ISSN: 0973-7510
E-ISSN: 2581-690X
Fluoroquinolones (FQ) are integral part of multidrug resistant/rifampicin resistant tuberculosis (MDR-TB/RR TB) treatment regimens. FQ resistance in MDR-TB lead to narrower treatment options and poor treatment outcomes. Hence proportion of FQ resistance among MDR-TB and cross-resistance among FQ in MDR-TB isolates was studied. This is the first study from North Coastal Andhra Pradesh, India. A cross-sectional study was conducted on 21,474 sputum samples from January-December 2018 & 833 culture isolates (rifampicin resistant detected by GeneXpert MTB / RIF assay & MDR-TB detected by direct and indirect GenoType MTBDRplus Assay) were analysed for FQ resistance by Mycobacterium Growth Indicator Tube (MGIT) for ofloxacin (Ofx) 2.0 µg/mL, levofloxacin (Lfx) 1.5 µg/mL & moxifloxacin (Mfx) 2.0 μg/mL. Among 833 culture isolates, 119 (14.2%) showed resistance to FQ which included 90 (10.8%) Ofx and Lfx resistance isolates but still sensitive to Mfx, 16 (1.92%) with resistance to all FQ tested, 12 Ofx monoresistance & 1 Lfx monoresistance. Mfx monoresistance was absent. Mfx sensitive isolates are 714 (85.7%) and were always sensitive to Ofx and Lfx. Cross-resistance was not complete. MDR-TB/RR TB is 4.1% among all TB patients. Additional resistance to FQ, among MDR-TB patients was seen in 14.2% patients.
Multidrug-resistant tuberculosis, fluoroquinolones, MGIT 960, cross-resistance
Infection with Mycobacterium tuberculosis resistant to isoniazid (H) and rifampicin (R) also called as MDR-TB or RR TB,1-3 leads to treatment with less potent, more toxic and expensive second-line anti-TB drugs (SLD). Fluoroquinolones (FQ) are crucial and integral part of drug resistant tuberculosis (DR-TB) treatment regimens1,3,4. Wide and illogic use of FQ, either for tuberculosis (TB) or other infections of respiratory tract, is responsible for increasing FQ resistance in MDR-TB, and lead to a situation where treatment options are narrowed1,3. FQ resistance and poor treatment outcomes in MDR-TB have been reported in studies4,5. Unfortunately, India has high TB burden and also is a high MDR-TB burden country,6 and by inference harbours large extensively-drug resistant tuberculosis (XDR-TB) population. FQ resistance may thus be a more prevalent than actually reported and can threaten TB control programmes1. Studies from India have demonstrated the prevalence of FQ resistance in MDR-TB patients1,3,7,8,9 but none from North Coastal Andhra Pradesh. Hence we studied the proportion of FQ resistance among MDR-TB/RR-TB and also the proportion of their cross‑resistance among MDR-TB/RR-TB isolates.
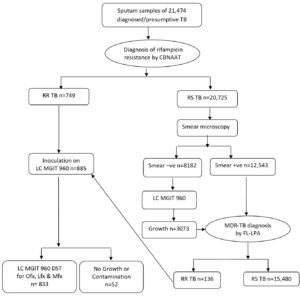
This study was performed at the Intermediate reference laboratory (IRL), Visakhapatnam, Andhra Pradesh, India which is a biosafety level 3 (BSL3) laboratory. It is certified and Quality assured by National Institute for Research in TB, Chennai; the WHO Supranational Reference Laboratory for testing of Mycobacterium tuberculosis. The Institutional Ethics Committee reviewed and approved the present research procedure and removed the right to seek informed consent. Under the Revised National Tuberculosis Control Program (RNTCP) for India now called as National Tuberculosis Elimination Programme (NTEP), universal drug susceptibility testing (DST) to at least R for all individuals diagnosed with TB through Cartridge Based Nucleic Acid Amplification Test (CBNAAT)/ GeneXpert MTB / RIF assay (http:/www.cepheid.com), available in district/CBNAAT laboratory which are WHO approved2. Two samples are collected per patient. One specimen to perform CBNAAT and the other is transported to the IRL for susceptibility testing by first line-probe assay (FL-LPA)/ GenoType MTBDRplus Assay (Hain Lifescience, Nehren, Germany) as applicable and further processing on liquid culture and drug susceptibility testing2. All sputum samples received in the laboratory have been considered for this study. A cross-sectional study was conducted on 21,474 sputum samples from January 2018 to December 2018 collected over a 12-month period. All samples were treated using the NALC-NaOH (N-acetyl-L-cysteine sodium hydroxide) method10 in the BSL3 laboratory in the Class II biosafety cabinet. The concentrated sample was used for further testing. A detailed workflow of the experiments is given in Fig. 1. Sputum samples with CBNAAT result as Mycobacterium tuberculosis detected and rifampicin resistance (RR) were inoculated in liquid culture (LC) system Mycobacteria Growth Indicator Tube (MGIT) 960. Smears from sputum with CBNAAT result as Mycobacterium tuberculosis detected and rifampicin sensitive (RS), were stained with fluorescent microscopy technique and were observed. Smear-positive samples were then subjected to FL-LPA/GenoType MTBDRplus Assay to detect resistance to R & H. Smear-negative samples were cultured using MGIT 960 (Becton Dickinson, Maryland, USA). Positive cultures were then subjected to FL-LPA. All RR-TB cultures then underwent DST by MGIT 960 for detection of resistance to Ofx, Lfx, & Mfx.
Fig. 1. Outline of experiments incorporated in the study. TB, Tuberculosis: CBNAAT, Cartridge Based Nucleic Acid Amplification Test; RR TB, Rifampicin resistance TB; RS TB, Rifampicin sensitive TB; LC MGIT, Liquid cultureMycobacteria Growth Indicator Tube; FL-LPA, first line line probe assay; DST, drug susceptibility testing; Ofx, ofloxacin; Lfx, levofloxacin; Mfx, moxifloxacin.
Sputum digestion and decontamination
Were performed in a class II biosafety cabinet by the NALC-NaOH method10 ,11. The pellet obtained after centrifugation was suspended in 1.0 mL of phosphate buffer (pH 6.8) and 500 µL was used for FL-LPA & the remaining suspension was stored.
Drug susceptibility testing by MGIT 960
Samples resistant to R, 500 µL were inoculated into a MGIT tube (Becton Dickinson & Co., Franklin Lakes, NJ). Positive MGIT tube (1–3 days) was then used for DST. The positive tubes by MGIT-960 underwent sterility check on brain heart infusion agar to rule out contamination. All isolates were identified as Mycobacterium tuberculosis complex by immunochromatography assay (SD MPT64TB Ag kit developed by Standard Diagnostics, South Korea). Ofx, Lfx and Mfx obtained from Sigma-Aldrich (St Louis, MO) were used for preparation of Drug stock solutions. The drug susceptibility testing was done according to the standard 1% proportionate method as per the manufacturer’s instructions11.
MGIT tubes were added with 800 µL of oleic acid-albumin-dextrose-catalase (OADC) supplement. For inoculation of MGIT tubes with culture, it was diluted in 1:5 ratio with sterile saline from 3-5 days old positive MGIT tube and direct inoculation was done for 0-2 days old positive tube. 100 μl of drug stock solutions were added to the MGIT tubes to have final concentrations of 2.0 μg/mL Ofx, 1.5 μg/mL of Lfx, and 2.0 μg/mL of Mfx. Growth control (GC) tube was also included. Culture for GC tube was diluted to 1:100 with sterile saline and then added to MGIT tubes without drug. MGIT tubes with drug were inoculated with 500 µL of the inoculum. Mycobacterium tuberculosis H37Rv strain was used as positive control.
GenoType MTBDRplus assay
FL-LPA was performed according to the manufacturer’s instructions12. 500 µL of the decontaminated sediment was used for DNA extraction using GenoLyse procedure. The supernatant containing DNA was used for amplification and hybridisation. The nitrocellulose strips were then fixed on paper for interpretation. Resistance interpretation was based on presence or absence of wild-type and mutation bands.
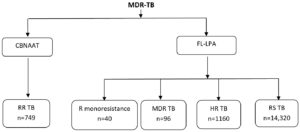
Of the 21,474 sputum samples from diagnosed TB and presumptive TB patients, 749 were Mycobacterium tuberculosis positive and RR, 20,589 were Mycobacterium tuberculosis positive and RS by GeneXpert/CBNAAT. Among RS TB, 12,543 were smear-positive and 8,182 were smear-negative. FL-LPA was performed on smear-positive RS TB samples. All smear-negatives RS TB samples were inoculated in LCMGIT 960. Among 8,182 samples inoculated in liquid culture, 3,073 cultures showed growth for Mycobacterium tuberculosis. Culture positive among smear negative is 37% . The cultures were then subjected to Genotype MTBDRplus/FL-LPA. A total of 15,616 samples were subjected to FL-LPA, of which 136 were RR TB. The FL-LPA and CBNAAT results are given in Fig. 2.
Fig. 2. Cartridge Based Nucleic Acid Amplification Test & first line line probe assay results. MDR-TB, Multidrug resistance tuberculosis; CBNAAT, Cartridge Based Nucleic Acid Amplification Test; FL-LPA, first line line probe assay; RR TB, Rifampicin resistance Tuberculosis; HR TB, Isoniazid resistance tuberculosis; RS TB, Rifampicin sensitive Tuberculosis.
Table (1):
Fluoroquinolones resistance among rifampicinresistance tuberculosis.
Total Rifampcin resistance |
Culture positive (%) |
Culture negative |
Culture lost |
Fluoroquinolone resistance (%) |
|---|---|---|---|---|
885 |
833 (94.1) |
42 |
10 |
119 (14.2) |
A total of 885 RR TB samples were inoculated into liquid culture drug susceptibility testing (LC-DST) ie. MGIT tube. Of the 885 samples, 52 showed contamination or very slow growth and finally lost after many attempts for culture and thus were not included in the study (Table 1). Samples which showed growth for Mycobacterium tuberculosis on MGIT medium were 833 and were subjected to susceptibility testing with Ofx (2 µg/mL), Lfx (1.5 ug/mL) and Mfx ( 2 ug/mL) at critical concentrations recommended by WHO. Among 833 isolates where FQ drug susceptibility pattern was available, Mfx sensitive isolates are 714 (85.7%) and were always sensitive to Ofx and Lfx. Mfx monoresistance was absent in the present study. A total of 119 (14.2%) isolates showed resistance to FQ (Table 2). Among 119 FQ resistance isolates, 12 were Ofx monoresistance, 1 Lfx monoresistance. Ofx and Lfx resistant isolates were 90 (10.8%) but they were sensitive to Mfx. Resistance to all fluoroquinolones tested ie. Ofx, Lfx and Mfx in RR TB were 16 (1.92%) only. Cross-resistance was not complete.
Table (2):
Drug susceptibility patterns in 833 multidrug resistance / rifampicin resistance tuberculosis isolates to fluoroquinolones.
| Drug resistance pattern | Number of isolates (%) |
|---|---|
| One drug resistance | |
| Ofx | 12(1.3) |
| Lfx | 01(0.1) |
| Mfx | 0 |
| Tow drug resistance | |
| Ofx+Lfx | 90(10.1) |
| Lfx+Mfx | 0 |
| Ofx+Mfx | 0 |
| Three drug resistance | |
| Ofx+Lfx+Mfx | 16(1.8) |
| All sensitive isolates | |
| Ofx+Lfx+Mfx | 714(80.8) |
Ofx, Ofloxacin; Lfx, Levofloxacin; Mfx, Moxifloxacin
WHO estimated incidence of MDR-TB/RR-TB in 2018 in India was 1,30,000.6 The first National Anti-Tuberculosis Drug Resistance Survey (NDRS) from India released on World TB Day, i.e., 24th March 2018 states incidence of MDR-TB is 6.19% (CI 5.54–6.90%) among all TB patients2,5.These figures are higher than that observed in the present study ie. MDR-TB/RR TB is 4.1% among all TB patients. Among MDR TB patients 89.1% of rifampicin monoresistant patients were detected by GeneXpert/CBNAAT and LPA methods. All rifampicin monoresistance were considered as MDR-TB and initiated on MDR-TB treatment under RNTCP as rifampicin monoresistance is considered as surrogate marker for MDR-TB2,7. One of the key findings of the NDRS report is that almost all RR-TB patients are resistant to isoniazid with or without other first or second line drugs2.
FQ especially Mfx, are integral part of standard regimen (shorter MDR-TB regimen and conventional MDR-TB regimen) for initiating treatment of MDR-TB/RR-TB at district DR-TB centre based on CBNAAT or FL-LPA2. FQ have the potential to become first line therapy for TB7. Latest Global tuberculosis report (2019), any FQ resistance among MDR-TB patients was 21 % globally6. In National Anti-Tuberculosis Drug Resistance Survey, additional resistance to FQ, among MDR-TB patients was shown to be 21.82 %2,5. Association of FQ resistance among individuals with resistance to first‑line TB drugs is evidence of acquired FQ resistance while on treatment for TB3. In the current study, additional resistance to FQ, among MDR-TB patients was seen in 14.2% patients and was lower. It may be due to early diagnosis of MDR/RR TB and satisfactory treatment due to availability of quality assured drug susceptibility testing services in the state and the implementation of programmatic management of DR-TB under RNTCP 7–8 years back itself. Studies from other reference laboratories in India reported additional resistance to FQ among MDR-TB as 16.1% 3, 17.1% 3, 31% 8 & 33% 7from isolates from Tamil Nadu, Kerala, Delhi and Karnataka respectively. The variations may be due to differences in study population and evolving DR-TB algorithms with time. The present study was performed after implementation of Universal DST. It may limit benefit of short course regime recommended by WHO for MDR-TB patients as resistance to FQ excludes the use of regime9. In a study from Uttar Pradesh, FQ resistance (with or without injectable SLD resistance) detected by second line LPA was high (58.4%) and stated those patients were not entitled for a shorter regimen9. Knowledge regarding individual’s drug resistance pattern before would help in instituting appropriate treatment regimen avoiding transmission of extensively DR-TB (XDR-TB) in the population7. Under Universal DST the algorithms for diagnosing TB, demonstration of susceptibilities of MDR-TB strains to FQ before initiation of treatment have been incorporated, so that according to DST results the treatment regimen can be changed2,8. With use of nuclei acid amplification test (Xpert MTB/RIF) and probe hybridization techniques (line probe assay), drug resistance are detected early4. Culture sensitivity is gold standard and liquid culture (MGIT-960) techniques which have excellent sensitivity and specificity is beneficial when used in high TB burden countries4.
Resistance to FQ is due to mutations in gyr A and gyr B genes which code for enzyme DNA gyrase involved in bacterial DNA replication3,7. FQ resistance among TB patients is due to the use of the drugs for TB as well as for other infections including respiratory as they are broad spectrum, affordable, have high bioavailability, good safety profile and convenient dosing4,7,8. FQ are available over the counter and are regularly prescribed. A retail store audit in India was conducted in 2004 by ORG IMS, reported two highly prescribed antibiotics as ciprofloxacin and Ofx, with gatifloxacin and Lfx being sixth and eighth most frequently prescribed13. FQs are used as first line drugs along with other TB drugs to shorten ATT or as a sequential addition to a failing first-line regimen3. Use of FQ before TB diagnosis was associated with FQ resistance, especially when FQ were used for over 10 days, >60 days ahead of TB diagnosis1. Development of resistance occurs even with very short duration of treatment and within 13 days of exposure to FQ4,8. Studies have reported FQ resistance among drug sensitive TB in India and abroad4,8. FQ resistance is a risk factor for development of XDR-TB.
FQs having significant antimycobacterial activity are Ofx, Lfx, gatifloxacin and Mfx3. Cross resistances among three FQ tested is common7 and was also seen in present study. Cross-resistance within the FQ class is reduced susceptibility to one FQ likely confers reduced susceptibility to all FQ1,7. In this study, 89.8% of MDR-TB isolates resistant to Ofx at base line were also resistant to Lfx and Mfx. Similar results were obtained in studies by Mamatha et al. and Ahmad et al.1,7 Lfx was almost always associated with resistance to other FQs (99%) in the present study. MDR-TB isolates resistant to Ofx and Lfx (13.4%) showed cross-resistance to Mfx 2 μg/mL and hence have an minimum inhibitory concentration (MIC) above that level and hence is not a suitable treatment option. High resistance above 3 mg/L makes FQ likely to be useless as peak Mfx level in humans is around 3 mg/L3. Resistance to Mfx alone was absent. Absence of resistance to Mfx alone and low cross-resistance is supportive evidence for testing of Mfx as candidate for detecting resistance to FQs and also their use in regimens for treatment of MDR-TB. In this study the concentrations of Mfx tested was 2.0 μg/mL as per PMDT guidelines2.Cross-resistance between earlier generation FQ and Mfx is present at lower concentration of 0.25μg/mL3. Mfx is active against strains with low levels of resistance (MIC, 0.5 μg/mL) and reduces mortality on treatment with high dose of Mfx3. With intermediate resistance (MIC, 2.0 μg/ml) it still responds when given along with other second line drugs but has higher relapse rates3,7. The current WHO recommendation is to use Mfx when there is resistance to early-generation FQ, such as Ofx. Superior pharmacokinetic profile of eight methoxy FQ like Mfx makes it a better antimycobacterial and sterilizing agent and thus are used as treatment for MDR-TB with Ofx/Lfx resistance as bacilli may still be susceptible even if cross-resistance is present3. In India FQ resistance is a problem and needs to be addressed by the policy makers urgently so as to control DRTB4. To know the geographical distribution of drug resistant strains, locate their hotspots and related ecological factors in high TB burden countries Shibabaw A et al. proposed the use of geographical information system.
The strengths of this study are large sample size, performance of testing at NTEP/RNTCP certified and quality assured BSL3 laboratory and on all MDR/RR isolates from sputum samples of program‑defined, presumed RR patients continuously for 1 year from a large geographical area. This reflects situation after implementation of Universal DST which is more realistic and usage of various diagnostic techniques including both conventional and molecular techniques for estimation and confirmation of resistance. Some of the shortcomings of this study are no treatment history and FQ exposure data and no follow up for treatment responses.
In conclusion, in present study MDR-TB/RR TB is 4.1% among all TB patients and additional resistance to FQ among MDR-TB patients was seen in 14.2%. Cross-resistance among FQ was incomplete. Resistance to Mfx alone was absent. Newer generation FQs are promising drugs in the treatment of drug-resistant TB but care should be taken regarding the rationale use of these drugs for the treatment of other diseases especially when other drugs are available. We also support the adoption of a FQ restriction policy in India and also efforts to create awareness among practising doctors to use FQ cautiously. Universal DST followed by an individualized regimen based on DST results to control TB in the country is necessary.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
ETHICS STATEMENT
This article does not contain any studies with human participants or animals performed by any of the authors.
AVAILABILITY OF DATA
All datasets generated or analyzed during this study are included in the manuscript.
- Ahmad V, Sidiq Z, Vashishtha H, et al. Additional Resistance to Moxifloxacin and Levofloxacin among MDR-TB Patients with Base Line Resistance to Ofloxacin at a Reference Laboratory. J Biotechnol Biomater. 2016;6(3):239.
Crossref - Guideline for PMDT in India 2017. https://tbcindia.gov.in, Accessed 4 March, 2020.
- Sanker P, Satheesan A, Ambika AP, Santhosh VT, Balakrishnan R, Mrithunjayan SK. High moxifloxacin cross-resistance levels among “newly identified” ofloxacin-resistant multidrug-resistant tuberculosis patients from South India: A ticking bomb or a tricky challenge? Biomed Biotechnol Res J. 2017;1:59-64.
Crossref - Sharma R, Singh BK, Kumar P, Ramachandran R, Jorwal P. Presence of Fluoroquinolone mono-resistance among drug-sensitive Mycobacterium tuberculosis isolates: An alarming trend and implications. Clin Epidemiol Glob Health. 2019:7(3);363-366.
Crossref - Mishra GP, Mulani JD. First National Anti-Tuberculosis Drug Resistance Survey (NDRS) from India – An Eye Opener. J Infectiology. 2018;1(2):26-29.
- Global tuberculosis report 2019. https://www.who.int, Accessed 29 February, 2020.
- Mamatha HG, Shanthi V. Baseline resistance and cross-resistance among fluoroquinolones in multidrug-resistant Mycobacterium tuberculosis isolates at a national reference laboratory in India. J Glob Antimicrob Resist. 2018;12:5-10.
Crossref - Sharma R, Sharma SK, Singh BK, Mittal A, Kumar P. High degree of fluoroquinolone resistance among pulmonary tuberculosis patients in New Delhi, India. Indian J Med Res. 2019;149(1):62-66.
Crossref - Singh PK, Jain A. Limited Scope of Shorter Drug Regimen for MDR TB Caused by High Resistance to Fluoroquinolone. Emerg Infect Dis. 2019;25(9):1760-1762.
Crossref - Kent PT, Kubica GP. Public health mycobacteriology: a guide for the level III laboratory. Atlanta, GA: US Department of Health and Human Services, US Centers for Disease Control and Prevention. 1985.
- Siddiqi SH, Ruesch-Gerdes S. MGIT procedure Manual for BACTEC MGIT 960 TB System.Franklin Lakes, NJ: Becton Dickinson. 2006.
- GenoType MTBDRplus VER 2.0 Instructions for Use.https://www.ghdonline.org, Accessed 2 January, 2020.
- ORG IMS Research Private Ltd. Retail Store Audit. New Delhi, India. 2004.
- Ahmad N, Javaid A, Sulaiman SAS, Ming LC, Ahmad I, Khan AH. Resistance patterns, prevalence, and predictors of fluoroquinolones resistance in multidrug resistant tuberculosis patients. Braz J Infect Dis. 2016;20(1):41-47.
Crossref - Shibabaw A, Gelaw B, Gebreyes W, Robinson R, Wang S-H, Tessema B. The burden of pre-extensively and extensively drug-resistant tuberculosis among MDR-TB patients in the Amhara region, Ethiopia. PLoS ONE. 2020;15(2):e0229040. Accessed 26 December 2020.
Crossref
© The Author(s) 2021. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.