ISSN: 0973-7510
E-ISSN: 2581-690X
Amid the global rise of antimicrobial resistance (AMR), essential oils (EOs) offer a promising natural alternative to conventional therapies due to their broad-spectrum antimicrobial, antioxidant, and anti-inflammatory properties. This study evaluated the therapeutic potential of EOs-eugenol (clove), rosemary, rosehip, lavender, clary sage, and lemongrass- against clinical pathogens, including multidrug-resistant strains. Agar well diffusion and broth microdilution assays revealed that eugenol exhibited the greatest antimicrobial efficacy (zone of inhibition: 25.0 ± 0.8 mm; MIC: 62.5-125 µg/mL), followed by rosemary and lavender oils. A ternary EO combination significantly enhanced antibacterial activity against E. coli, K. pneumoniae, A. baumannii, and MRSA. Antioxidant activity, quantified using the DPPH radical scavenging assay, yielded the lowest IC50 for the EO combination (11.78 ± 0.70 µg/mL). HRBC membrane stabilization assays demonstrated comparable anti-inflammatory efficacy for the combination and eugenol (IC50: 28.19-36.18 µg/mL) relative to diclofenac. GC-MS profiling identified 40 bioactive constituents, notably eucalyptol, a-pinene, and eugenol, underpinning the observed pharmacological effects. These findings substantiate the therapeutic potential of EO formulations in addressing infection, oxidative stress, and inflammation. Statistical analysis confirmed significant differences across treatments, validating the efficacy of EO interventions. These findings highlight the therapeutic relevance of EOs, particularly in combination, as multi-target agents capable of addressing infection, oxidative stress, and inflammation. The data support their development as adjunctive or alternative options to combat AMR-driven clinical challenges.
Essential Oils, Antimicrobial Resistance, Bioactive Compounds, Anti-inflammation, Gas Chromatography-Mass Spectroscopy
Despite the current emphasis on synthetic medications dominating the healthcare system, medicinal plants continue to be a historical and traditional source of therapeutic compounds. Currently, over 80% of the global population, primarily residing in developing countries, continues to rely on plant-based medicines as their primary form of healthcare treatment.1 Essential Oils (EOs), also referred to as plant essences, volatile oils, ethereal oils, or aetheroleum, are intricate natural blends of evaporative, fat-dissolving, and fragrant compounds typically extracted from aromatic plants.2 EOs are constituted by terpenes and terpenoids, typically containing 20-60 chemical substances, though some may have hundreds of components. The primary constituents, comprising up to 85% of the total composition, broadly define their biological properties. Despite their lower concentrations, minor components play a vital role by synergistically enhancing biological activities.3
Presently, around 3000 EOs have been characterized, with roughly 300 holding commercial values across various industries, serving diverse applications in perfumery, cosmetics, healthcare products, dental care, and agricultural practices. They also function as natural food preservatives, flavour enhancers, and traditional remedies, demonstrating their versatility across pharmaceutical, food safety, and wellness sectors.4 These are a wide class of secondary metabolites, possessing diverse biological activities, encompassing antibacterial, antioxidant, anti-inflammatory, enzyme-inhibitory, sedative, anxiolytic, antidepressant, and anti-allergic activities.5
Despite the widespread availability of synthetic antimicrobials, the emergence of Antimicrobial Resistance (AMR) has created an urgent need for alternative therapeutic approaches. AMR poses a global health crisis and is escalating the Multidrug-resistant (MDR) strains of pathogenic bacteria such as Escherichia coli, Staphylococcus aureus, Salmonella typhi, and Citrobacter koseri.6 Global modelling prediction suggests that AMR contributes to a substantial fatality burden by 2050, with an estimated 1.91 million deaths (range: 1.56-2.26 million) directly caused by drug-resistant infections and 8.22 million deaths (range: 6.85-9.65 million) linked to AMR-related complications.7
In response to the crisis, EOs emerged as potent antimicrobial agents due to their complex chemical composition and multiple mechanisms of action. EOs function by disrupting the microbial cell membrane and inhibiting the efflux pump, liable for antimicrobial resistance in Gram-negative bacteria. Additionally, various modes of action are to be widely documented, including the interference of peptidoglycan synthesis in the bacterial cell wall through penicillin-binding proteins (PBPs) in Gram-positive bacteria.8 This multi-target approach remarkably reducing the likelihood of antimicrobial resistance when compared to single target antibiotics.
Microbial infections are often accompanied by inflammatory responses characterized by immune cell recruitment and the release of inflammatory mediators, leading to alterations in tissue structure and function. If unresolved, acute inflammation can progress to chronic inflammation, contributing to the pathogenesis of diseases such as cancer, autoimmune disorders, pulmonary conditions, and neurodegenerative diseases.9 Key molecular regulators of inflammation include pro-inflammatory cytokines and enzymes such as Cyclooxygenase-2 (COX-2), inducible Nitric Oxide Synthase (iNOS), Myeloperoxidase (MPO), and transcription factors including Nuclear Factor kappa B (NF-kB) and Peroxisome Proliferator-Activated Receptor-gamma (PPAR-g).10 EOs have been shown to modulate these signaling pathways, resulting in suppressed expression of inflammatory mediators and attenuation of inflammatory responses.11
Oxidative stress, arising from excessive production of reactive oxygen species (ROS) and free radicals, leads to cellular damage and is closely associated with the development of chronic diseases. Bioactive constituents present in EOs, particularly polyphenolic compounds, exhibit strong antioxidant activity by scavenging ROS such as hydroxyl radicals, superoxide radicals, hydrogen peroxide, singlet oxygen, and nitric oxide. Through these mechanisms, EOs help restore redox balance and mitigate oxidative damage at the cellular level.12,13
An important and distinguishing feature of EOs is their synergistic activity, wherein the combined presence of naturally occurring constituents within crude EOs produces enhanced biological effects compared to isolated components evaluated individually. This cooperative interaction among bioactive compounds leads to amplified pharmacological outcomes, underscoring the therapeutic superiority of whole EOs over single-compound formulations.1
Based on these considerations, the EOs selected for this comparative study represent diverse botanical families and distinct phytochemical profiles, each contributing unique biofunctional properties. Eugenol, the principal bioactive constituent of Syzygium aromaticum (clove), exhibits strong antimicrobial, anti-inflammatory, and antioxidant activities attributed to its phenolic structure.14 Rosmarinus officinalis L. (Lamiaceae) is recognized for its rich EO composition and associated bioactivities, particularly antioxidant and antimicrobial effects.15 EOs from Rosa canina L. (Rosaceae) demonstrate notable antioxidant and anti-inflammatory properties, largely due to their high content of phenolic acids, carotenoids, tocopherols, and vitamins.16 Lemongrass essential oil (LEO), derived from the Poaceae family, contains bioactive constituents such as citral, geraniol, citronellol, and germacrene-D, conferring broad pharmacological activities including antibacterial, antifungal, antiviral, anticancer, and antioxidant effects.17 Similarly, EOs obtained from Salvia sclarea L. and Lavandula angustifolia (Lamiaceae) exhibit potent antimicrobial efficacy against predominant bacterial strains.18 This study aims to evaluate the antimicrobial, antioxidant, and anti-inflammatory activities of selected essential oils and to identify their bioactive constituents using GC-MS analysis, correlating phytochemical composition with observed biological effects.
Therapeutic-grade essential oils of clove, lavender, rosemary, rosehip, lemongrass, and clary sage were purchased from AV Herbals Pvt. Ltd., New Delhi. Clinical bacterial strains were obtained from PSG Institute of Medical Sciences & Research, Coimbatore. Culture media and reagents (Mueller-Hinton agar, DMSO, DPPH, etc.) were procured from HiMedia Laboratories, Mumbai. Standard antibiotics, ascorbic acid, and diclofenac sodium were used as controls. GC-MS analysis was performed using a Shimadzu GCMS-QP2010 Plus system.
Collection of essential oils
Six commercially available essential oils, Clove oil (Eugenol, Syzygium aromaticum), Lavender oil (Lavandula angustifolia), Rosemary oil (Rosmarinus officinalis), Rosehip oil (Rosa canina), Lemongrass oil (Cymbopogon citratus), and Clary sage oil (Salvia sclarea) were purchased from certified suppliers (AV Herbals Pvt. Ltd., New Delhi). All oils were of therapeutic grade (>98%) and stored in airtight amber glass vials to protect them from light and oxidative degradation. Owing to their hydrophobic nature, the oils were uniformly dissolved in 1% (v/v) dimethyl sulfoxide (DMSO) prior to use, and the same solvent system was consistently employed across all experimental assays with appropriate solvent controls included.
Collection of microbial strains
Clinical microbial isolates used in this study were archival strains obtained from routine diagnostic procedures at the Department of Microbiology, PSG Institute of Medical Sciences & Research, Coimbatore. All isolates were fully anonymised, and no patient identifiers or associated clinical data were accessed. The pathogens used in this study included Methicillin-resistant Staphylococcus aureus (MRSA), Escherichia coli, Pseudomonas aeruginosa, Klebsiella pneumoniae, Streptococcus pyogenes, and Acinetobacter baumannii. Pure cultures were maintained on nutrient agar slants at 4 °C and sub-cultured periodically to ensure viability for subsequent antimicrobial susceptibility testing.
Assessment of antimicrobial activity of essential oils using agar well diffusion method
The antimicrobial activity of the selected essential oils was evaluated using the agar well diffusion method.18 Broth cultures of test pathogens were adjusted to a 0.5 McFarland standard (~1 × 10x CFU/mL) and swabbed onto Mueller-Hinton agar plates. Wells of 8 mm diameter were aseptically cut and loaded with essential oils at concentrations of 10, 25, 50, 75 and 100 μg/mL, selected based on preliminary screening studies and previously reported effective ranges for essential oils against multidrug-resistant pathogens. A volume of 25 µL of oil was added to each well. The plates were incubated at 37 °C for 24 hours. Chloramphenicol and dimethyl sulfoxide (DMSO) served as the positive and negative controls, respectively. Zones of inhibition were measured post-incubation.
Determination of minimum inhibitory concentration (mic) of essential oils
The MIC of essential oils was determined using the broth microdilution method in a sterile 96-well microtiter plate, following Clinical and Laboratory Standards Institute (CLSI) guidelines with slight modifications for the hydrophobic nature of essential oils. Serial two-fold dilutions of each oil were prepared in Mueller-Hinton broth. Microbial inocula were adjusted to a 0.5 McFarland standard (~1 × 10x CFU/mL). Each well received 100 µL of diluted oil and 100 µL of microbial suspension. Wells containing broth with 1% DMSO served as negative controls, while a standard antibiotic served as the positive control. Following incubation at 37 °C for 24 hrs, absorbance was measured at 600 nm using a microplate reader, and MIC values were defined as the lowest concentration showing no visible microbial growth.
Determination of antioxidant potential by dpph scavenging activity
The antioxidant activity of the essential oils was evaluated using the 2,2-diphenyl-1-picrylhydrazyl (DPPH) assay as described by Eid et al.12 0.5 mM DPPH solution was prepared in methanol. Essential oil samples were prepared at varying concentrations (10, 25, 50, 75, 100, and 150 µg/mL). To 0.5 mL of essential oil, 2.5 mL of DPPH solution was added to make a final volume of 3 mL. Ascorbic acid served as the positive control, and methanol as the negative control. Samples were incubated in the dark at room temperature for 30 minutes, and absorbance was measured at 517 nm using a UV-Vis spectrophotometer. The percentage of scavenging activity was calculated using the formula:
Scavenging activity (%) = Absorbance of Control – Absorbance of Sample / Absorbance of Control × 100
IC50 values were calculated accordingly.
Determination of anti-inflammatory activity by hrbc membrane stabilization assay
In vitro anti-inflammatory activity was assessed using the human red blood cell (HRBC) membrane stabilization assay.19 Fresh human blood (5 mL) was collected from a healthy volunteer after obtaining informed consent. The study involved minimal risk and did not include patient identifiers or clinical interventions; therefore, formal ethical committee approval was not mandatory, in accordance with institutional guidelines. The collected blood was centrifuged at 2500 rpm for 5 min, and the packed cells were washed with an equal volume of isosaline. This washing step was repeated until the supernatant became clear. A 10% (v/v) HRBC suspension was prepared in normal saline and stored at 4 °C until use.
To assess membrane stabilization, 0.5 mL of HRBC suspension was mixed with 1 mL isosaline buffer (pH 7.4), 2 mL of hyposaline (0.25%), and 1 mL of EO at varying concentrations (10, 25, 50, 75, 100 µg/mL). Samples were incubated at 56 °C for 30 minutes and centrifuged at 2500 rpm for 5 minutes. Absorbance was measured at 560 nm, and the percentage of inhibition was calculated as:
Inhibition (%) = [Absorbance of Control – Absorbance of Sample / Absorbance of Control] × 100
Diclofenac sodium served as the positive control, and the solution containing only isosaline buffer without EO was used as the negative control.
Phytochemical profiling of essential oil by gc-ms analysis: insights into bioactive constituents
Gas chromatography-mass spectrometry (GC-MS) analysis of the essential oil combination was performed using a Shimadzu GC-MS-QP2010 Plus instrument with an RXI-5Sil MS capillary column. Helium was used as the carrier gas in linear velocity mode. The injection was done in split mode (10:1), with an injector temperature of 280 °C. The oven temperature was initially held at 70 °C for 4 minutes, then increased at 5 °C/min to 250 °C and held for 10 minutes.
The column flow rate was 1.02 mL/min, total flow 14.4 mL/min, and purge flow 3.0 mL/min. The system was equilibrated for 3.0 minutes. The ion source and interface temperatures were maintained at 200 °C and 280 °C, respectively. The solvent cut time was 2.00 minutes, detector gain was set to 0.97 kV, and the scanning range was m/z 40-600 at a speed of 2000 with a scan time of 0.30 seconds. Compounds were identified by comparing spectra against the NIST 2020 and WILEY 08 libraries, and only peaks with high spectral similarity were considered.20
Synergistic Antimicrobial Efficacy Of Combinatorial Essential Oils Via Agar Disc Diffusion Assay
Based on initial antimicrobial screening, essential oils showing significant activity against both Gram-positive and Gram-negative bacteria (eugenol, rosemary, and rosehip oils) were selected for combination studies.8 A ternary blend was prepared in equal volumes (1:1:1). Sterile discs were loaded with 20 µL of the combination. Chloramphenicol (30 µg) and ketoconazole (30 µg) served as positive controls for bacterial and fungal strains, respectively.
Anti-inflammatory and antioxidant potential of combinatorial essential oils
The antioxidant and anti-inflammatory properties of the combined essential oil formulation were assessed using the DPPH scavenging assay and HRBC membrane stabilization assay, respectively, as described above.
Statistical analysis
All experiments were performed in triplicate, and results are expressed as mean ± standard deviation. Statistical analyses were conducted using one-way or two-way analysis of variance (ANOVA), followed by Tukey’s post-hoc test. Statistical significance was set at p < 0.05, and exact p-values along with appropriate effect sizes (h² for ANOVA) are reported consistently across all result sections. All analyses were performed using IBM SPSS Statistics version 20.0.
Assessment of antimicrobial activity of essential oils using agar well diffusion method
The antimicrobial activity of six essential oils, viz eugenol, rosemary, rosehip, lavender, clary sage, and lemongrass were evaluated using the agar well diffusion method against selected microbial strains. Zones of inhibition (ZOI) were measured in millimetres after a 24-hour incubation at 37 °C. All tests were performed in triplicate (n = 3), and the results are expressed as mean ± standard deviation (Table 1). Each essential oil was tested at concentrations of 10, 25, 50, 75, and 100 µg/mL. A dose-dependent increase in antimicrobial activity was observed, with higher concentrations yielding larger zones of inhibition. Eugenol demonstrated the highest overall efficacy, showing strong inhibitory activity across all tested organisms, including polymicrobial cultures (25.0 ± 0.8 mm at 100 µg/ml). It also exhibited large inhibition zones against K. pneumoniae (23.0 ± 0.8 mm), E. coli (21.7 ± 0.5 mm), and A. baumannii (21.3 ± 0.5 mm), likely due to its phenolic structure that disrupts bacterial membranes and interferes with cellular processes.
Table (1):
Antimicrobial activity of individual essential oils against clinically relevant bacterial pathogens assessed by the agar well diffusion method
| Pathogens | EO’s | Concentration of EO (µg/mL) | |||||
|---|---|---|---|---|---|---|---|
| 10 | 25 | 50 | 75 | 100 | PC | ||
| Average zone of inhibition (in mm) | |||||||
| MRSA | EU | 18.0 ± 0.8 | 19.3 ± 0.9 | 20.3 ± 0.5 | 20.3 ± 0.5 | 23.0 ± 0.8 | 22.0 ± 0.8 |
| RM | 16.0 ± 0.8 | 17.3 ± 0.5 | 20.7 ± 0.5 | 21.0 ± 0.8 | 22.7 ± 0.5 | 21.7 ± 0.9 | |
| RH | 13.3 ± 0.5 | 15.0 ± 0.8 | 17.3 ± 0.5 | 19.0 ± 0.8 | 21.7 ± 0.5 | 21.0 ± 0.8 | |
| LV | 13.0 ± 0.8 | 14.7 ± 0.9 | 16.3 ± 0.5 | 18.3 ± 0.5 | 20.7 ± 0.9 | 20.0 ± 0.8 | |
| CS | 14.0 ± 0.8 | 15.3 ± 0.5 | 16.0 ± 0.8 | 20.7 ± 0.5 | 22.0 ± 0.8 | 21.3 ± 0.5 | |
| LG | 18.3 ± 0.5 | 19.3 ± 0.5 | 20.7 ± 0.5 | 21.0 ± 0.8 | 21.3 ± 1.2 | 21.0 ± 0.8 | |
| S. pyogenes | EU | 18.3 ± 0.5 | 19.0 ± 0.8 | 19.3 ± 0.5 | 20.7 ± 0.9 | 21.7 ± 0.5 | 23.0 ± 0.8 |
| RM | 15.0 ± 0.8 | 16.3 ± 0.5 | 17.7 ± 0.5 | 18.3 ± 0.5 | 20.7 ± 0.5 | 21.3 ± 0.5 | |
| RH | 12.3 ± 0.5 | 13.7 ± 0.5 | 16.3 ± 0.5 | 18.7 ± 0.5 | 21.0 ± 0.8 | 20.7 ± 0.5 | |
| LV | 13.3 ± 0.5 | 15.3 ± 0.9 | 16.7 ± 0.5 | 17.7 ± 0.9 | 21.3 ± 0.9 | 21.0 ± 0.8 | |
| CS | 15.3 ± 0.5 | 16.0 ± 0.8 | 17.7 ± 0.5 | 19.3 ± 0.5 | 20.0 ± 0.8 | 23.3 ± 1.2 | |
| LG | 12.7 ± 0.9 | 16.0 ± 0.8 | 17.7 ± 0.9 | 20.0 ± 0.8 | 21.3 ± 0.5 | 21.0 ± 0.8 | |
| E. coli | EU | 15.7 ± 0.9 | 16.3 ± 0.5 | 19.0 ± 0.8 | 20.7 ± 0.5 | 21.7 ± 0.5 | 19.0 ± 0.8 |
| RM | 18.0 ± 0.8 | 19.3 ± 0.5 | 20.0 ± 0.8 | 20.3 ± 0.5 | 21.3 ± 0.9 | 20.0 ± 0.8 | |
| RH | 16.0 ± 0.8 | 17.3 ± 0.5 | 18.7 ± 0.9 | 19.3 ± 0.5 | 22.3 ± 1.2 | 21.3 ± 0.5 | |
| LV | 18.0 ± 0.8 | 19.7 ± 0.5 | 21.3 ± 0.5 | 22.0 ± 0.8 | 23.0 ± 0.8 | 20.7 ± 0.9 | |
| CS | 16.0 ± 0.8 | 17.3 ± 0.5 | 19.0 ± 0.8 | 20.3 ± 0.5 | 21.7 ± 0.5 | 20.0 ± 0.8 | |
| LG | 16.0 ± 0.8 | 18.3 ± 0.5 | 19.7 ± 0.5 | 21.0 ± 0.8 | 21.7 ± 0.5 | 19.0 ± 0.8 | |
| K. pneumoniae | EU | 17.3 ± 0.9 | 18.0 ± 0.8 | 21.0 ± 0.5 | 21.7 ± 0.5 | 23.0 ± 0.8 | 22.7 ± 0.5 |
| RM | 14.7 ± 0.5 | 15.7 ± 0.5 | 16.3 ± 0.9 | 19.0 ± 0.8 | 19.7 ± 0.5 | 20.7 ± 0.9 | |
| RH | 13.7 ± 0.5 | 14.7 ± 0.5 | 16.0 ± 0.8 | 19.3 ± 0.5 | 22.0 ± 0.8 | 21.3 ± 0.5 | |
| LV | 11.0 ± 0.8 | 13.0 ± 0.8 | 13.7 ± 0.5 | 15.0 ± 0.8 | 17.3 ± 0.5 | 18.3 ± 0.5 | |
| CS | 14.3 ± 0.5 | 15.7 ± 0.5 | 17.3 ± 0.5 | 19.3 ± 0.5 | 20.3 ± 0.5 | 21.7 ± 0.5 | |
| LG | 11.0 ± 0.8 | 12.3 ± 0.5 | 14.7 ± 0.5 | 16.7 ± 0.9 | 20.0 ± 0.8 | 19.7 ± 0.5 | |
| A. baumannii | EU | 14.3 ± 0.9 | 16.3 ± 0.5 | 18.3 ± 0.5 | 20.7 ± 0.5 | 21.3 ± 0.5 | 18.3 ± 0.5 |
| RM | 15.0 ± 0.8 | 17.3 ± 0.5 | 19.0 ± 0.8 | 20.3 ± 0.5 | 22.3 ± 0.5 | 21.3 ± 0.5 | |
| RH | 12.0 ± 0.8 | 12.7 ± 0.9 | 14.0 ± 0.8 | 16.3 ± 0.9 | 18.3 ± 0.5 | 16.7 ± 0.5 | |
| LV | 14.0 ± 0.8 | 16.7 ± 0.5 | 19.0 ± 0.8 | 20.3 ± 0.5 | 22.3 ± 0.9 | 21.3 ± 0.5 | |
| CS | 11.7 ± 0.5 | 12.7 ± 0.5 | 14.7 ± 0.5 | 17.3 ± 0.5 | 21.0 ± 0.8 | 20.7 ± 0.9 | |
| LG | 15.3 ± 0.9 | 17.3 ± 0.9 | 18.7 ± 0.5 | 18.0 ± 0.8 | 19.0 ± 0.0 | 20.3 ± 0.5 | |
| P. aeruginosa | EU | 15.0 ± 0.8 | 16.3 ± 0.5 | 17.7 ± 0.5 | 18.7 ± 0.5 | 20.3 ± 0.5 | 19.0 ± 0.8 |
| RM | 14.3 ± 0.5 | 16.0 ± 0.8 | 16.7 ± 0.5 | 19.0 ± 0.8 | 21.0 ± 0.8 | 19.7 ± 0.9 | |
| RH | 13.7 ± 0.5 | 15.0 ± 0.8 | 17.0 ± 0.8 | 17.7 ± 0.5 | 20.0 ± 0.8 | 19.7 ± 0.5 | |
| LV | 13.7 ± 0.5 | 15.0 ± 0.8 | 16.0 ± 0.8 | 17.3 ± 0.5 | 19.7 ± 0.5 | 18.3 ± 0.5 | |
| CS | 12.3 ± 0.5 | 13.7 ± 0.5 | 14.3 ± 0.5 | 16.0 ± 0.8 | 17.7 ± 0.5 | 18.7 ± 0.5 | |
| LG | 11.3 ± 0.5 | 11.7 ± 0.9 | 13.7 ± 0.5 | 15.0 ± 0.8 | 17.0 ± 0.8 | 19.0 ± 0.8 | |
| Polymicrobial culture | EU | 20.3 ± 0.5 | 21.0 ± 0.8 | 22.3 ± 0.9 | 22.3 ± 0.5 | 25.0 ± 0.8 | 24.3 ± 0.5 |
| RM | 13.0 ± 0.8 | 14.7 ± 0.5 | 16.0 ± 0.8 | 17.7 ± 0.5 | 20.3 ± 0.5 | 20.3 ± 0.5 | |
| RH | 14.3 ± 0.5 | 15.3 ± 0.5 | 15.0 ± 0.8 | 19.7 ± 0.5 | 20.7 ± 0.5 | 20.3 ± 0.5 | |
| LV | 14.7 ± 0.5 | 16.0 ± 0.8 | 18.3 ± 0.9 | 19.0 ± 0.8 | 21.7 ± 0.5 | 21.0 ± 0.8 | |
| CS | 12.7 ± 0.5 | 14.3 ± 0.5 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 21.0 ± 0.8 | |
| LG | 0.0 ± 0.0 | 10.7 ± 0.5 | 14.7 ± 0.5 | 16.0 ± 0.8 | 17.3 ± 0.5 | 19.7 ± 0.5 | |
EU: Eugenol, RM: Rosemary oil, RH: Rosehip oil, LV: Lavender oil, CS: Clary sage oil, LG: Lemongrass oil, PC: Positive Control
Rosemary oil showed significant antimicrobial activity, particularly against MRSA (22.7 ± 0.5 mm), E. coli (21.3 ± 0.9 mm), and A. baumannii (22.3 ± 0.5 mm). Its bioactivity is attributed to components like 1,8-cineole and a-pinene.6 Rosehip oil displayed moderate activity, especially against E. coli (22.3 ± 1.2 mm) and K. pneumoniae (22.0 ± 0.8 mm), with slightly reduced efficacy against P. aeruginosa and A. baumannii. Its effects are possibly linked to its high content of polyunsaturated fatty acids and antioxidants.16 Lavender oil was effective against both Gram-negative and Gram-positive strains, notably E. coli (23.0 ± 0.8 mm), A. baumannii (22.3 ± 0.9 mm), and MRSA (20.7 ± 0.9 mm), attributed to its main constituents namely linalool and linalyl acetate.6,18 Clary sage exhibited moderate efficacy against monocultures such as MRSA (22.0 ± 0.8 mm) and A. baumannii (21.0 ± 0.8 mm) but showed no inhibition against polymicrobial cultures at concentrations above 25 µg/ml, suggesting potential antagonistic effects or degradation in complex environments.21 Lemongrass oil showed selective activity, particularly against MRSA (21.3 ± 1.2 mm), S. pyogenes (21.3 ± 0.5 mm), and E. coli (21.7 ± 0.5 mm), while its effect on polymicrobial cultures was relatively weak (17.3 ± 0.5 mm at 100 µg/ml). This may be due to the variable stability or efficacy of citral, its principal active compound.17
Among the tested essential oils, eugenol exhibited the most potent and consistent broad-spectrum antimicrobial activity, including against polymicrobial cultures, likely due to its phenolic structure that disrupts bacterial membranes and cellular functions. Rosemary oil also showed strong efficacy, particularly against multidrug-resistant strains, attributable to bioactive monoterpenes such as 1,8-cineole and a-pinene. Lavender and rosehip oils demonstrated moderate, strain-dependent activity, reflecting their mixed phytochemical composition and indirect antimicrobial mechanisms. In contrast, clary sage and lemongrass oils displayed selective or reduced efficacy in polymicrobial settings, possibly due to limited stability or antagonistic interactions in complex microbial environments. Overall, these findings highlight the importance of selecting essential oils based on infection type and microbial complexity rather than relying on uniform antimicrobial performance.
Determination of minimum inhibitory concentration of essential oils
The minimum inhibitory concentrations (MICs) of six essential oils, viz, eugenol, rosemary, rosehip, lavender, clary sage, and lemongrass were evaluated against selected clinically relevant pathogens using the broth microdilution method, with a concentration range from 1000 to 1 µg/mL. The MIC values are presented in Table 2.
Table (2):
Minimum Inhibitory Concentration (MIC) of different Essential Oils against Test Microorganisms
| Test organism | Minimum Inhibitory concentration of Selected Essential Oils (µg/mL) | |||||
|---|---|---|---|---|---|---|
| EU | RM | RH | LV | CS | LG | |
| MRSA | 62.5 | 62.5 | 250 | 125 | 250 | 62.5 |
| S. pyogenes | 62.5 | 62.5 | 62.5 | 250 | 250 | 250 |
| E. coli | 62.5 | 62.5 | 250 | 500 | 250 | 250 |
| K. pneumoniae | 125 | 62.5 | 500 | 250 | 500 | 250 |
| P. aeruginosa | 125 | 62.5 | 250 | 125 | 500 | 125 |
| A. baumannii | 125 | 125 | 250 | 250 | 250 | 250 |
| Polymicrobial | 125 | 250 | 125 | 125 | 500 | 250 |
The results revealed notable variations in antimicrobial efficacy among the oils. Eugenol demonstrated the most potent and consistent activity, exhibiting MIC values of 62.5 µg/mL against MRSA, Streptococcus pyogenes, and E. coli, and 125 µg/mL against A. baumannii, P. aeruginosa, K. pneumoniae, and polymicrobial cultures. This broad-spectrum effectiveness can be attributed to eugenol’s phenolic structure, which enables it to disrupt bacterial membranes, inhibit enzymes, and interfere with quorum sensing.
Rosemary oil displayed comparable antimicrobial potency, with MICs of 62.5 µg/mL for MRSA, Streptococcus, E. coli, P. aeruginosa, and K. pneumoniae, and 125 µg/mL for A. baumannii and polymicrobial cultures. Its efficacy is likely due to active constituents such as 1,8-cineole, camphor, and a-pinene, which are known to compromise microbial cell wall integrity and metabolic processes. Lemongrass oil showed selective activity, with MICs of 62.5 µg/mL against MRSA, 125 µg/mL against P. aeruginosa, and 250 µg/mL for the remaining organisms. The presence of citral, a key bioactive compound, may explain its effectiveness, although reduced efficacy against certain strains suggests limitations in complex infections.
Lavender oil exhibited variable antimicrobial activity, with MIC values ranging from 125 µg/mL (MRSA, Pseudomonas) to 500/ µg/mL (E. coli). Its inconsistent performance may stem from variability in its phytochemical composition and relatively lower concentrations of antimicrobial constituents like linalool and linalyl acetate, or possible interference by antagonistic components. Rosehip oil showed uniform inhibitory effects at 250 µg/mL for most pathogens, with reduced efficacy against K. pneumoniae (MIC: 500 µg/mL). Despite the higher MICs, its activity across a wide range of organisms suggests potential therapeutic value, possibly owing to its rich profile of flavonoids, phenolic acids, and volatile compounds.22 Clary sage oil demonstrated the weakest antimicrobial activity, with MICs of 250 µg/mL against MRSA, Streptococcus, E. coli, and A. baumannii, and 500 µg/mL for P. aeruginosa, K. pneumoniae, and polymicrobial cultures. The diminished performance may reflect either a low concentration of active agents or the presence of constituents that counteract antimicrobial action.6,18
Among the tested strains, P. aeruginosa generally showed higher resistance, consistent with its well-established intrinsic mechanisms such as efflux pumps and biofilm formation. Interestingly, despite the complexity of polymicrobial cultures, eugenol and rosemary oil maintained appreciable activity, indicating their potential in managing mixed-pathogen infections.18,23 The lower MIC values observed for eugenol and rosemary oil against MDR organisms indicate high antimicrobial potency at reduced concentrations, which is clinically advantageous for minimizing toxicity, lowering effective doses, and reducing resistance pressure. Their sustained activity against polymicrobial cultures further supports their potential translational application as adjunct or alternative agents in managing complex and drug-resistant infections. In summary, the MIC findings confirm the superior antimicrobial efficacy of eugenol, followed closely by rosemary oil, across a spectrum of Gram-positive and Gram-negative bacteria. The differential susceptibility patterns highlight the need for careful selection of essential oils based on the target organism and the intended clinical application.
Determination of antioxidant potential of essential oils by dpph scavenging activity
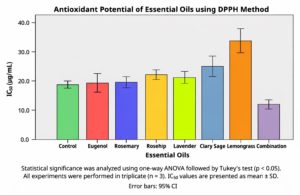
The antioxidant potential of the selected essential oils and their combination was assessed using the DPPH radical scavenging assay. The IC50 values, representing the concentration required to inhibit 50% of DPPH radicals, were calculated for each group (n = 3), and the results are summarized in Figure 1. Statistical analysis using one-way ANOVA indicated significant differences among the treatment groups (F7,16 = 121.271, p < 0.001). Levene’s test confirmed homogeneity of variances (F7,16 = 0.873, P = 0.548).
Figure 1. Determination of Antioxidant activity of Essential oils and combination using DPPH scavenging assay
The mean IC50 values (µg/mL ± SD) were as follows: Combination (11.78 ± 0.70), Control (18.39 ± 0.56), Eugenol (19.12 ± 1.08), Rosemary (19.59 ± 0.75), Lavender (20.92 ± 0.79), Rosehip (21.78 ± 0.67), Clary sage (24.58 ± 1.30), and Lemongrass (33.61 ± 1.52). The overall mean IC50 across treatments was 21.22 ± 5.98/ µg/mL, with values ranging from 11.05 to 34.92/ µg/mL.
Post hoc analysis using Tukey’s HSD test revealed five distinct homogeneous subsets, confirming that the combination treatment had significantly greater antioxidant activity than all other groups (P < 0.001). Lemongrass exhibited the weakest antioxidant potential. Based on IC50 values, the antioxidant potency of the treatments can be ranked as:
Combination > Control > Eugenol > Rosemary > Lavender > Rosehip > Clary sage > Lemongrass.
Further comparisons using Fisher’s LSD test supported most pairwise differences and identified additional significant contrasts not captured by the more conservative Tukey’s test. These findings underscore the substantial variability in antioxidant capacity among the tested essential oils. Eugenol and rosemary oils demonstrated relatively strong antioxidant activity, with IC50 values of 19.12 µg/mL and 19.59 µg/mL, respectively. Eugenol, a phenolic compound abundant in clove oil, is well-known for its ability to donate hydrogen atoms via its hydroxyl group, thereby neutralizing free radicals.24 Similarly, rosemary essential oil contains potent antioxidants such as rosmarinic acid, carnosic acid, and other phenolic diterpenes that enhance its radical scavenging capability.25
Lavender (IC50 = 20.92 µg/mL) and rosehip oil (21.78 µg/mL) exhibited moderate antioxidant activity, consistent with their reported phenolic content and traditional medicinal use. These oils are rich in monoterpenes and select phenolic compounds, though at lower levels than those found in eugenol or rosemary.26 On the other hand, clary sage (24.58 µg/mL) and lemongrass (33.61 µg/mL) showed comparatively weaker antioxidant performance. The lower activity of Lemongrass can be attributed to its citral-based composition, which, although possessing antioxidant properties, is less effective than phenolic constituents in scavenging free radicals. The reduced antioxidant potential of these oils likely stems from their chemical profiles, which are dominated by monoterpene hydrocarbons rather than phenolic structures.20
Notably, the combination of essential oils displayed the lowest IC50 value (11.78 µg/mL), indicating synergistic antioxidant effects. This superior activity suggests that blending essential oils may enhance radical scavenging through complementary or additive mechanisms involving multiple bioactive compounds. Previous studies have reported similar synergism in essential oil mixtures, where the total antioxidant activity exceeds that of the individual components.27 In conclusion, the DPPH assay results indicate that essential oils vary significantly in their antioxidant capacities, influenced by their phytochemical composition. While eugenol and rosemary show strong individual performance, combining oils may offer an effective strategy to maximize antioxidant potential for therapeutic applications.
Determination of anti-inflammatory activity by hrbc membrane stabilization assay
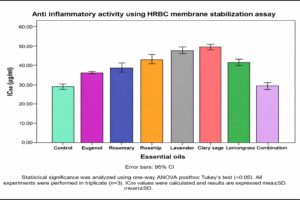
The anti-inflammatory activity of the selected essential oils and their combination was evaluated using the human red blood cell (HRBC) membrane stabilization assay. The IC50 values, representing the concentration required to inhibit 50% hemolysis, were determined for each treatment group (n = 3). The results are presented in Figure 2, with statistical analysis conducted using one-way ANOVA followed by Tukey’s HSD post hoc test.
Figure 2. Determination of anti-inflammatory activity of Essential oils and combination by HRBC membrane stabilization assay
The mean IC50 values (µg/mL ± SD) ranged from 28.19 ± 0.75 µg/mL (Combination) to 49.67 ± 0.56 µg/mL (Clary sage). The control group exhibited an IC50 of 28.48 ± 0.62 µg/mL, indicating that lower IC50 values correlated with higher membrane stabilization potential and, hence, greater anti-inflammatory efficacy. Levene’s test confirmed the assumption of homogeneity of variances (F7,16 = 0.854, P = 0.561), validating the ANOVA, which revealed highly significant differences among the treatments (F7,16 = 386.778, P < 0.001), with an effect size of ח² = 0.994, indicating that 99.4% of the variance was due to treatment differences. Tukey’s HSD post hoc test identified five homogeneous subsets. The Combination treatment (28.19 µg/mL) and Control (28.48 µg/mL) formed one subset with no significant difference (P = 1.000), suggesting equivalent membrane-stabilizing activity. Eugenol (36.18 µg/mL), Rosemary (38.67 µg/mL), and Lemongrass (41.70 µg/mL) formed distinct subsets, demonstrating moderate potency. The least effective treatments were Rosehip (43.11 µg/mL), Lavender (47.94 µg/mL), and Clary sage (49.67 µg/mL), which required higher concentrations to achieve comparable stabilization effects.
These findings highlight significant variability in the anti-inflammatory properties of essential oils. Among the individual treatments, Eugenol exhibited the highest potency, likely due to its phenolic hydroxyl group enabling efficient interaction with phospholipid bilayers through hydrogen bonding and hydrophobic interactions. These mechanisms stabilize the red blood cell membrane and reduce hemolysis, aligning with eugenol’s known anti-inflammatory and antioxidant properties.28
Rosemary and Lemongrass showed moderate efficacy. Their activity is attributed to monoterpenes such as 1,8-cineole and citral, respectively, which protect cellular membranes by reducing lipid peroxidation and protein denaturation.9,17 In contrast, Clary sage, Lavender, and Rosehip demonstrated weaker membrane-stabilizing capacity. Their higher IC50 values suggest either a lower concentration of effective bioactives or different modes of action that are less suited to direct membrane stabilization.16 Notably, the combination treatment, although yielding the lowest mean IC50 (28.19 µg/mL), was statistically indistinguishable from the control. This suggests that anticipated synergistic effects were absent. Instead, antagonistic interactions or dilution effects within the multi-component blend may have masked the individual potencies of its constituents, a finding contrary to previous expectations for enhanced efficacy in essential oil mixtures.
The substantial F-statistic and high effect size strongly indicate that treatment type has a decisive impact on anti-inflammatory activity, with minimal variability within groups. These results underscore the therapeutic promise of Eugenol, Rosemary, and Lemongrass as effective membrane stabilizers and support their further exploration in inflammation models. Additionally, the findings warrant deeper investigation into the dose-dependent interactions in essential oil combinations to optimize formulations for synergistic rather than antagonistic outcomes.
Phytochemical profiling of essential oil by gc-ms analysis: insights into bioactive constituents
Gas Chromatography-Mass Spectrometry (GC-MS) analysis was employed to identify and quantify the chemical constituents of the essential oil sample. A total of 40 compounds were detected, encompassing a wide range of monoterpenes, sesquiterpenes, oxygenated derivatives, and fatty acid esters, with retention times spanning from 3.594-47.175 minutes. The identified compounds, along with their retention times and relative peak area percentages, are detailed in Table 3 and visualized in the chromatogram in Figure 3.
Table (3):
GC-MS Profile of Essential Oil Constituents
Peak |
RT (min) |
Area (%) |
Compound Name |
Present in |
Known Properties |
|---|---|---|---|---|---|
1 |
3.594 |
0.11 |
3-Hexen-1-ol |
Rosehip oil |
Antimicrobial & Antioxidant |
4 |
5.101 |
0.91 |
3-Carene |
Rosemary oils |
Antimicrobial, penetration enhancer |
6 |
5.38 |
26.17 |
α-Pinene |
Rosemary, Clove oil |
Antimicrobial, Anti-inflammatory & Bronchodilator |
7 |
5.827 |
4.96 |
Camphene |
Rosemary |
Antimicrobial, expectorant, antioxidant |
9 |
6.6 |
3.89 |
β-Pinene |
Rosemary, Rosehip oil |
Antimicrobial, bronchodilator, anti-inflammatory |
10 |
6.86 |
0.36 |
Geranyl isovalerate |
Rosemary, Rosehip oil |
Fragrance component, antimicrobial |
11 |
7.42 |
0.22 |
α-Phellandrene |
Rosehip, Eucalyptus oils |
Antimicrobial, antifungal |
12 |
7.496 |
0.29 |
trans-β-Ocimene |
Rosemary oil |
Antimicrobial, fragrance, insect repellent |
14 |
7.994 |
1.44 |
p-Cymene |
Antimicrobial, anti-inflammatory, antioxidant |
|
15 |
8.134 |
2.74 |
D-Limonene |
Rosemary oil |
Antimicrobial, antioxidant, anti-inflammatory |
16 |
8.247 |
30.22 |
Eucalyptol |
Rosemary oil |
Antimicrobial, anti-inflammatory, expectorant, bronchodilator |
18 |
9.875 |
0.19 |
(+)-4-Carene |
Pine, Rosemary oils |
Antimicrobial, anti-inflammatory |
19 |
10.386 |
1.17 |
Linalool |
Rosemary oil |
Antimicrobial, sedative, anti-inflammatory, analgesic |
20 |
10.896 |
0.12 |
Isobornyl formate |
Rosemary, Pine oils |
Fragrance component, antimicrobial |
21 |
11.825 |
0.44 |
Camphor |
Rosemary oil |
Antimicrobial, analgesic, anti-inflammatory, antiviral |
23 |
12.413 |
10.36 |
Isoborneol |
Rosemary |
Antimicrobial, anti-inflammatory, antiviral |
24 |
12.917 |
0.22 |
Terpinen-4-ol |
Clove oil |
Antimicrobial, anti-inflammatory, antifungal |
26 |
13.508 |
1.21 |
Ascaridole |
Chenopodium oil |
Antimicrobial, anthelmintic, antifungal |
27 |
14.816 |
0.31 |
Linalyl acetate |
Rosehip oil |
Antimicrobial, fragrance, sedative |
28 |
14.968 |
0.05 |
2,6-Octadien-1-ol, 3,7-dimethyl |
Rosehip oil |
Fragrance component, Antimicrobial, Antioxidant |
29 |
15.931 |
0.25 |
Bornyl acetate |
Rosemary, Rosehip oil |
Antimicrobial, fragrance, anti-inflammatory |
30 |
17.603 |
0.15 |
α-Terpinyl acetate |
Rosehip oil |
Antimicrobial, fragrance, antifungal |
32 |
19.614 |
0.09 |
Caryophyllene |
Clove oil |
Anti-inflammatory, antimicrobial, analgesic |
33 |
20.476 |
7.05 |
Eugenol |
Clove oil |
Antimicrobial, anti-inflammatory, analgesic, antioxidant |
35 |
31.919 |
0.29 |
l-(+)-Ascorbic acid 2, 6-dihexadecanoate |
Rosemary oil |
Antioxidant, stabilizer |
36 |
35.24 |
0.61 |
6-Octadecenoic acid, (Z)- |
Rosemary oil, Rosehip oil |
Emollient, antimicrobial |
37 |
35.327 |
0.57 |
Oleic Acid |
Rosehip |
Emollient, penetration enhancer, anti-inflammatory |
38 |
35.668 |
0.17 |
Octadecanoic acid |
Rosemary oil, Rosehip oil |
Emollient, antimicrobial |
40 |
47.175 |
0.43 |
Stigmast-5-en-3-ol |
Plant sterols |
Anti-inflammatory, wound healing, skin barrier repair |
The analysis revealed a complex and pharmacologically rich composition, with eucalyptol (1,8-cineole) being the most abundant compound at 30.22% (RT = 8.247 min), followed by a-pinene at 26.17% (RT = 5.38 min). Together, these two monoterpenes accounted for over 56% of the total oil content, signifying their central role in the oil’s bioactivity. Other notable constituents included isoborneol (10.36%), eugenol (7.05%), camphene (4.96%), and β-pinene (3.89%). Minor constituents comprised D-limonene (2.74%), 3-cyclohexene-1-methanol (2.70%), linalool (1.17%), p-cymene (1.44%), and (+)-3-carene (0.91%). Fatty acids such as oleic acid (0.57%) and 6-octadecenoic acid (0.61%) were also identified, indicating the presence of lipid-derived components.
The dominance of eucalyptol is particularly significant, given its well-established antimicrobial, anti-inflammatory, and mucolytic activities, which make it a promising agent for wound healing and respiratory applications.29 a-Pinene, the second major constituent, is a monoterpene hydrocarbon known for its broad-spectrum antimicrobial and anti-inflammatory effects, and its ability to enhance skin permeability, thus improving the topical delivery of active compounds.
Isoborneol, a monoterpenoid alcohol (10.36%), contributes to the oil’s antiviral and antibacterial properties, while eugenol (7.05%) a phenolic compound commonly found in clove oil adds antioxidant, antimicrobial, and analgesic properties.6 Additional compounds such as camphene, b-pinene, and D-limonene are known for promoting transdermal penetration and supporting tissue regeneration.24,27
The oxygenated monoterpenes, which form the most pharmacologically significant fraction, are dominated by eucalyptol, followed by camphor (0.44%) and isoborneol. These components are associated with anti-inflammatory, bronchodilatory, and analgesic activities. Linalool (1.17%) and linalyl acetate (0.31%), although present in smaller amounts, are known to exhibit antibacterial, sedative, and antioxidant effects.11 The identification of bornyl acetate (0.25%) also suggests a contribution to both aroma and therapeutic functionality.
Phenolic compounds, particularly eugenol, were identified at two retention times (0.44% and 7.05%), reaffirming its key role in the oil’s synergistic antimicrobial and analgesic effects.1 Despite its lower abundance, the presence of caryophyllene (0.09%), a sesquiterpene, adds further value due to its anti-inflammatory and cannabinoid receptor-binding capabilities.
The detection of higher molecular weight constituents such as stigmast-5-en-3-ol (0.43%) – a phytosterol – suggests potential for wound healing, skin barrier repair, and improved oil stability.30 Similarly, fatty acids like oleic acid (0.57%) not only stabilize the oil but may also enhance penetration and bioavailability of active constituents.31
Overall, the GC-MS chemical fingerprint illustrates a well-balanced profile dominated by eucalyptol and a-pinene, complemented by a diverse range of bioactive constituents with known antimicrobial, antioxidant, anti-inflammatory, and transdermal-enhancing effects. The combination of these compounds likely contributes to the observed synergistic therapeutic potential, supporting the oil’s traditional uses and its application in modern medicinal and topical formulations.
Synergistic antimicrobial efficacy of combinatorial essential oils using agar disc diffusion assay
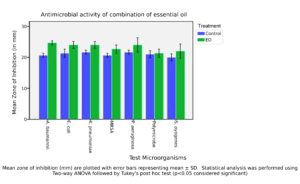
The antimicrobial efficacy of a combinatorial essential oil (EO) formulation was evaluated against seven clinically significant bacterial strains using the agar disc diffusion method. Zones of inhibition were measured in millimetres and compared to control treatments, with results summarized in Figure 4. Statistical significance was determined using two-way ANOVA followed by Tukey’s post hoc test (P < 0.05). The EO combination exhibited significantly enhanced antimicrobial activity compared to controls for A. baumannii (24.67 ± 0.57 mm vs. 20.67 ± 0.57 mm), E. coli (24.00 ± 1.00 mm vs. 21.33 ± 1.15 mm), K. pneumoniae (24.00 ± 1.00 mm vs. 21.67 ± 0.57 mm), and MRSA (22.67 ± 1.15 mm vs. 20.67 ± 0.57 mm). These differences were statistically significant (P < 0.05), confirming the superior antibacterial potential of the EO blend.
Figure 4. Antimicrobial activity of essential oil combination against test microorganisms using agar disc diffusion assay
Although increases in inhibition zones were also observed for P. aeruginosa, S. pyogenes, and polymicrobial cultures, these were not statistically significant (p > 0.05), likely due to greater response variability or inherent resistance mechanisms. The order of microbial susceptibility to the EO combination was as follows: A. baumannii > E. coli > K. pneumoniae = P. aeruginosa > MRSA = S. pyogenes > Polymicrobial. The two-way ANOVA confirmed that both treatment type and microbial strain had significant effects on antimicrobial outcomes (p < 0.05), with robust interaction effects validating the influence of EO combinations across multiple pathogens.
These findings demonstrate that the EO combination offers a broad-spectrum antimicrobial effect, particularly potent against multidrug-resistant Gram-negative bacteria, such as A. baumannii and E. coli, as well as the Gram-positive MRSA strain. This enhanced efficacy likely results from synergistic interactions among key bioactive components including eugenol, 1,8-cineole, geraniol, linalool, and citral, which are known to disrupt bacterial membranes, interfere with energy metabolism, and inhibit biofilm formation.32
Consistent with prior studies,3,28 the combination of phenolic-rich and monoterpene-based essential oils appears to result in additive or synergistic effects, improving efficacy over individual oil applications. In the case of A. baumannii, a pathogen known for efflux pump mechanisms and membrane impermeability, the EO combination may enhance permeability and exert cumulative stress on microbial targets.8
The enhanced antimicrobial activity observed with essential oil combinations underscores the importance of synergistic interactions in combating multidrug-resistant pathogens. Although only modest improvements were noted against highly resilient organisms such as Pseudomonas aeruginosa and polymicrobial cultures, these trends suggest that formulation limitations may restrict efficacy rather than intrinsic antimicrobial potential. Future formulation strategies offer promising avenues to overcome these constraints.33 Nanoencapsulation methods, including polymeric nanoparticles, lipid-based nanocarriers, and nanoemulsions, can improve EO stability, protect volatile bioactive compounds from degradation, improve aqueous dispersibility, and enable sustained release at the site of infection. These systems can also enhance penetration across bacterial membranes and biofilms, which are major contributors to resistance in MDR pathogens. Additionally, integration of EOs into hydrogel-based delivery platforms could enable localized, controlled release while maintaining effective concentrations over prolonged periods, particularly for topical applications such as wound infections, burns, and diabetic ulcers. Hydrogels may further support synergistic interactions between EO components and improve biocompatibility and patient compliance. Therefore, the observed synergistic antimicrobial trends, combined with advances in nanotechnology and biomaterial-based delivery systems, support the future development of EO-based formulations as adjunct or alternative therapies for managing resistant and polymicrobial infections.
This study demonstrated the concentration-dependent antimicrobial, antioxidant, and anti-inflammatory activities of six essential oils, viz. eugenol, rosemary, rosehip, lavender, clary sage, and lemongrass and their combination. Eugenol showed the highest individual efficacy, while the combinatorial formulation exhibited enhanced antimicrobial activity, particularly against multidrug-resistant strains like A. baumannii and MRSA. The EO combination also showed strong antioxidant capacity (lowest IC50) and comparable anti-inflammatory activity to standard controls. GC-MS profiling identified key bioactive compounds such as eucalyptol, a-pinene, isoborneol, and eugenol, which likely contribute to the observed therapeutic effects.
These findings highlight the broad-spectrum potential of essential oils, especially in combination, as promising natural alternatives or adjuncts to conventional therapies. Further research should explore formulation optimization, mechanisms of action, and in vivo efficacy to advance clinical applications.
ACKNOWLEDGMENTS
The authors would like to thank the Management and the DST-FIST Scheme, Department of Microbiology at PSG College of Arts & Science, Coimbatore, for providing the necessary facilities and continuous support to carry out this research work.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
VD conceptualized the study. MP and RR supervised the study and provided resources. SD performed data collection. VD and GC designed the experiments. VD and SD performed the experiments. VD and SD performed preliminary data analysis. VD and GC performed data interpretation. VD wrote the manuscript. GC revised the manuscript and provided technical and intellectual input. MP and RR reviewed and approved the final manuscript for publication.
FUNDING
None.
DATA AVAILABILITY
The data generated and analysed during this study are included in the manuscript, and the data supporting the findings of this study are available from the corresponding author upon reasonable request
ETHICS STATEMENT
Not applicable.
INFORMED CONSENT
Written informed consent was obtained from the participant before enrolling in the study.
- Awojide SH, Fadunmade EO, Adegboye AA, et al. A comparative study on the synergistic activities of fractions and crude essential oil of Syzygium aromaticum. Bull Natl Res Cent. 2024;48(1):7.
Crossref - de Sousa DP, de Assis Oliveira F, Arcanjo DDR, et al. Essential Oils: Chemistry and Pharmacological Activities—Part II. Biomedicines. 2024;12(6):1185.
Crossref - Hulankova R. Methods for Determination of Antimicrobial Activity of Essential Oils In Vitro—A Review. Plants. 2024;13(19):2784.
Crossref - Mitoshi M, Kuriyama I, Nakayama H, et al. Suppression of allergic and inflammatory responses by essential oils derived from herbal plants and citrus fruits. Int J Mol Med. 2014;33(6):1643-1651.
Crossref - Wu J, Hu W, Chen J, Hu J, Ke C, Sheng Z. Characterizing the essential oil composition and assessing the antioxidant and antimicrobial properties of two compositae taxa: Gerbera delavayi Franch. and Gerbera piloselloides (L.) Cass. Front Plant Sci. 2025;16:1527525.
Crossref - Puvaca N, Milenkoviז J, Galonja Coghill T, et al. Antimicrobial activity of selected essential oils against selected pathogenic bacteria: In vitro study. Antibiotics. 2021;10(5):546.
Crossref - Stamova S, Ermenlieva N, Tsankova G, Nikolova SP, Georgieva E. Synergistic Antimicrobial Activity of Essential Oils and Vitamin C: Mechanisms, Molecular Targets and Therapeutic Potential. Appl Sci. 2025;15(8):4294.
Crossref - Iseppi R, Mariani M, Condo C, Sabia C, Messi P. Essential oils: A natural weapon against antibiotic-resistant bacteria responsible for nosocomial infections. Antibiotics. 2021;10(4):417.
Crossref - Korinek M, Handoussa H, Tsai YH, et al. Anti-Inflammatory and Antimicrobial Volatile Oils: Fennel and Cumin Inhibit Neutrophilic Inflammation via Regulating Calcium and MAPKs. Front Pharmacol. 2021;12:1-20.
Crossref - Spisni E, Petrocelli G, Imbesi V, et al. Antioxidant, anti-inflammatory, and microbial-modulating activities of essential oils: Implications in colonic pathophysiology. Int J Mol Sci. 2020;21(11):1-27.
Crossref - Yang J, Lee SY, Jang SK, Kim KJ, Park MJ. Anti-Inflammatory Effects of Essential Oils from the Peels of Citrus Cultivars. Pharmaceutics. 2023;15(6):1595.
Crossref - Eid AM, Jaradat N, Shraim N, et al. Assessment of anticancer, antimicrobial, antidiabetic, anti-obesity and antioxidant activity of Ocimum Basilicum seeds essential oil from Palestine. BMC Complement Med Ther. 2023;23(1):221.
Crossref - Wintola OA, Olajuyigbe AA, Afolayan AJ, Coopoosamy RM, Olajuyigbe OO. Chemical composition, antioxidant activities and antibacterial activities of essential oil from Erythrina caffra Thunb. growing in South Africa. Heliyon. 2021;7(6):e07244.
Crossref - Elbestawy MKM, El-Sherbiny GM, Moghannem SA. Antibacterial, Antibiofilm and Anti-Inflammatory Activities of Eugenol Clove Essential Oil against Resistant Helicobacter pylori. Molecules. 2023;28(6):2448.
Crossref - Sahlabgi A, Lupuliasa D, Stanciu G, et al. The Development and Comparative Evaluation of Rosemary Hydroalcoholic Macerate-Based Dermatocosmetic Preparations: A Study on Antioxidant, Antimicrobial, and Anti-Inflammatory Properties. Gels. 2025;11(3):149.
Crossref - Negrean OR, Farcas AC, Nemes SA, Cic DE, Socaci SA. Recent advances and insights into the bioactive properties and applications of Rosa canina L. and its by-products. Heliyon. 2024;10(9):e30816.
Crossref - Mukarram M, Choudhary S, Khan MA, et al. Lemongrass essential oil components with antimicrobial and anticancer activities. Antioxidants. 2022;11(1):20.
Crossref - Puskarova A, Buckova M, Krakova L, Pangallo D, Kozics K. The antibacterial and antifungal activity of six essential oils and their cyto/genotoxicity to human HEL 12469 cells. Sci Rep. 2017;7(1):8211.
Crossref - Alonazi MA, Jemel I, Moubayed N, Alwhibi M, El-Sayed NNE, Ben Bacha A. Evaluation of the in vitro anti-inflammatory and cytotoxic potential of ethanolic and aqueous extracts of Origanum syriacum and Salvia lanigera leaves. Environ Sci Pollut Res. 2021;28(16):19890-19900.
Crossref - Mirghani MES, Liyana Y, Parveen J. Bioactivity analysis of lemongrass (Cymbopogan citratus) essential oil. Int Food Res J. 2012;19(2):569-575.
- Raveau R, Fontaine J, Verdin A, et al. Chemical composition, antioxidant and anti-inflammatory activities of clary sage and coriander essential oils produced on polluted and amended soils-phytomanagement approach. Molecules. 2021;26(17):5321.
Crossref - Jarrar N, Abu-Hijleh A, Adwan K. Antibacterial activity of Rosmarinus officinalis L. alone and in combination with cefuroxime against methicillin-resistant Staphylococcus aureus. Asian Pac J Trop Med. 2010;3(2):121-123.
Crossref - Klancnik A, Guzej B, Kolar MH, Abramovic H, Mozina SS. In vitro antimicrobial and antioxidant activity of commercial rosemary extract formulations. J Food Prot. 2009;72(8):1744-1752.
Crossref - Julianus Sohilait H, Kainama H. Free Radical Scavenging Activity of Essential Oil of Eugenia caryophylata from Amboina Island and Derivatives of Eugenol. Open Chem. 2019;17(1):422-428.
Crossref - Saini A, Pandey A, Sharma S, et al. Assessment of antioxidant activity of rosemary (Rosmarinus officinalis) leaves extract. J Pharmacogn Phytochem. 2020;9(3):14-17
- Marin I, Sayas-Barbera E, Viuda-Martos M, Navarro C, Sendra E. Chemical composition, antioxidant and antimicrobial activity of essential oils from organic fennel, parsley, and lavender from spain. Foods. 2016;5(1):18.
Crossref - Mohamed AA, Alotaibi BM. Essential oils of some medicinal plants and their biological activities: a mini review. J Umm Al-Qura Univ Appl Sci. 2023;9(1):40-49.
Crossref - Mateen S, Rehman MT, Shahzad S, et al. Anti-oxidant and anti-inflammatory effects of cinnamaldehyde and eugenol on mononuclear cells of rheumatoid arthritis patients. Eur J Pharmacol. 2019;852:14-24.
Crossref - Kabotso DEK, Neglo D, Gaba SE, et al. In Vitro Evaluation of Rosemary Essential Oil: GC-MS Profiling, Antibacterial Synergy, and Biofilm Inhibition. Pharmaceuticals. 2024;17(12):1653.
Crossref - Bakhtiar Z, Eghlima G, Hatami M, Mirjalili MH. Quantification of fatty acids in seed oil and important bioactive compounds in Iranian Rosa canina L. ecotypes for potential cosmetic and medicinal uses. Sci Rep. 2023;13(1):1-15.
Crossref - Galgano M, Capozza P, Pellegrini F, et al. Antimicrobial Activity of Essential Oils Evaluated In Vitro against Escherichia coli and Staphylococcus aureus. Antibiotics. 2022;11(7):979.
Crossref - Wasim Akram SA, Arokiarajan MS, Christopher JJ, et al. Antimicrobial and antioxidant study of combined essential oils of Anethum Sowa Kurz. and Trachyspermum ammi (L.) along with quality determination, comparative histo-anatomical features, GC-MS and HPTLC chemometrics. Sci Rep. 2024;14(1):27010.
Crossref - Damasceno ROS, Pinheiro JLS, Rodrigues LHM, et al. Anti-Inflammatory and Antioxidant Activities of Eugenol: An Update. Pharmaceuticals. 2024;17(11):1505.
Crossref
© The Author(s) 2026. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.