Nontyphoidal Salmonella infections are common and remain a significant public health problem. The increasing cases of non-typhoidal Salmonella confirmed cases in past years from the United States, Europe, and low-income countries indicated that control programs aimed at inhibiting the contamination of food animals along the food chain had been largely ineffective. The majority of non-typhoidal Salmonella infections are mild gastroenteritis that rarely demands antimicrobial treatment. Salmonella is a gram-negative, multidrug-resistant, facultatively anaerobe bacterium. Salmonella is composed of approximately 2500 serotypes among which Salmonella enterica is responsible for more than 99% of human salmonellosis and morbidity. Salmonella enterica is an enteropathogenic foodborne bacteria isolated from animals that are responsible for zoonotic infections in humans, animals, and birds. Children who are young or immunocompromised, as well as those with underlying health issues like sickle cell disease, are highly susceptible to invasive disease. Antimicrobial treatment might be needed in case of invasive infections like meningitis, bacteremia, and osteomyelitis. Resistance to third-generation cephalosporins and fluoroquinolones in non-typhoidal Salmonella is a worrying concern in the 21st century. This review specifies various non-typhoidal Salmonella serotypes infections and their cases.
Antibacterial, Bacteraemia, Gastroenteritis, Immunocompromised, Osteomyelitis
Salmonella terminology is immensely complicated and has been a concern of immense controversy for many years. In the end, estimating the species and subspecies that represent the Salmonella genus is incredibly challenging.1 Facultative cellular anaerobic characteristics and Gram-negative rod nature of Salmonella enterica induce 1.3 billion illnesses year after year. It is representative of the Enterobacteriaceae family. Salmonella is an enteroinvasive bacterium that, when consumed, dodges gastric protection, multiplies, and breaches the mucosal surfaces of the intestinal epithelium. Intense gastroenteritis infection is the prevalent type of Salmonella infection. The period of incubation might variegate between 4-72 hours after consuming deteriorated food and water too. Critical bacterial fever & chills, vomiting, seasickness, diarrhoea, and abdominal discomfort are perhaps the most typical signs and symptoms. If fever occurs it normally dwindles within 72 hrs. Diarrhoea with blood typically limits itself and lasts for 3-7 days. This bacterium is removed from the body through faeces after inflammation, which can take up to 5 weeks. In youngsters, excretion may be prolonged. Salmonella excretion that lasts for more than 8 weeks after the infection is unusual in young patients and adults. The optimal amount of bacteria required to make a healthy individual ill is 106 – 108 non-typhoidal Salmonella.
To date, approximately 2500 unique serotypes or serovars of Salmonella bongori and Salmonella enterica have already been found. Salmonella is a commonly encountered and persistent which may persist for several weeks or months in dry and moist situations. Although all serotypes induce illness in people, some are specific to a particular host and can only be identified in either one or some species of animals, which includes Salmonella enterica serotype Dublin in cattle and Salmonella enterica serotype Choleraesuis in pigs. Generally, such serotypes induce gastroenteritis, which is typically benign and does not require medical attention; nevertheless, sickness could be acute in youngsters, aged people, and compromised immune systems individuals. Salmonella enterica serotype Enteritidis and Typhimurium, are the prevalent Salmonella serotypes transmitted to humans from infected animals in various regions around the globe. It is typically distinguished by an immediate onset of acute fever, abdominal discomfort, diarrhoea, uneasiness, and in rare cases vomiting. Humans get infected with non-typhoidal Salmonella infections by coming in contact with different farm animals, pets, reptiles, and broilers. 2
Salmonella is perhaps the most frequently isolated pathogen after Campylobacter when a presumptive diagnosis of diarrhoea is sought. The well-known Salmonella enteric serovar Heidelberg strain is ubiquitous and is regularly acquired from victims of salmonellosis in the North American region.3 Salmonella can thrive on plants; thus, fruits and vegetables could be a way for Salmonella to enter the food chain.4 NTS infection in animal-based foods occurs as a consequence of contamination in the feed or even the environment.5,2 It is the most predominant bacterial source of food contamination across the globe.6 Non-typhoidal salmonellosis is probably minor; however, in youngsters, the aged, and individuals with disabilities, it can cause life-risking consequences. The extent of NTS infection is determined by the host immune response, Salmonella serotype, and infective dosage. There is a matter of discussion that the evolution of Salmonella has not been properly researched to explain various complications within or across subspecies, and hence the ability to cause foodborne sickness or even more serious invasive illness remains unexplored.7
In the study of Taliha et al.8 three patients were detected with invasive non-typhoidal Salmonella infections in which one patient was suffering from a urinary tract infection and spondylodiscitis which was given medical treatment and surgery. The second patient was detected with an abdominal prosthetic aortic aneurysm which was given long-term antimicrobials. The third patient was detected with a thoracic aortic aneurysm and cutaneous abscesses which were treated with antimicrobials and surgery. For future research, the study suggests application of antimicrobials in the field of agriculture and veterinary should be encouraged to avoid the emergence of antimicrobial resistance. A vaccine against NTS that is both effective and safe is anxiously awaited.
Recent Study of Marchello et al.9 provided current information on the fatalities and complications related to the global burden of non-typhoidal Salmonella invasive disease, including in regions lacking up-to- date and strong national research data. A lacuna of this study is the inability to investigate the relationship between non-typhoidal Salmonella invasive disease and other comorbid conditions besides HIV and malaria.
Based on the findings of Rule et al.10 the patient was detected with acute bacterial meningitis. To treat the most common causes of bacterial meningitis, empiric antibiotic therapy with intravenous ceftriaxone was given. To minimize intracranial inflammation and avoid long-term neurological sequellae, dexamethasone intravenous corticosteroid therapy was incorporated. However, a limitation of the approach was the incapability to provide handling clinicians with quick microbiological results. Due to this conventional culture was conducted on this patient’s CSF, resulting in a four-day turnaround time before final identification.
In the study of Albert et al.11 NTS bacteremia was detected in 30 of 53,860 and 31 of 290,36 blood cultures in two hospitals of Kuwait. 13 different serovars were cultured in both hospitals and the majority of the cases were due to S. Enteritidis (all sequenced type (ST11) and S. Typhimurium (all ST19). Ampicillin, ciprofloxacin and tetracycline were the most prominent antibiotics and showed 36.1% to 50.8% prevalence. Limitations showed that the hospitalized cases didn’t represent community cases where a wide range of the severity of cases can occur, and the community population at risk was not defined clearly. Therefore polymerase chain reaction (PCR) and conventional blood cultures methods may not be sensitive enough to recognize all true positive cases.
The study investigated by Sanni et al.12 stated that 150 samples were accumulated from three local areas of government in every five states of the North Central zone of Nigeria. Besides of Salmonella enterica, Klebsiella pneumoniae was found in 92.9% of the specimens, Salmonella arizonae in 0.2%, Lactobacillus bulgaris in 0.9%,
S. paratyphi in 1.9% and S. typhi in 2.3% of all specimens. Mixed infections with Lactobacillus bulgaris, S. arizonae, Klebsiella pneumoniae, and S. paratyphi were reported in 392 of the 416 S. enterica-positive specimens (94.5%). There were some limitations in this work. Firstly no complete serotyping of all categorised confirmed cases was conducted, as this could have revealed all of the Salmonellae serotypes collected during the 18-month period. Secondly, many laboratories were used to ascertain NTS positivity, and not all farm cases were forwarded for laboratory analysis, among which some may have been salmonellosis. This resulted in misclassification which may have increased or decreased the overall prevalence calculated in the study. The objective of the present study is to give a detailed comprehensive review of non-typhoidal Salmonella infections and their consequences in humans.
Non-typhoidal Salmonella antimicrobial-resistant property
Antimicrobial resistance condition is a worldwide issue in the management of human and animal health care. Assessments of AMR pathogen transmission between humans and animals might provide insight into an essential pathway through which AMR pathogens invade humans. However, determining the transmitting path can be difficult (either pass on from animals to people or people to animals). Furthermore, AMR Salmonella illnesses exacerbate the situation by increasing the likelihood of blood infections due to treatment inefficacy.13 Although salmonellosis from pets has been proposed as a potential AMR transmission pathway, there is a lack of concrete data for this fact.14 Pet dogs can get Salmonella via ingestion of contaminated food or coming into contact with infectious waste or other diseased animals. Humans might be infected because of getting in contact with a diseased dog. According to one investigation, 107 (84 per cent) of the 128 Salmonella-infected individuals previously had interacted with dogs before becoming ill.15
Intrinsic and iatrogenic susceptibility of the host
Salmonellosis of the gastrointestinal system and its chronic complications are linked to a broad range of ailments and therapies that are mainly accountable for the several defence mechanisms against intestinal and intracellular pathogenic organisms. Hypoacidity in newborns, pernicious anaemia, or brought about by antacids and H-2 blockers can make people more susceptible to salmonellosis. Multiple perilous aspects can lead to salmonellosis which includes changes in the endogenic intestinal microbiota, rheumatological illnesses, diabetes, tumours, and reticuloendothelial blockade because of sickle-cell disorder, malaria, and bartonellosis. Therapeutic immunosuppression of all types and HIV infection are major reasons for salmonellosis. Various anatomical disruptions, such as UTIs, atherosclerotic endovascular lesions, kidney stones, gallstones, schistosomiasis, and prosthetic devices, could be the sole cause of persistent Salmonella disease.
Non-typhoidal Salmonella infections of key serotypes
Reptiles, poultry, and amphibians are widely known reservoirs for Salmonella spp. disease.16 However, in a few patients, the causative agent is unknown.17,18 According to a Chinese investigation conducted between 2005-2011, S.newport was among the ten leading serovars involved in clinical patient trials.19 Salmonellosis is a complicated disorder having a myriad of causes influencing death rates. Various serotypes appear to be related to specific spreading channels according to their roles in naturally occurring ecological systems.20 Salmonella Enteritidis was mostly linked to the ingestion of turkey, eggs, chicken, and sprouts, while Salmonella Typhimurium was linked to beef, dairy products, pork, and vegetables, while S. Newport to fruits & veggies.21 Although these are self-limiting, zoonotic infections can also induce irritable bowel syndrome problems, ulcerative colitis illness, and Crohn’s disease too, as well as extra-intestinal consequences such as rheumatoid arthritis, Guillain-Barre, and reactive arthritis.22
S. Heidelberg promotes many invasive human illnesses, such as dilated cardiomyopathy and septicemia.23 The condition can sometimes present as a feverish invasive disease, typically without diarrhoea, with bacteremia, meningitis, or focal disorders that can be fatal if left untreated or incorrectly managed.24-26 In 2017, the disease accounted for an estimated 535000 non-typhoidal Salmonella invasive illnesses and 77500 fatalities.27 Among six subspecies of S. enterica the foodborne illnesses are most often observed in 1 subspecies, notably S. enterica subspecies I. Some other S. enterica subspecies are II (salamae), IIIa (arizonae), IIIb (diarizonae), IV (houtenae), and VI (indica). It is very rare when subspecies II-IV lead to disease, but they hardly seem like people’s clinical care hazards and are also correlated with professions comprising wildlife, animals, and reptiles that are rarely eaten.28-30
Selection for just the fittest Salmonella enterica subspecies I serotype able to produce food-related ailments is obvious in a particular subspecies. It’s also known that many S.enterica serotypes can induce sickness under ideal circumstances; hence, all food and feedstuff components must pass quality control tests for humans and domestic animals. In the United States annually, very few serotypes, principally S. infantis, S, typhimurium, S. newport, S. enteritidis, and S. javiana, are the reason for 80% of reported salmonellosis cases.31,32 Microbiological studies demonstrated a higher prevalence of S.newport,
S. Mississippi, and S. javiana in western Tennesee countries in comparison to all other Tennessee countries.33 Surprisingly, a similar pattern was documented in Louisiana, as per the credentials of the Louisiana Office of Public Health.34 Salmonella type III and type IV secretory systems are some of its major virulence factors (T3SSs, T$SSs). These key Salmonella virulence factors influence host cell physiology to promote illness.35
Non-typhoidal serotypes, like S. enteritidis and S. Typhimurium, are commonly encountered serotypes linked to foodborne outbreaks. They have enormous epidemiologic importance because they survive in food production environments and have the tendency to expand extensively and fast throughout communities. Contamination of domestically marketed ice cream, chocolate cake, and smoked salmon outbreaks involving cucumbers are a few instances.36-39 Non-typhoidal Salmonella species are associated with 550 million diarrheal diseases around the globe.40 NTS can reside in infected individuals for extended periods without showing clinical manifestations.41 Asia’s Southeast region is a key hub for salmonellosis caused by non-typhoidal Salmonella. As per the data, this region accounted for 20.69 per cent of cases and 26.93 per cent of deaths of whole world non-typhoidal salmonellosis.42
Infections of the gastrointestinal tract instigated by non-typhoidal Salmonella
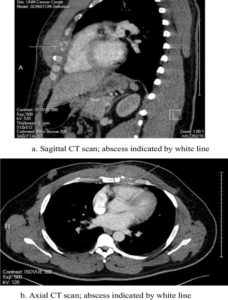
Gastroenteritis is the foremost reason for morbidity in children globally, particularly in developing nations, accounting for roughly 1.87 million deaths in children under the age of five per year.43 It typically begins 12-38 hours after the pathogen is consumed, with cramping of the abdomen, nausea, diarrhoea, and 100.5-102.2 °F fever. Although the faeces is runny, it also has a paste-like mucilaginous consistency and contains a modest amount of polymorphonuclear leukocytes and occult blood. Stool culture continues to remain positive after 4 to 5 weeks also. Mild leucocytes could be found. Around 70% of patient populations will experience fever. Tenderness is discovered during an abdominal investigation. Typically, the ailment is relatively benign and last for 1-4 days. Many serious, long-term illnesses occur frequently. When diarrhoea stops, reactive arthritis affects 10-30% of adult human populations and can last for several weeks or months. This condition causes pain & inflammation in the knees, hips, and Achilles tendon also. Salmonella enteritis may progress more slowly in susceptible hosts. Preterm infants, premature children, and children with primary/secondary immune deficiency may experience problems for several weeks. Salmonella enteritis can induce toxic megacolon formation, bowel incursion, systemic toxicity, and even death in sick individuals with inflammatory bowel disorders, especially active ulcerative colitis. In immunocompromised adults, Salmonella gastroenteritis is a self-limiting infectious disease. A case report of Castlemain et al.44 stated that a healthy man 39-year-old who was previously successfully treated with Salmonella gastroenteritis and bacteremia was found Salmonella Oranienburg positive. S. Oranienburg is a serovar responsible for the current gastroenteritis breakout linked to interaction with a pet turtle. Figures 1a and b showed a chest CT scan of a patient infected with S.Oranienburg.
Non-typhoidal Salmonella extraintestinal focal infections (EFI)
NTS extraintestinal focal infections are entirely unusual and have unknown danger variables. Pleuropulmonary infection, spinal osteomyelitis, and mycotic aneurysm are all some examples of EFIs.45 When possible, these infections must be depleted. For the cure of a medically eliminated focus of soft tissue in a normal host, a minimum period of 14 days of antimicrobial prophylaxis is recommended. Because Salmonella species are known to persist in polluted areas, treatment for 4-6 weeks is usually recommended. Osteomyelitis and joint-related infections are more refractory to treat in sickle-cell anaemic individuals. Bacteremia or focal infection tends to affect 8% of people (which are septic arthritis, osteomyelitis, and meningitis). Focal infections, most prevalently in the bones and central nervous system, can happen in newborns (particularly those under 3 months old) and immunocompromised children.46 However, NTS disease is linked to a variety of illnesses besides enteritis, with a broad range of severity in particular risk groups like extraintestinal manifestations (meningitis, osteomyelitis, deep soft tissue, septic arthritis, pneumonia, and bacteremia.
Bacteremia and Endovascular Disorder: Intense Complications
Bacteremia is much more likely to happen in immunocompromised hosts. Bacteremia, a life-threatening condition, affects about 5% of people who have a gastrointestinal disorder resulting from non-typhoidal Salmonella. Non-typhoidal Salmonella bacteremia is more noxious. S. Choleraesuis, S. Heidelberg, and S. Typhimurium, on the other hand, can normally cause a detrimental bacteremia syndrome which can last for 1 week and is characterized by a protracted fever, malaise, headache, and shivers but scarcely diarrhoea. Salmonella may cause recurrent bacteremia or other invasive infections like septic arthritis in hosts. In adults, the progression of infective endarteritis, particularly embroilment of the abdominal aorta, is a frightening contraindication to Salmonella bacteremia.
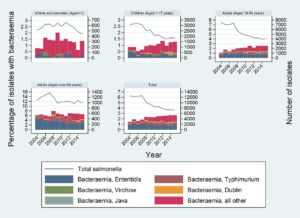
Bacteremic Salmonella is typically treated with a particular antimicrobial agent, such as third-generation cephalosporin, quinolone 2001, and ampicillin. Figure 2 depicts total NTS instances over a specific period by age category and proportion of infectious bacteremia infection.47 Bacteremia is the most widely known systemic infection, with the occurrence of Salmonella enteritis ranging from 2% to 47% depending on the region.48 Despite the overall reduction in salmonellosis incidence during the last five years due to better hygiene, Salmonella persists as the second leading source of bacteremia in youngsters between the age of 3-59 months, as per the largest multicenter study on pediatric invasive bacterial infections in South Korea.49
Figure 2. The Overall number of non-typhoidal Salmonella reported cases by age category, proportion to bacteremia, and the five major serovars.47
Non-typhoidal Salmonella: Reason behind meningitis in children with immune deficiency
A leading source of chronic bacterial meningitis in newborns and infants in developing countries is non-typhoidal Salmonella. Abscesses, subdural empyema, Seizures, and hydrocephalus are common complications of NTS meningitis in youngsters, and children may experience serious delayed development and impairment. Meningitis caused by NTS has an elevated mortality rate and long-term neurologic disorders.50 Meningitis caused by NTS is much less common, and the exact prevalence is uncertain. Little research on meningitis caused by NTS or S. enterica ser. Typhi in children has already been publicly released, and most of them involved infants and malfunctioned children. All three pediatric sufferers were given meropenem to treat multidrug non-typhoidal Salmonella, and there was a significant improvement. Even after antibiotic therapy, the survival rate of NTS meningitis can range from complete recovery to fatality, and neural complications like epilepsy, focal problems, and cerebral palsy usually occur.51 Various inflammation peaks were detected in youngsters of age five and adults fifteen. In 196 sick people, NTS had been secluded from the cerebrospinal fluid, and in 82 sick people from feces, blood, or any other body spots. During the period, the mean rate of infection of NTS meningitis was 5 per 10000000 annually as shown in Figure 3.52
Figure 3. From 2003 to 2013, there were 256 incidents of nontyphoidal Salmonella meningitis [n=256]. B) Total NTS meningitis incidents (n=111) by category of age and morbidity from 2003 to 2013. Single patients who retrieved had no age registered. C) Total NTS meningitis instances (n=91) by category of age and condition of HIV, 2003-2013. The age of a single HIV-diagnosed host was not registered.52
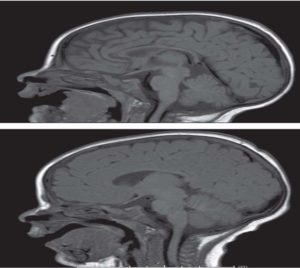
Because NTS meningitis has an elevated morbidity & mortality rate, it is pivotal to administrate relevant antibiotics as soon as possible. Adult HIV-positive people are at a significantly greater risk of getting salmonellosis, which can progress to severe complications like meningitis even after antibiotic prophylaxis.53 Third-generation cephalosporins, luckily are frequently used as an early empirical antimicrobial prophylaxis in children with suspected bacterial meningitis. In a report by Monica et al.,54 a two-month-old Italian boy infant was admitted as he was suffering from loss of appetite, elevated fever, irritability, and blood traces were present in diarrhoea when investigated showed the presence of Salmonella enteritidis. Brain ultrasound confirmed periencefalic purulent suffusion, which was affirmed by brain imaging technique i.e., MRI (Figure 4a). A third brain MRI indicated some re-absorption of the frontal collection at the end of the prophylactic antibiotic treatment (Figure 4b). Adult meningitis has also been linked to S. Virchow.55
Figure 4. In a report of two month old boy who suffered from Salmonella enteritidis, the results of the first brain MRI, which were supported by the cerebral ultrasound, showed periencefalic purulent suffusion (fig 4a) and a last brain MRI after the antibiotic treatment ended showed a slight resorption of the frontal accumulation (fig 4b)
Complications of osteomyelitis caused by Non-Typhoidal Salmonella
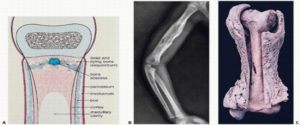
Vertebral osteomyelitis is a very infrequent NTS complication with an undiscovered global prevalence.56 Salmonella osteomyelitis, an unusual typhoid infection, can happen because of hematogenous damage to the joints and bones caused by a bacterial episode. It can also be dispersed from infected areas nearby or through penetrating trauma. Distress in the particular bone which has upper redness, elevated body temperature, and paleness are possible symptoms. Diagnosis and treatment must be done as soon as possible because if not, it can result in long-lasting functional impairment afterwards. Salmonella osteomyelitis can be seen infrequently in hospitals, accounting for 0.8 per cent of all infections caused by Salmonella and only 0.45 per cent of overall osteomyelitis are triggered by NTS, primarily in sickle cell anaemic hosts.57 Capillary obstruction by intravascular sickling might desiccate and siege the gastrointestinal tract in patients with sickle cell anaemia, allowing Salmonella bloodstream incursions and raising the risk of osteomyelitis. A role for the complement system and impaired opsonization has also been proposed.58,59 In immunocompromised individuals with no hemoglobinopathy, non-typhoidal Salmonella osteomyelitis is unusual. Surgical debridement and long-term antibiotics are part of the healing process. In the United States, out of 0.04 per cent, 51,964 cases of NTS infections remained closely linked to bone and joint illnesses. It has also been related to other scenarios such as connective tissue disorders, prior trauma, and immunocompromised conditions such as cancer malignancies, (Figure 5).60
Figure 5. Osteomyelitis. Within the bone, an infection nidus disperses through the medullary (cancellous) and cortical bone, causing periosteal growth of new bone [A]. The diseased bone finally dies (sequestrum) and is surrounded by a shell of periosteal new bone (involucrum). Roentgenogram of the humerus [B]; remember that the whole femur is necrotic, with extensive new periosteal bone wrapping and forming a new shell contour in a gross femur of a cow [C] (involucrum)60
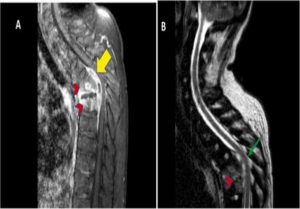
Patients suffering from sickle cell illness are prone to acquire osteomyelitis. Osteomyelitis clinical manifestations in sickle cell illness are equivalent to vasooclusive crisis, with little or no diagnostic standard. It has been reported in the study of Weisman et al.61 that twenty-eight children who were suffering from sickle cell disease were treated for osteomyelitis. Based on the results of MRI, and other clinical diagnoses revealed that osteomyelitis was confirmed in 3 patients, and the probability was in 6 patients and assumed in 19 patients. Another sickle cell illness sufferer mentioned in Elnour et al.62 study needed surgery to cure vertebral-related osteomyelitis and an epidural inflammation, healed by 4 weeks of IV ceftriaxone treatment, and 2 weeks of trimethoprim/sulfamethoxazole treatment. This increased his chances of contracting an intricate Salmonella infection (Figure 6).
Figure 6. A contrast MRI of the spine revealed focal spondylodiscitis in level T5-T6 with a multiloculated intraspinal epidural lesion stretched from level T4-T7 underneath the unaffected posterolateral longitudinal ligament, compacting the cord with a modest spinal canal with modest stenosis of the spinal canal, vertebral obliteration with prevalent kyphosis, and a huge bilateral paraspinal phlegmon with lesions.62
Septic arthritis: A rare cause of non-typhoidal
Salmonella
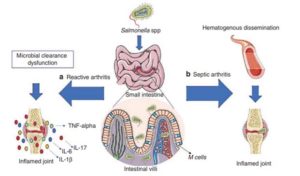
Salmonella arthritis is a relatively uncommon joint ailment that accounts for about 1% of total infected case scenarios.63 Organisms that induce the problem of septic arthritis can often be a non-typhoidal Salmonella species, and it typically manifests as one of the metastatic illnesses in children and young adults who have previously experienced bacteremia.64 Salmonella septic arthritis is commonly caused by hematogenous spread. Hardly 10 to 15% of rare situations result in a positive blood culture of specimens.65 Many non-typhoid Salmonella serotypes have already been known to trigger septic arthritis or osteomyelitis in humans, which would include S. Enteritidis, S.Typhimurium, S. Typhi, S. Newport, S. Choleraesuis, S. Virchow, and S. OhiO.66,57,67-71 Genomic identification of Salmonella Virchow monophasic variant in the study of Wang et al.72 showed that this variant was responsible for knee joint septic arthritis. Surgical treatment and antimicrobial therapy are common treatments. Acute septic arthritis is relatively uncommon in immune-competent children. Bacterial infections that begin in the skin (after cutaneous lesions) or throat are the leading triggers (like Staphylococcus or Streptococcus species). Salmonella species exhibit reactive and septic arthritis in the joints. (Figure 7) shows how Salmonella species invade the small intestine and how inflamed joints lead to septic arthritis and reactive arthritis.73
Figure 7. Pathophysiology of Salmonella arthritis. (a) After intestinal colonization and infection, a person with genetic and environmental sensitivity related to factors that affect microbial clearance will result in reactive arthritis. (b) After intestinal colonization and infection, hematogenous dissemination can occur, leading to the development of Salmonella bacilli and finally causing septic arthritis.73
Urinary tract infections
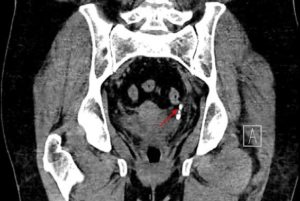
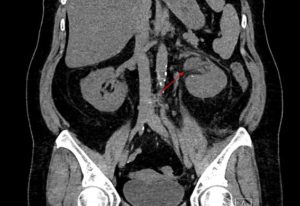
Urinary tract infection by NTS is uncommon and is also typically linked with degenerative changes in the genitourinary tract, immunodeficiency, and chronic conditions like diabetes. Immediate urethral inflammation, which is likely to occur in females, and hematogenous dispersal from gastroenteritis illness are the modes of inflammation.74 Serious diseases that increase the risk of Salmonella UTI involve cardiovascular or liver problems, cancer, and acute hemodialysis.74-76 The much more widely known Salmonella-caused UTI illnesses are nephrocalcinosis and nephrolithiasis. Primary hyperparathyroidism is associated with an increased risk of nephrolithiasis. Furthermore,77 indicated that 25-33% of non-typhoidal salmonellosis in those patients who have undergone kidney transplants had urinary tract infections, whereas no individuals with a heart transplant had non-typhoidal salmonellosis-acquired urinary tract infections. Non-typhoidal Salmonella UTI is primarily linked to tract structural abnormalities. The CT scan report of the abdomen/ pelvis disclosed chronic occluding calculi in distally present left ureter, left renal oedema, and hydroureteronephrosis, Figure 8 and Figure 9.78
Only a few investigations have been undertaken to ascertain the etiology of NTS in people. Recognition of serotype dispersion, and also drug sensitivity patterns is critical for developing effective therapeutic and control strategies. This article guides us about different infections which are triggered by NTS, how they affect our body, and the way our body encounters them. Recent advancements in sequencing and bioinformatics, as well as the emergence of new versions to review interrelations between host and cell, have granted critical perceptions in understanding the gene’s role and their regulation process, which might help in providing explanations of resistance against Salmonella infections. The immense population of sufferers is children under the age of five. Both experimental outcomes and medical outcomes indicate that this pathogen is highly adaptable to various antimicrobial selective pressures.79 Salmonella vigilance in both people and animals is mandatory to effectively control the associated infections.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
NC and PR conceived the study. PR, VC, and JC performed literature review. PR wrote the first draft. PR, VC and JC worked on subsequent drafts. NC reviewed and revised the manuscript. All authors read and approved the final manuscript for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
This article does not contain any studies with human participants or animals performed by any of the authors.
- Agbaje M, Begum RH, Oyekunle MA, Ojo OE, Adenubi OT. Evolution of Salmonella nomenclature: a critical note. Folia Microbiol. 2011;56(6):497-503.
Crossref - Centers for Disease Control and Prevention. Reptile- associated salmonellosis–selected states, 1996-1998. Morb Mortal Wkly Rep. 1999; 48(44): 1009-1013
- Centre for Disease Control and Prevention. National Salmonella Surveillance Overview. 2013. https://www. cdc.gov/nationalsurveillance/pdfs/salmonella-annual- report-2013-508c.pdf.
- Silva C, Calva E, Maloy S. One Health and food-borne disease: Salmonella transmission between humans, animals, and plants. Microbiol Spectr. 2014;2(1):OH- 0020-2013.
Crossref - Hoelzer K, Moreno AI, Wiedmann M. Animal contact as a source of human non-typhoidal salmonellosis. Vet Res. 2011;42(1):1-27.
Crossref - Centre for Disease Control and Prevention. Foodborne Germs and Illnesses. 2020. www.cdc.gov/foodsafety/ foodborne-germs.html. Accessed August 27, 2020.
- Criscuolo A, Issenhuth-Jeanjean S, Didelot X, et al. The speciation and hybridization history of the genus Salmonella. Microb Genom. 2019;5(8):e000284.
Crossref - Taliha MD, Balti E, Maillart E, et al. Invasive non- typhoidal Salmonella infection complicated by metastatic infections: Report of three cases. IDCases. 2022;28:e01498.
Crossref - Marchello CS, Birkhold M, Crump JA, et al. Complications and mortality of non-typhoidal Salmonella invasive disease: a global systematic review and meta-analysis. Lancet Infect Dis. 2022;22(5):692-705.
Crossref - Rule R, Mbelle N, Sekyere JO, Kock M, Hoosen A, Said M. A rare case of Colistin-resistant Salmonella Enteritidis meningitis in an HIV-seropositive patient. BMC Infect Dis. 2019;19(1):806.
Crossref - Albert MJ, Bulach D, Alfouzan W, et al. Non-typhoidal Salmonella blood stream infection in Kuwait: Clinical and microbiological characteristics. PLoS Negl Trop Dis. 2019;13(4):e0007293.
Crossref - Sanni AO, Onyango J, Usman A, Abdulkarim LO, Jonker A, Fasina FO. Risk Factors for Persistent Infection of Non-Typhoidal Salmonella in Poultry Farms, North Central Nigeria. Antibiotics. 2022;11(8):1121.
Crossref - Divek VTN, Venkitanarayanan K, Kollanoor A. Antibiotic-resistant Salmonella in the food supply and the potential role of antibiotic alternatives for control. Foods. 2018;7(10):167-291.
Crossref - Christidis T, Hurst M, Rudnick W, Pintar KDM, Pollari F. A comparative exposure assessment of foodborne, animal contact and waterborne transmission routes of Salmonella in Canada. Food Control. 2020;109:106899.
Crossref - Centers for Disease Control and Prevention (CDC). https://www.cdc.gov/salmonella/pettreats-07-19/ Acessed on 26 March 2020.
- McLeod N, Lastinger A, Bryan N, Kieffer T, Wolfe T. Salmonella neck abscess in a diabetic. ID Cases. 2019;17:e00541.
Crossref - Arroyo SQ, Villalba NL, Mezard-saint V, Zulfiqar AA, Ortiz MBA, Zamorano NF. Thyroid abscess due to Salmonella enteritidis in an immunocompetent patient. Revue Medicale de Liege. 2019;74(11):563- 565.
- Manzar N, Almuqamam M, Kaushik K, et al. Primary non-typhoidal Salmonella infection presenting as a splenic abscess in a healthy adolescent male. Le Infezioni Medicina. 2019;27:77-81.
- Wu C, Yan M, Liu L, Lai J, Chan EW, Chen S. Comparative characterization of nontyphoidal Salmonella isolated from humans and food animals in China, 2003-2011. Heliyon. 2018;4(4):1-18.
Crossref - Cheng RA, Eade CR, Wiedmann M. Embracing Diversity: Differences in Virulence Mechanisms, Disease Severity, and Host Adaptations Contribute to the Success of Nontyphoidal Salmonella as a Foodborne Pathogen. Front Microbiol. 2019;10:1-20.
Crossref - Snyder TR, Boktor SW, M’ikanatha, NM. Salmonellosis outbreaks by Food Vehicle, Serotype, Season, and Geographical Location, United States, 1998 to 2015. J Food Prot. 2019;82(7):1191-1199.
Crossref - Esan OB, Pearce M, van Hecke O, et al. Factors associated with Sequelae of Campylobacter and Non- typhoidal Salmonella Infections: A Systematic Review. EBioMedicine. 2017;15:100-111.
Crossref - Hoffmann M, Zhao S, Pettengill J, et al. Comparative Genomic Analysis and Virulence Differences in Closely Related Salmonella enterica Serotype Heidelberg Isolate from Humans, Retail Meats, and Animals. Genome Biol Evol. 2014;6(5):1046-1068.
Crossref - Feasey NA, Dougan G, Kingsley RA, Heyderman RS, Gordon MA. Invasive non-typhoidal Salmonella disease: an emerging and neglected tropical disease in Africa. Lancet. 2012;379(9835):2489-99.
Crossref - Haselbeck AH, Panzner U, Im J, Baker S, Meyer F, Marks F. Current perspectives on invasive nontyphoidal Salmonella disease. Curr Opin Infect Dis. 2017;30(5):498-503.
Crossref - Roth GA, Abate D, Abate KH, et al. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392:1736-88.
Crossref - Stanaway JD, Parisi A, Sarkar K, et al. The global burden of non-typhoidal Salmonella invasive disease: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Infect Dis. 2019;19(12):1312-1324.
Crossref - Bjelland AM, Sandvik LM, Skarstein MM, Svendal L, Debenham JJ. Prevalence of Salmonella serovars isolated from reptiles in Norwegian zoos. Acta Vet Scand. 2020;62(1):3.
Crossref - Hydeskov HB, Guardabassi L, Aalbaek B, Olsen KE, Nielsen SS, Bertelsen MF. Salmonella prevalence among reptiles in a zoo education setting. Zoonoses Public Health. 2013;60(4):291-295.
Crossref - Trotta A, Del Sambro L, Galgano M, et al. Salmonella enterica Subsp. houtenae Associated with an Abscess in Young Roe Deer (Capreolus capreolus). Pathogens. 2021;10(6):654.
Crossref - Control CFD. An Atlas of Salmonella in the United States, 1968-2011: Laboratory-based Enteric Disease Surveillance; CDC, Ed.; US Department of Health and Human Services: Atlanta, GA, USA. 2013;248.
- Tack DM, Ray L, Griffin PM, et al. Preliminary Incidence and Trends of Infections with Pathogens Transmitted Commonly Through Food-Foodborne Diseases Active Surveillance Network, 10 U.S. Sites, 2016-2019. MMWR Morbidity Mortality Weekly Report. 2020:509- 514.
Crossref - TDH. Interactive dashboard for selected reportable diseases and events. In Communicable and Environmental Diseases and Emergency Preparedness (CEDEP) 2010-2012 Annual Report. https://www. tn.gov/content/dam/tn/health/documents/cedep- weeklyreports/AnnualReport2010-12.pdf. Accessed on 21 January 2020.
- LPH. Salmonella annual report 2018. In Louisiana Office of Public Health-Infectious Disease Epidemiology Section; 2018. http://ldh.la.gov/assets/oph/Center- PHCH/Center-CH/infectious-epi/Annuals/Salmonella_ LaIDAnnual_2018.docx.pdf. Accessed on 21 January 2020.
- Byndloss M, Rivera-Chavez F, Tsolis R, Baumler A. How bacterial pathogens use type III and type IV secretion systems to facilitate their transmission. Curr Opin Microbiol. 2017;35:1-7.
Crossref - Eun Y, Jeong H, Kim S, et al. A large outbreak of Salmonella enterica serovar Thompson infections associated with chocolate cake in Busan, Korea. Epidemiol Health. 2019;41:e2019002.
Crossref - Friesema I, de Jong A, Hofhuis A, et al. Large outbreak of Salmonella Thompson related to smoked salmon in the Netherlands, August to December 2012. Euro Surveill. 2014;19(39):20918.
Crossref - Hennessy TW, Hedberg CW, Slutsker L, et al. A national outbreak of Salmonella enteritidis infections from ice cream. The Investigation Team. N Engl J Med. 1996;334(2):1281-1286.
Crossref - Laughlin M, Bottichio L, Weiss J, et al. Multistate outbreak of Salmonella Poona infections associated with imported cucumbers, 2015-2016. Epidemiol. Infect. 2019;147:e270.
Crossref - World Health Organization. 2020. https://www.who. int/en/news-room/fact-sheets/detail/ Salmonella– (non-typhoidal).
- Marzel A, Desai PT, Goren A, et al. Persistent infections by nontyphoidal Salmonella in humans: Epidemiology and Genetics. Clin Infect Dis. 2016;62(7):879-886.
Crossref - World Health Organization. Estimates of the Global Burden of Foodborne Disease. 2020. https://extranet. who.int/sree/FERG/FERG_DataFiles.zip.
- Boschi-Pinto C, Velebit L, Shibuya K. Estimating child mortality due to diarrhea in developing countries. Bull World Health Organ. 2008;86(9):710-717.
Crossref - Castlemain BM, Castlemain BD. Case report: post-salmonellosis abscess positive for Salmonella Oranienburg. BMC Infect Dis. 2022;22(1):337.
Crossref - Chen PL, Chang CJ, Wu CJ. Extraintestinal focal Infectious in adults with non typhoid Salmonella bacteremia: predisposing factors and clinical outcome. J Intern Med. 2007;261(1):91-100.
Crossref - Crump JA, Sjolund-Karlsson M, Gordon MA, Parry CM. Epidemiology, clinical presentation, laboratory diagnosis, antimicrobial resistance, and antimicrobial management of invasive Salmonella infections. Clin Microbiol Rev. 2015;28(4):901-937.
Crossref - Katiyo S, Pebody BM, Minaji M, et al. Epidemiology and Outcomes of Nontyphoidal Salmonella Bacteremias from England, 2004 to 2015. J Clin Microbiol. 2019;57(1):1-35.
Crossref - Wen SC, Best E, Nourse C. Non-typhoidal Salmonella infections in children: Review of literature and recommendations for management. J Paediatr Child Health. 2017;53(10):936-941.
Crossref - Lee JH, Cho HK, Kim KH. Etiology of invasive bacterial infections in immunocompetent children in Korea (2006-2010): a retrospective multicenter study. J Korean Med Sci. 2011;26(2):174-183.
Crossref - Wu HM, Huang WY, Lee ML, Yang AD, Chaou KP, Hsieh LY. Clinical features, acute complications, and outcome of Salmonella meningitis in children under one year of age in Taiwan. BMC Infect Dis. 2011;11:30.
Crossref - Moon HJ, Lee Y, Han MS. Nontyphoidal Salmonella meningitis in an immunocompetent child. Pediatric Infection and Vaccine. 2022;29(1):54-60.
Crossref - Keddy KH, Sooka A, Musekiwa A, et al. Clinical and Microbiological Features of Salmonella Meningitis in a South African Population, 2003-2013. Clin Infect Dis. 2015;61(4):272-282.
Crossref - Subramoney EL. Non-typhoidal Salmonella infections in HIV-positive adults. South African Medical Journal. 2015; 105(10): 805-807
- Monica F, Valentina C, Lisa M, et al. Unusual meningitis caused by nontyphoidal Salmonella in an Italian infant: a case report. Acta Biomedica. 2019;90(2):333-38.
Crossref - Lombardi D, Malaspina S, Strippoli A, Lucarelli C, Luzzi I, Ripabelli G. Salmonella enterica serovar Virchow meningitis in a young man in Italy: a case report. J Med Case Rep. 2014;8:139.
Crossref - Librianto D, Suwarto S, Imran D, et al. An extremely rare case of upper thoracic Salmonella infection. Orthop Res Rev. 2021;13:107-112.
Crossref - McAnearney S, McCall D. Salmonella osteomyelitis. Ulster Medical Journal. 2015;84:171-172.
- Sanchez AA, Mazurek MT, Clapper MF. Salmonella osteomyelitis presenting as fibrous dysplasia. A case report. Clin Orthop Relat Res. 1996;330:185-189.
Crossref - Anand AJ, Glatt AE. Salmonella osteomyelitis and arthritis in sickle cell disease. Semin Arthritis Rheum. 1994;24(3):211-121.
Crossref - Jones TF, Ingram LA, Cieslak PR, et al. Salmonellosis outcomes differ substantially by serotype. J Infect Dis. 2008;198(1):109-114.
Crossref - Weisman JK, Nickel RS, Darbari DS, Hanisch BR, Diab HY. Characteristics and outcomes of osteomyelitis in children with sickle cell disease: A 10-year single-center experience. Pediatr Blood Cancer. 2020;67(5):e28225.
Crossref - Elnour S, Hashim M, Ibrahim H. Disseminated non- typhoidal Salmonella infection with salmonella pneumonia and vertebral osteomyelitis in sickle cell disease: A case report. IDCases. 2022;27:e01390.
Crossref - Shanthi M, Sekar U, Sridharan KS. Septic arthritis of hip caused by Salmonella typhi: A case report. Case Rep Infect Dis. 2012;464527.
Crossref - Kurniawan A, Sitorus IP, Loho T, Hutami WD. A rare case of septic arthritis of the knee caused by Salmonella typhi with preexisting typhoid fever in a healthy, immunocompetent child-A case report. Int J Surg Case Rep. 2021;78:76-80.
Crossref - Al Nafeesah AS: Nontyphoidal Salmonella septic arthritis of the elbow in a healthy infant. Pan Afr Med J. 2015;22:357.
Crossref - Salem KH. Salmonella osteomyelitis: A rare differential diagnosis in osteolytic lesions around the knee. J Infect Public Health. 2014;7(1):66-69.
Crossref - Kato H, Ueda A, Tsukiji J, Sano K, Yamada M, Ishigatsubo Y. Salmonella enterica serovar Ohio septic arthritis and bone abscess in an immunocompetent patient: a case report. J Med Case Rep. 2012;6:204.
Crossref - Weston N, Moran E. Salmonella Newport causing osteomyelitis in a patient with diabetes. BMJ Case Rep. 2015.
Crossref - Katsoulis E, Pallett A, Bowyer GW. Septic arthritis of the knee by Salmonella monte video. Ann R Coll Surg Engl. 2004;86(4):272-274.
Crossref - Sy AM, Sandhu J, Lenox T. Salmonella enterica serotype Choleraesuis infection of the knee and femur in a nonbacteremic diabetic patient. Case Rep Infect Dis. 2013;2013:506157.
Crossref - Morgan MG, Forbes KJ, Gillespie SG. Salmonella septic arthritis: a case report and review. Journal of Infection. 1990;21(2):195-203.
Crossref - Wang Z, Xu H, Chu C, Tang Y, Li Q, Jiao X. Genomic identification of the emerging multidrug-resistant Salmonella Virchow monophasic variant causing septic arthritis of the knee joint. Authorea Preprints. 2022.
Crossref - Tobon GJ, Garcia-Robledo JE, Nieto-Aristizabal I. Salmonella arthritis. Infections and the Rheumatic Diseases. 2019;41-47.
Crossref - Jehangir A, Poudel D, Fareedy SB, Salman A, Qureshi A, Jehangir Q, Richard A. Group D Salmonella urinary tract infection in an immunocompetent male. Case Rep Infect Dis. 2015;1-2.
Crossref - Longo D, Fauci A, Kasper D, Hauser S. Harrison’s Principles of Internal Medicine. Edn 18, McGraw-Hill Education, Columbus. 2011;1278-1280.
- Tena D, Gonzalez-Praetorius ssA, Bisquert J. Urinary tract infection due to nontyphoidal Salmonella: Report of 19 cases. Journal of Infection. 2007;54(3):245-249.
Crossref - Hsu RB, Lin FY. Nontyphoid Salmonella infection in heart transplant recipients. Am J Med Sci. 2008;336(5):393- 396.
Crossref - Altaf A, Tunio N, Tunio S, Zafar MR, Bajwa N. Salmonella Urinary Tract Infection and Bacteremia Following Non-Typhoidal Salmonella Gastroenteritis: An Unusual Presentation. Cureus. 2020;12(12):e12194.
Crossref - Chen CL, Su LH, Chiu CH. Transcription modulation of gene expression in Salmonella enterica serotype Choleraesuis by sub-inhibitory concentrations of ciprofloxacin. Food Res Int. 2012;45(2):973-77.
Crossref
© The Author(s) 2023. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.