006). In addition, PAH can be formed during certain food processes as drying, cooking, smoking, grilling and frying (Ferrares et al., 2008; Martorell, et al., 2010; Alomirah et al., 2011). The presence of aromatic compounds in the environment and foods causes adverse effects on the health of humans and animals (Agudo, 2010; Rubio et al., 2006; Alomirah et al., 2011). Moreover MAH and PAH have been reported to be cytotoxic and genotoxic for humans (EFSA, 2008). An effective and economical solution to this problem is to use biotechnological tools for biodegradation of these compounds. Although biodegradation can be mediated under aerobic and anaerobic conditions or both, higher presence of sulfates in sediments and soils contaminated with PAH suggest the possibility of sulfate being the preferred terminal electron acceptor by anaerobic microorganisms (Lu et al., 2011). Previously anaerobic biodegradation of benzene, toluene, ethylbenzene, naphthalene, 2-methyl naphthalene, fluorene and phenanthrene, mediated by bacterial consortia under sulfate-reducing conditions have been documented (Musat and Widdel, 2008; Dou et al., 2009; Musat et al., 2009; Selesi et al., 2010; Chen et al., 2011; Mathava et al., 2011; Yan et al., 2012; Sun et al., 2014). Different sulfate reducing bacteria (SRB) have reported for the degradation of various hydrocarbons, such as Desulfosporosinus meridiei (Robertson et al., 2001), Desulfovibrio carbonoliphilus (Allen et al., 2008), Desulfotomaculum sp., Desulfomicrobium sp., Desulfobacter sp. and Desulfobulbus sp. (Guan et al., 2012). While most have been isolated from oil contaminated sites, interestingly Desulfotomaculum ruminis, was isolated from rumen of sheep (Coleman, 1960). In this regard, Spring et al., (2012) opined that this bacterium developed in the tract of ruminants because consumption of food contaminated with aromatic compounds. Studies on anaerobic bacteria capable of metabolizing PAH and MAH, will improve current strategies employed for bioremediation of MAH and PAH contaminated sites. The aim of this paper was to study the potential of a pure culture anaerobic bacterial strain isolated from an anaerobic consortia, developed from bovine rumen fluid to metabolize MAH and PAH under anaerobic and sulfate reducing conditions.
Collection and isolation of SRB
Anaerobic bacterial strain, C1Fd, was isolated from a consortium developed of bovine rumen that metabolized aromatic compounds such as phenol, catechol, 4-amino benzene, and p-phenylenediamine, under sulfate reducing conditions (Morlett et al., 2008b).
The mineral culture medium consisted of the following salts and solutions (l-1): K2HPO4, 0.5 g; NH4Cl, 1.0 g; Na2SO4, 4 g; MgSO4, 1.0 g; CaCl2, 0.1 g; yeast extract, 1.0 g; FeSO4, 0.1 g; cysteine HCl, 0.25 g; Na2SO3, 0.25 g; C6H8O6, 0.1 g; sodium lactate, 10.0 ml; ethanol, 1.0 ml; resazurin solution (0.1%), 0.1 ml; trace elements solution, 1 ml. The trace element solution contained (l-1): MgCl2·6H2O, 2.5 g; MnCl2·4H2O, 0.6 g; NaCl, 1 g; FeCl2·4H2O, 0.1 g; CoCl2·6H2O, 0.1 g; AlCl3, 0.01 g; H3BO3, 0.01 g; Na2MoO4·2H2O, 0.01 g; CuCl2·2H2O, 0.01g; CaCl2·2H2O, 0.1 g; ZnCl2, 0.1 g. The culture medium was prepared under nitrogen atmosphere; pH was adjusted to 7 and was sterilized in an autoclave at 121°C/15 min.
The colony isolated from consortium, was purified in solid medium by successive spread on sterile agar plates under anaerobic conditions, then this colony was transferred to reactors (100 ml) containing 50 ml of culture medium, under sterile conditions, 4-amino benzene was added and anaerobic (atmosphere of N2) sulfate reducing conditions. The purified bacterial strain was inoculated at 10% v/v, with cell concentration of 1 x 107/ml. The strain was maintained in mineral culture medium, with MAH and PAH as the sole source of carbon and energy, under the conditions described above.
Morphological and biochemical characterization of the strain isolate
Different bacterial traits such as: Gram character, sporulation color, opacity, form, elevation and consistency were evaluated. The biochemical characterization was carried out by microplate analysis using MicroScan AutoScan4.
Molecular identification of the strain
DNA extraction. The grown cells were centrifuged at 10625 x g/10 min, and the DNA was extracted from 0.1 g of wet biomass by using the modified CTAB protocol (Wagner et al., 1987). The integrity and concentration of DNA was evaluated, by agarose gel electrophoresis at 1% and was quantified based on the A260 and A280 nm using the program DNA quantification Gen5 version 1.11 (EPOCH Microplate Spectrophotometer, BioTek® Instruments).
Amplification of 16S rDNA gene. 16S rDNA gene of the extracted DNA was amplified using the primers 16S f 5´AGGAGGTGATCCAACCGCA3´ y 16S r 5´AACTGGAGGAAGGTGGGGAT3´ (Greisen et al., 1994). The final volume of the reaction was 25 µl, which contained: deionized water 14.5 µl, Buffer 10x/ MgCl3 50 mM 3.5 µl, dNTP´s 10 mM 0.5 µl, primers f and r 10 mM 2 µl, Taq polymerase paq5000 (Agilent Technologies) and template DNA 2 µl. PCR was performed in a thermocycler (Axygen Maxygen) by employing the following conditions: 1 cycle of initial denaturation at 95°C/5 min, 35 cycles of denaturation at 94°C/1 min, annealing at 54°C/1 min and polymerization at 72°C/1 min, 1 cycle of final polymerization at 72°C/5 min and was held at 4°C. Amplified PCR products were visualized on agarose gels 1.5%, by electrophoresis at 100 V/40 min in a Thermo EC105® chamber. Subsequently the gels were stained with ethidium bromide (5 µg/ml) and observed in UV transilluminator (Spectroline Ultraviolet Transiluminator Longliffet ®), and the bands were analyzed by UVP Mini Darkroom GDS-800 System software Labswork 4.5. Molecular weight marker 100 bp Axygen (Biosciences) was run along with the samples to determine the size of the amplicon.
Sequencing of 16S DNAr gene. Amplified product was sequenced by the method Taq FS Dye Terminator Cycle Sequencing Fluorescence- Based Sequencing in an automatic sequencer (Perkin Elmer of Applied Biosystem®; model 3730). Obtained sequences were aligned and analyzed using BLAST (Basic Logical Aligment Search Tool) of the database of NCBI (National Center for Biotechnology Information) of GenBank.
Growth curve of the strain
The growth of the bacterial strain C1Fd was determined in batch reactors (100 ml) containing 45 ml of mineral medium with glucose, as carbon source, under anaerobic, sulfate reduction conditions at 37°C. The reactors were inoculated with 10% (v/v) of strain at concentration of 1 x 107 cells/ml. Then, small aliquots were taken of the reactors and optical density (OD) readings at 600 nm were measured using a microplate in the EPOCH Microplate Spectrophotometer (BioTek® Instruments).
Anaerobic degradation of MAH and PAH
While benzene and toluene were tested as model compounds of MAH, naphthalene and phenanthrene were used as PAH model compounds in this study. Degradation kinetics of MAH and PAH by C1Fd strain were determined according to the conditions established; mineral medium as mentioned previously with 10 mM of benzene, toluene, naphthalene and phenanthrene as carbon source, SO4-2 as electron acceptor at 2.5 mM, pH 7 and at 37±1°C. Mineral culture medium was prepared under anaerobic conditions and in N2 atmosphere and was sterilized in an autoclave at 121°C/15 min (Morlett et al., 2008b) and subsequently the desired aromatic compound was added before inoculation (10% v/v at a concentration at 1 x 107 cells/ml). Batch reactors with aromatic compounds, but without addition of SO42- were used as negative control, while reactors with glucose and SO42- were used as positive control (Morlett et al., 2008a). The kinetics of the biodegradation of each of the test aromatic compounds of this study was determined periodically, viz, 0, 24, 48 and 72 h.
Analytical methods
Concentration of MAH and PAH during the degradation kinetics, was determined by a gas chromatograph (GC) fitted with Flame Ionization Detector (Varian Start 3600) and a capillary column CP Wax 52CB (N. CP8745) of 30 m x 0.25 mm x 0.15 µm. The column temperature was programed from 50 to 150°C, at the rate of 1°C/min, injector and detector temperature was 250 and 300°C, respectively. Helium was used as carrier gas (30 ml/min).
Morphological and biochemical characterization of the strain
C1Fd strain was isolated from a rumen fluid derived bacterial consortium degrading different aromatic compounds. Macroscopic nature of the colony and microscopic characteristics of the isolated bacterial strain is shown in Table I.
Table (1):
Macroscopic and microscopic characteristics of isolated SRB, C1Fd strain
| Characteristics | C1F d |
|---|---|
| Colony | |
| Color | pink |
| Form | round |
| Brightness | lustreless |
| Elevation | flat |
| Consistency | smooth |
| cell morphology | bacillus |
| Gram | + |
| sporeforming | + |
| Biochemical tests | |
| Glucose | + |
| Mannitol | + |
| Lactose | + |
| Trehalose | + |
| Mannose | – |
| Arabinose | + |
| Ribose | – |
| Pyruvate | + |
| Arginine | + |
| Indoxilphosphatasa | – |
| Phosphatasa | + |
| PNP-ß-D galactopyranoside | + |
| Voges proskauer | + |
| Bile Aesculin | – |
| NaCl 6.5 % | + |
| Novobiocin | – |
PNP: p-Nitrophenyl; NaCl: sodium chloride; (+) positive test; (-) negative test.
Biochemical tests demonstrated that the isolated bacteria strain is capable of growing on various substrates (Table I). Further, it was observed that the C1Fd strain recorded growth at high salt concentrations (6.5% of NaCl), and was in accordance to the previous report that SRB can grow under high salt conditions (Barton and Hamilton, 2007). In addition, the sensitivity of strain to the antibiotics; Clindamycin (0.5 µg/ml), Oxacillin (0.25 µg/ml) and penicillin (0.03 µg/ml) was observed.
Molecular identification of the isolated strain using 16S rDNA gene sequencing
For molecular identification of the strain C1Fd, a 400 bp amplicon (Fig. 1) obtained from the extracted DNA (100 ng/ml) under the conditions and by using the primers mentioned earlier.
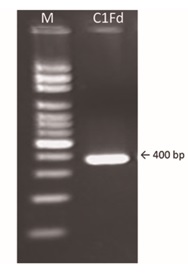
Fig. 1. PCR amplification of the 16S rDNA gene from C1Fd strain. Lane 1: molecular weight 100 bp marker (M); lane 2: amplicon of the SRB strain C1Fd on agarose gel 1.5%.
The obtained amplicon was purified, sequenced (GenBank access number is KT633556) and based on 97% similarity of sequence, the strain was determined to be Bacillus genus. A phylogram showing the phylogenetic relationship of Bacillus sp. C1Fd strain, with other sulfate-reducing bacteria was constructed by maximum-likelihood method using Mega5 Program (Fig. 2) and the phylogram was bootstrapped to 1000 times. It can be seen that the isolate of this study was grouped as a separate clade from the compared SRB. This suggested that this isolate is distant from many of the previously described SRB and further tests are necessary for identification at specie level. Besides, it can be observed that C1Fd strain is closely related to Bacillus sp. G9C (N. access AY062011), a clone directly related to the genus Desulfotomaculum sp. strain DSM 7440 (N. access Y11579).
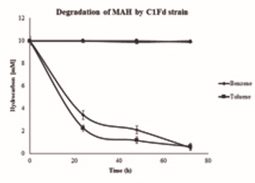
Fig. 2. Kinetics on degradation of toluene and benzene by CF1d strain. The bars in the line graph perpendicular to the time reflect the standard deviation of the measurements
Laban et al. (2009) reported that there are few Gram positive anaerobic bacteria which can degrade hydrocarbons and that these bacteria are closely related to the Desulfotomaculum genus. Additionally, the isolate due to its sporulation character can survive for longer periods in anoxic environments.
Anaerobic degradation of aromatic compounds
C1Fd strain growth was tested with different MAH for their potential to degrade these compounds. An insignificant lag phase was observed when C1Fd was grown on benzene and toluene (Fig. 3). It was also observed that benzene was resistant to bacterial degradation during the first 20 h and was found degraded later, but at lower rate than toluene until 72 h. These observations are in accordance with previously reports by Caldwell and Suflita (2000) and Coates et al., (2002); since the metabolism of aromatic compounds by sulfate reducing bacteria involve activation of the aromatic ring by methylation, hydroxylation or carboxylation. Thus, toluene having a methyl group is a molecule readily available for biodegradation, unlike the benzene, which first had to be modified before it is bioavailable. C1Fd strain removed 7.7±0.25 mM of toluene in 24 h, while the benzene removal during the same period was 6.57 ± 0.39 mM. However, there was no significant difference in the degradation of these aromatic compounds by C1Fd strain at 72 h (Fig. 3). This implies that the initial lag phase in degradation of benzene is only because of activation and later the strain was able to metabolize both compounds relatively at the same % of degradation (Foght, 2008; Carmona et al., 2009).
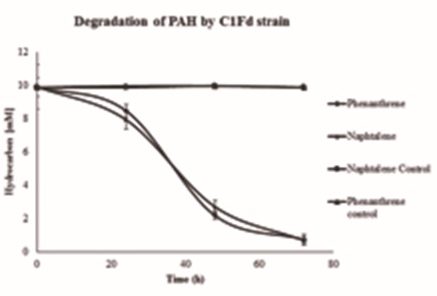
Fig. 3. Study the removal of naphthalene and phenanthrene (PAHs) by CF1d strain. The bars in the line graph perpendicular to the time reflect the standard deviation of the measurements.
Removal of benzene and toluene by C1Fd strain was on average 9.4±0.25 mM, in about 3 days. Previous reports on the degradation of benzene by different bacterial consortia under sulfate reducing conditions, demonstrated a removal from 9.3 ¼M to 0.32 mM between of 4 to 90 days or more (Kleinsteuber et al., 2008; Musat et al., 2008; Laban et al., 2009; Cervantes et al., 2011; Chen et al., 2011). In case of toluene, C1Fd strain removed 9.3±0.27 mM in three days. Earlier reports indicated that removal of toluene by bacterial consortia under sulfate reduction conditions ranged from 0.2 to 0.5 mM in a period of 4 to 200 days (Kazy et al., 2010; Chen et al., 2011; Fowler et al., 2012; Sun et al., 2014).
Degradation kinetics of phenanthrene and naphthalene by C1Fd strain are presented in Fig. 4. A lag period of about 24 h was recorded during the growth of C1Fd strain on both compounds. It must be noted that the C1Fd strain did not show a distinct lag phase when grown on both MAH compounds tested in this study. The strain degraded about 9.2±0.24 and 9.1±0.29 mM for naphthalene and phenanthrene respectively.
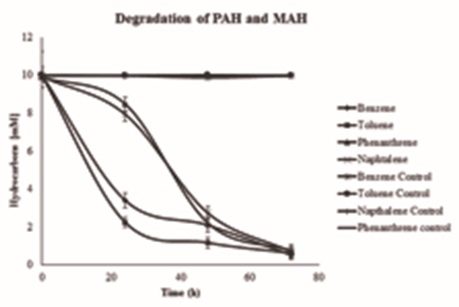
Fig. 4. Comparison of MAH and PAH removal by C1Fd strain. The bars in the line graph perpendicular to the time reflect the standard deviation of the measurements.
There are few reports on the biodegradation of PAHs by pure culture of SRB, In general, removal of PAH by bacterial consortia are widely reported. A removal of naphthalene from 0.22 to 5 mM was reported for periods between 20 to 60 days (Dou et al., 2009; Musat et al., 2009). While in the present work, a removal of 9.4±0.25 mM was found in a span of 3 days. In the case of degradation of phenanthrene by bacterial consortia, several authors have reported a time period of 3 to 240 days for the removal 0.28 mM (Tsai et al., 2009; Su et al., 2012; Yan et al, 2012.); while in the present work, a degradation of 9.13 mM was observed within 3 days by the pure culture of C1Fd.
The kinetics of removal of MAH and PAH by a sulfate reducing bacterial strain is shown in Fig. 5. The near absence of the lag phase during the growth of C1Fd strain was observed in the presence of MAH compounds tested in this study, while it was about 24 h for both tested PAH compounds. The duration of exponential phase was also clearly different for MAH and PAH compounds and this could be attributed to the complex nature of PAH, which requires activation of the molecule to increase its bioavailability for metabolism by C1Fd. However, there was no significant difference of removal was observed between MAH and PAH at 72 h.
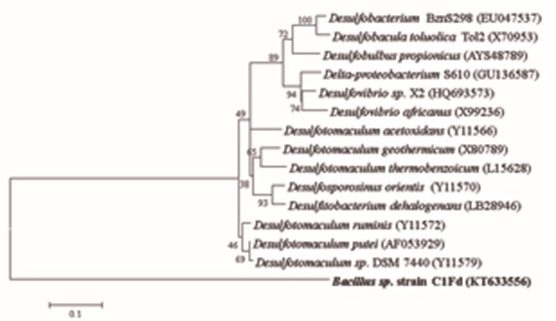
Fig. 5. Phylogenetic relationship of Bacillus sp. C1Fd (color bold) to different SRB strain based on the 16S rDNA gene sequences by the Maximum-likelihood analysis. The accession numbers of SRB are in parentheses.
Although several authors have reported the degradation of MAH and PAH by different microbial consortia, there are few reports on biodegradation of these compounds by pure culture strains, specifically by sulfate reducing bacteria (Singlenton et al., 2009; Tsai et al., 2009). Nevertheless, the potential of pure culture isolate of this study (C1Fd) to degrade MAH and PAH compounds almost at similar rate at 72 h is clearly demonstrated. These results clearly indicate the the metabolic capacities of Bacillus sp. C1Fd for potential applications in bioremediation of aromatic compounds. Characterization of enzymes from this strain is in progress to enhance its potential for bioremediation of hydrocarbon-contaminated sites.
ACKNOWLEDGMENTS
The authors thank the National Council for Science and Technology (CONACYT) of Mexico for the financial support through project N. 183204. First Author Sandra L. Villarreal Morales also thank CONACYT for awarding her scholarship for obtaining her Master’s degree in Science (Food Science and Technology). This publication was supported by PFCE-SEP 2016.
- Agudo A. 2010. Polycyclic aromatic hydrocarbons (PAH): approaching the problem as occupational hazard. Catalan Institute of Oncology and Department of Occupational Health and the Environment MCA-UGT. Edit UGT Confederal Executive Committee. Spain (in Spanish).
- Allen T.D., P.F. Kraus, P.A. Lawson, G.R. Drake, D.L. Balkwill and R.S. Tanner. Desulfovibrio carbinoliphilus sp. nov., a benzyl alcohol-oxidizing, sulfate-reducing bacterium isolated from a gas condensate-contaminated aquifer. Int. J. Syst. Evol. Micr., 2008; 58: 1313–1317.
- Alomirah H., S. Al-Zenki, S. Al-Hooti, S. Zaghloul, W. Sawaya, N. Ahmed and K. Kannan. Concentrations and dietary exposure to polycyclic aromatic hydrocarbons (PAHs) from grilled and smoked foods. Food Control. 2011; 22: 2028-2035.
- Barton L.L and W.A. Hamilton (eds). Sulphate-reducing Bacteria Environmental and Engineered Systems. Cambridge University Press. New York, USA 2007.
- Becerril S.M., N.L. Tello, J.A Morlett and N. Balagurusamy. Chapter 7 Microbiology and anaerobic metabolic pathaways of degradation of xenobiotics compounds, pp. 126-142 .In Proteomes and Genomes of XIX Century: Environmental Biotechnology. Rodriguez R., O. Soto, J.L. Martínez and C.N. Aguilar (eds). University Autonomous of Coahuila, Mexico (In Spanish) 2008.
- Caldwell M.E. and J.M. Suflita. Detection of phenol and benzoate as intermediates of anaerobic benzene biodegradation under different terminal electron-accepting conditions. Environ. Sci. Technol. 2000; 34: 1216–1220.
- Carmona M., M.T. Zamarro, B. Velázquez, R.G. Durante, J.F. Juárez, J.A. Valderrama, M.J. Barragán, J.L. García. and E. Díaz. Anaerobic catabolism of aromatic compounds: a genetic and genomic view. Microbiol. Mol. Biol. R., 2009; 73(1): 71–133.
- Cervantes F.J, A.R. Mancilla, T.E.E. Ríos, S.A.G. Alpuche and L. Montoya-Lorenzan. Anaerobic degradation of benzene by enriched consortia with humic acids as terminal electron acceptors. J. Harzard. Mater. 2011; 195: 201- 207.
- Chen L., F. Liu, Y.L. Liu, H.Z. Donga and P.J.S. Colberg. Benzene and toluene biodegradation down gradient of a zero-valent iron permeable reactive barrier. J. Harzard. Mater. 2011; 188: 110- 115.
- Coates J.D., R. Chakraborty and M.J. McInerney. Anaerobic benzene biodegradation – a new era. Res. Microbiol., 2002; 153: 621–628.
- Coleman G.S. A sulphate-reducing bacterium from the sheep rumen. J. Gen. Microbiol., 1960; 22: 423-436.
- Dou J., X. Liu and A. Ding. Anaerobic degradation of naphthalene by the mixed bacteria under nitrate reducing conditions. J. Hazard. Mater, 2009; 165: 325–331.
- EFSA. Polycyclic aromatic hydrocarbons in food scientific opinion of the panel on contaminants in the food chain (Question No. EFSA-Q-2007-136). EFSA Journal. 2008; 724: 1–114.
- Ferrares E., G. Andreottola and I.A. Oprea. Remediation of PAH contaminated sediments by chemical oxidation. J. Hazard. Mater, 2008; 152: 128-139
- Foght J. Review anaerobic biodegradation of aromatic hydrocarbons: pathways and prospects. J. Mol. Microbiol. Biotechnol., 2008; 15: 93–120.
- Fowler S.J., X. Dong, C.W. Sensen, J.M. Suflita and L.M. Gieg. Methanogenic toluene metabolism: community structure and intermediates. Environ. Microbiol, 2012; 14: 754–764.
- Greisen K., M. Loeffelholz, A. Purohit and D. Leong. PCR primers and probes for the 16S rRNA gene of most species of pathogenic bacteria, including bacteria found in cerebrospinal fluid. J. Clini. Microbiol, 1994; 32(2): 335-351.
- Guan J., L.P. Xia, L.Y. Wang, J.F. Liu, J.D. Gu and B.Z. Mu. Diversity and distribution of sulfate-reducing bacteria in four petroleum reservoirs detected by using 16S rRNA and dsrAB genes. Int. Biodeterior. Biodegr, 2013; 76: 58-66.
- Kazy S.K., A.L. Monier and P.J.J. Alvarez. Assessing the correlation between anaerobic toluene degradation activity and bssA concentrations in hydrocarbon-contaminated aquifer material. Biodegradation, 2010; 21: 793–800.
- Kleinsteuber S., K.M. Schleinitz, J. Schleinitz, H. Harms, H.H. Richnow and C. Vog. Molecular characterization of bacterial communities mineralizing benzene under sulfate-reducing conditions. FEMS Microbiol. Ecol, 2008; 66: 143–157.
- Laban N.A., D. Selesi, C. Jobelius and R.U. Meckenstock. Anaerobic benzene degradation by Gram-positive sulfate-reducing Bacteria. FEMS Microbiol. Ecol, 2009; 68: 300–331.
- Lu X.Y., T. Zhang and H.H.P. Fang. Bacteria-mediated PAH degradation in soil and sediment. App. Microbiol. Biot, 2011; 89(5): 1357-1371.
- Martorell I., G. Perelló, R. Martí-Cida, V. Castell, J.M. Llobet and J.L. Domingo. Polycyclic aromatic hydrocarbons (PAH) in foods and estimated PAH intake by the population of Catalonia, Spain: Temporal trend. Environ. Int., 2010; 36: 424–432.
- Mathava K., L. Wei-Chin, T. Jen-Chieh and L. Jih-Gaw. Versatility of fluorine metabolite (phenol) in fluorine biodegradation by sulfate reducing culture. Int. Biodeter. Biodegr., 2011; 65: 522-526.
- Morlett J.A., R. Rodríguez, H. Barrera and N. Balagurusamy. 2008a. Chapter 8 Relationship between nitrate-, sulfate reducing bacteria and methanogens in anaerobic biodegradation of aromatic compounds, pp. 143- 156. In Proteomes and Genomes of XIX Century: Environmental Biotechnology. Rodriguez R., O. Soto, J.L. Martínez and C.N. Aguilar (eds). University Autonomous of Coahuila, Mexico (In Spanish).
- Morlett J.A., N. Balagurusamy and M.J. Rodríguez. Testing the efficiency of biofilm formed on natural substratum, Coyonoxle, Opuntia imbricata for anaerobic degradation of aromatic compounds. Eng. Life. Sci., 2008b; 8(4): 425-430.
- Musat F. and F. Widdel. Anaerobic degradation of benzene by a marine sulfate-reducing enrichment culture, and cell hybridization of the dominant phylotype. Environ. Microbiol., 2008; 10(1): 10-19.
- Musat F., A. Galushko, J. Jacob, F. Widde, M. Kube, R. Reinhardt, H. Wilkes, S. Schink and R. Rabus. Anaerobic degradation of naphtalene and 2-methylnaphthalene by strains of marine sulfate-reducing bacteria. Environ. Microbiol., 2009; 11: 209-219.
- Robertson W.J., J.P. Bowman, P.D. Franzmann and B.J. Mee. Desulfosporosinus meridiei sp. nov., a spore forming sulfate reducing bacterium isolated from gasoline-contaminated groundwater. Int. J. Syst. Evol. Micr., 2001; 51: 133–140.
- Rubio A.C., M.R. Álvarez and T.A. Hardisso. Polycyclic aromatic hydrocarbons (PAHs) in fishery products: Review (In Spanish). Rev. Toxicol., 2006; 23(001): 1-6.
- Selesi D., N. Jehmlich, M. Von Bergen, F. Schmidt, T. Rattei, P. Tischler, T. Lueders and R.U. Meckenstock. Combined genomic and proteomic approaches identify gene clusters involved in anaerobic 2-methylnaphthalene degradation in the sulfate-reducing enrichment culture N47. J. Bacteriol., 2010; 192(1): 1295–306.
- Singleton D.R., R.L. Guzman and M.D. Aitken. Characterization of a polycyclic aromatic hydrocarbon degradation gene cluster in a phenanthrene-degradation Acidovorax strain. Appli. Environ. Microbiol., 2009; 75(9): 2613-2620.
- Spring S., M. Visser, M. Lu, A. Copeland, A. Lapidus, S. Lucas, J-F. Cheng, C. Han, R. Tapia, L.A. Goodwin and others. Complete genome sequence of the sulfate-reducing firmicute Desulfotomaculum ruminis type strain (DLT). Stand. Genomic. Sci., 2012; 7: 304-319.
- Su L., H. Zhou, G. Gou, A. Zhao and Y. Zhao. Anaerobic biodegradation of PAH in river sediment treated with different additives. Procedia. Environ. Sci., 2012; 16: 311–319
- SunW., X. Dun and A.M. Cupples. Identification of Desulfosporosinus as toluene-assimilating microorganisms from a methanogenic consortium. Int. Biodeterior. Biodegrad., 2014; 88: 13-19.
- Tamura K., D. Peterson, N. Peterson, G. Stecher, M. Nei and S. Kumar. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol., 2011; 28(10): 2731- 2739.
- Tsai J.C., M. Kumar and J.G. Lin. Anaerobic biotransformation of fluorene and phenanthrene by sulfate-reducing bacteria and identification of biotransformation pathway. J. Hazard. Mater., 2009; 164: 847- 855.
- Wagner D. B, N. Song, H. Cai, J.H. Tay and H. Jiang. Enhanced degradation of phenanthrene and pyrene in freshwater sediments by combined employment of sediment microbial fuel cell and amorphous ferric hydroxide. J. Hazard. Mater., 2012; 199-200: 217-225.
- Yan Z., G.R. Furnier, M.A. Saghay-Maroof, S.M. Williams, B.P. Dancik and R.W. Allard. Chloroplast DNA polymorphisms in lodgepole and jack pines and their hybrids. Proc. Natl. Acad. Sci. USA., 1987; 84(7): 2097–2100.
© The Author(s) 2017. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


