ISSN: 0973-7510
E-ISSN: 2581-690X
Fasciola hepatica and Fasciola gigantica are two trematodes (liver flukes) that cause water- and food-borne disease Fasciolosis. Many animals are involved in the life cycle of the Fasciola spp., such as sheep, cattle, and camels, and humans are the definitive host. This work investigated the prevalence of Fasciola among imported and local slaughtered sheep and the relation between infection rate and seasons of the year. A total of 720,290 local and imported sheep (251,701 and 468,489, respectively) were slaughtered in a Jeddah slaughterhouse (Western region of Saudi Arabia) over the periods of 2017-2018, 2018-2019, and 2019-2020. In the three years of inspection, the infection rate at slaughter was higher among imported than local sheep. In 2017-2018, both local and imported sheep had the highest level of infection in the spring and the lowest in the winter. However, in 2018-2019, the highest rate of infection for both was documented in the summer and the lowest in the winter. Lastly, in 2019-2020, the highest rate of infection for local sheep was observed in the summer of 2020 and the lowest rate of infection was observed in the spring of 2020. Among imported sheep, the highest rate of infection was in the summer and the lowest was in winter for both 2019 and 2020. To control this disease, there is the need to apply for successful integrated programs and practice adaptive management techniques.
Fasciola hepatica, Fasciola gigantica, Imported, Local, Sheep, Fasciolosis
Parasitic infections are considered serious health issues and are prevalent throughout most developing countries.1 Annual losses due to infection by the trematodes Fasciola hepatica and Fasciola gigantica (liver flukes) in farm animals are estimated to US$3 billion or more.2 Economically, more than one factor contributes to the losses, including medication costs, meat condemnation, lower wool quality, lower milk production, and more.3 Liver flukes are considered one of the most common ruminant parasites worldwide, and they are spread throughout many regions.4 Fasciola hepatica and F. gigantica are the etiological agents for the zoonotic disease fascioliasis.5 This disease is widespread among farm animals favoring sheep and cattle as hosts and humans can be indirectly infected as well.6
Saudi Arabia attracts thousands of foreign workers annually from all over the globe, including the countries of India, Pakistan, Indonesia, Egypt, Sri Lanka, and the Philippines. Intestinal parasites are endemic in those countries; however, all workers are medically examined in their countries as well as after their arrival in Saudi Arabia. Still, studies have revealed that intestinal parasites and protozoa among those populations prevail.7 Further, studies and literature regarding fasciolosis are still scarce.
Fasciola hepatica is widely distributed but it is particularly abundant in temperate regions and cool high-altitude areas in the tropics and subtropics, while F. gigantica is found mostly in the tropical zones. The geographical distribution of F. gigantica and F. hepatica is dependent on the existence of snails in the environment because snails play a major role as intermediate hosts for the parasitic species.6 A previous survey revealed that 700 million domestic animals around the world were infected with liver flukes.8
The route of infection is oral, which suggests that the infection is transferred from animals to human. In fact, cattle, sheep, and goats can harbor the infection after ingestion of the metacercarial cysts from soil or drinking water. Afterwards, the cysts travel through the intestinal wall and hepatic tissues until they reach the bile duct.9 Fascioliasis is characterized by chronic, subacute, or acute inflammation of the infected liver and the bile ducts. As sequela of improper liver functions and liver cirrhosis, submandibular edema appears, followed by anemia leading to general intoxication and mortality in severe prolonged cases.10 Published study shows a current incidence of about 17 million human cases of fascioliasis, with about 91 million being at risk of infection.11 The Middle East, Asia, many parts of the United States, South Africa, and parts of Europe are considered geographic areas of concerns for the spread of Fasciola species.12-13
Metacercariae are the infective stage of Fasciola species, and humans can harbor the infection through eating contaminated watercress or drinking contaminated water.14 In general, both F. gigantica and F. hepatica have an identical life cycle,15 which depends upon two separate hosts. The first is an intermediate host, and the second is the final host. The final host can be any of several herbivorous mammals in addition to humans. Members of the Lymnaeidae family (commonly known as pond snails) are the corresponding intermediate hosts for Fasciola species.16 Human fascioliasis has been recorded in many countries, with Bolivia, Peru, Cuba, China, Spain, the Nile Delta in Egypt, and central areas of Vietnam and Northern Iran being considered as endemic regions.17 Bovine fascioliasis is the focal source of human fascioliasis. The closer the source of fresh water is, the more the spread of fascioliasis. According to this theory, the Middle East and North Africa regions, particularly Egypt, Ethiopia, Iran, Iraq, and Syria, are hyperendemic areas for human fascioliasis.16
The prevalence of fascioliasis does not differ by the sex of the infected host, regardless of the rate of infection in the area. However, some studies have observed a slight increase in male hosts compared to the female,18 which could be due to girls and women spending less time with livestock, particularly during pregnancy and lactation periods.
Molecular biology techniques have become widely used in identifying microorganisms.3 Regardless of the advances in modern techniques, persistent challenges include identifying reinfection after treatment and differentiating between acute and chronic stages in the detection of resistant strains.19 Furthermore, vaccination against fascioliasis is not available, and anthelminthic drugs are the only treatment options for fascioliasis.20 The objective of this study was to evaluate infection of sheep with Fasciola spp. in Saudi Arabia.
The taxonomic position of Fasciola may be summarized as follows according to Sy et al.:21
Kingdom: Animalia
Phylum: platyhelminths
Class: Trematoda
Subclass: Digenea
Family: Fasciolidae
Genus: Fasciola
Species: Fasciola gigantica
Fasciola hepatica
Animals
A total of 720,290 sheep, including local (251,701) and imported (468,589) sheep, were slaughtered in a Jeddah slaughterhouse (Western region of Saudi Arabia) over the period of 2017-2018, 2018-2019, and 2019-2020. The prevalence of fascioliasis in slaughtered sheep was determined by tri-weekly visits to the main slaughterhouse in Jeddah from July 2017 to July 2018 (36,440 local and 72,813 imported sheep), from August 2018 to August 2019 (143,535 local and 232,409 imported sheep), and from September 2019 to December 2020 (71,726 local and 163,367 imported sheep), the last data collection was taken in more than a year due to disrupted by the COVID-19 pandemic.
Examination of Slaughtered Sheep
After a slaughtered sheep was skinned, the outer surface of the carcass was examined by visual observation for any abnormal lesion and then the abdominal cavity was opened to check the internal organs. For each sheep, the liver was inspected by making multiple deep incisions in the lobes and the gallbladder was opened using a knife. Observations were confirmed by the veterinarians throughout the meat inspection visits.
Statistical Analysis
All the data collected were analyzed to determine the prevalence of fascioliasis in imported and local slaughtered sheep for 3 years and its relationship to seasonal infections. The differences in infection rate between local and imported sheep in different years and seasons were determined using analysis of variance (Correlations). The statistical tests were performed using a software package (IBM® SPSS® Statistics, SA, version 22). A p value < 0.05 was considered as significant.
Infection Rate
In the first year (2017-2018), the infection rate was higher among imported slaughtered sheep (2.12%) than in local slaughtered sheep (0.66%), as shown in Table 1. The same trend was found in the following years. In 2018-2019, the imported slaughtered sheep had an infection rate of 0.62% and the local slaughtered sheep had a rate of 0.12%. Similarly, the imported sheep had a high rate of infection (1.19%) in the year 2019-2020 compare to the local slaughtered sheep (0.27%). Table 1 shows a significant difference in the rate of infection between local and imported sheep (p < 0.01).
Table (1):
A comparison between the Fasciolosis infection rates of local and imported sheep over the course of the 3 years of inspection.
Year |
Slaughtered local sheep |
Infected local sheep |
Rate of infection (%) |
Slaughtered imported sheep |
Infected imported sheep |
Rate of infection (%) |
|---|---|---|---|---|---|---|
2017-2018 |
36,440** |
241** |
0.66 |
72,813** |
1542** |
2.12 |
2018-2019 |
143,535** |
179** |
0.12 |
232,409** |
1448** |
0.62 |
2019-2020 |
71,726** |
192** |
0.27 |
163,367** |
1951** |
1.19 |
**. Correlation is significant at the 0.01 level (2-tailed).
Relation Between Infection Rate and Season
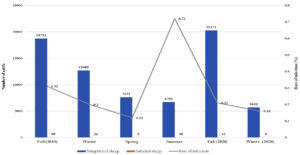
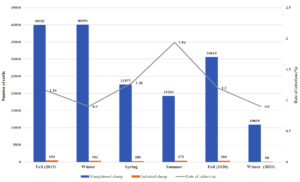
During the 3-year span of the study, a correlation was found between the season in which the local and imported sheep were slaughtered and the prevalence of fascioliasis, as shown in Fig. 1a, b for 2017-2018, Fig. 2a, b for 2018-2019, and Fig. 3a, b for 2019-2020.
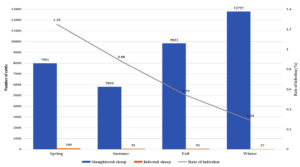
Fig. 1a. Seasonal recordings of the infection rate in the local cattle for the year July 2017 – July 2018.
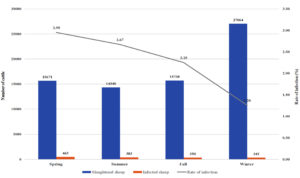
Fig. 1b. Seasonal recordings of the infection rate in the imported cattle for the year July 2017 – July 2018.
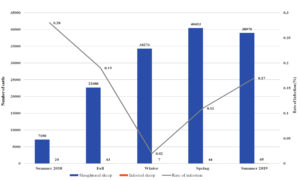
Fig. 2a. Seasonal recordings of the infection rate in the local cattle for the year August 2018 – August 2019.
Fig. 2b. Seasonal recordings of the infection rate in the imported cattle for the year August 2018 – August 2019.
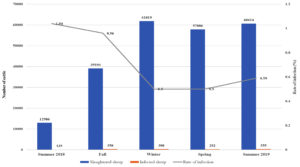
Fig. 3a. Seasonal recordings of the infection rate in the local cattle for the year September 2019 – December 2020.
Fig. 3b. Seasonal recordings of the infection rate in the imported cattle for the year September 2019 – December 2020.
Relation Between Infection Rate and Season for July 2017 to July 2018
For the year 2017-2018, the highest rate of infection in local sheep was observed in the spring (1.25%), and the lowest rate of infection was observed in the winter (0.29%). Similarly, the imported sheep had the highest rate of infection in the spring (2.95%) and the lowest in the winter (1.26%). The imported sheep had a higher rate of infection than the local breed in all seasons, and the difference in the infection rates of local and imported sheep was with high significant values. In 2017, fall and summer had significant differences in infection rates between the groups.
Relation Between Infection Rate and Season for August 2018 to August 2019
For the local sheep in 2018-2019, the highest rate of infection was observed in the summer of 2018 (0.28%), and the lowest rate of infection was observed in the winter (0.02%). A similar trend was observed in the imported sheep, which had the highest rate of infection in the summer (1.04%) and the lowest in the winter and spring (0.50 and 0.50%, respectively). In comparisons between local and imported sheep, the imported animals always had the highest rate of infection in all seasons except summer. For both types of sheep, there was an obvious elevation in infection rates as the weather got warmer in the spring and summer. A comparison of all years showed a significant difference in the infection rates of local and imported sheep for the year 2018-2019.
Relation Between Infection Rate and Season for September 2019 to December 2020
For the local sheep in 2019-2020, the highest rate of infection was observed in the summer of 2020 (0.72%), and the lowest was observed in the spring of 2020 (0.12%). The imported sheep showed a similar trend, with the highest rate of infection occurring in the summer (1.94%) and the lowest in the winters of 2019 and 2020 (0.90% and 0.90%, respectively). However, in comparisons of all seasons between local and imported sheep, the highest rate of infection occurred in imported sheep in all seasons. For both types of sheep, there was an obvious elevation in infection rates as the weather got warmer in the spring and summer.
Fasciola is a ubiquitous parasite worldwide, with animal infection reaching up to 90% prevalence in some areas.22 Magzoub and Kasim23 were the first to publish the prevalence of animal fascioliasis in Saudi Arabia. They found that imported slaughtered cattle have higher infected in the Eastern, Northern, and Western regions. Further, imported sheep had similar results in previous studies in different cities include Riyadh and Taif, but the infection rate in local sheep was much lower than in imported sheep.24- 25 These results are agreed with our results.
A correlation between infection rate and season has been observed in previous studies in various countries.26-28
A previous study evaluated the prevalence of fascioliasis in a Jeddah slaughterhouse.29 The examined animals included 2470 imported sheep, 142 imported cattle, 5 imported camels, and 4050 local sheep. Infection with fascioliasis in imported animals was 30.14%, 4.22%, and 0% for sheep, cattle, and camels, respectively, and local sheep were free from fascioliasis. Degheidy et al.25 conducted a study on the prevalence of fascioliasis as well as liver abscesses in infected animals at an Al-Taif slaughterhouse. Their results showed that of the 18,925 examined sheep, 3501 (18.5%) were condemned because of fascioliasis and liver abscesses. In Riyadh, a comparative study between imported and local slaughtered sheep revealed that the incidence in imported sheep was higher than that in the local sheep.24
Due to the religious position of Saudi Arabia among Muslims, especially at the time of a pilgrimage, the number of slaughtered animals increases dramatically (up to the millions). The ability on increasing in demand of livestock is highly affected by the prevalence of fascioliasis in the country, which highlights the strong need for control of imported livestock. In a study on slaughtered cattle and goats in Taiz, Yemen, by Hezam et al.,30 prevalence of fascioliasis was 5.47% for cattle and 3.14% for goats. The study also showed that adult and male goats were more susceptible to the disease than younger and female ones. On the contrary, female cattle were more likely to be infected than male. Babiker et al.31 conducted a retrospective study in Sudan, using records on fascioliasis collected between 1998 and 2007 from four slaughterhouses. Their study revealed the prevalence of fascioliasis to be 6.05% for cattle and 2.37% for sheep.
In Egypt, Hussein and Khalifa32 collected 297 fecal samples to study the prevalence of fascioliasis in cattle, buffalo, and sheep and found a positivity rate of 30.3%. In addition, the study revealed higher incidences among buffalo compared with cattle, with the lowest infection rate occurring among sheep.
In 2011, a survey study on animal fascioliasis in the Hawaler region, Kurdistan, Iraq, showed infection rates of 4.11%, 3.63%, and 3.44% in sheep, goats, and cattle, respectively. The study also revealed great variations in infection rates between different seasons, with rates being higher in the summer.33 Furthermore, Hussain and Zghair34 conducted an epidemiological study on animal fascioliasis in Karbala, Iraq, and reported 3.61% and 5.77% infection rates for cattle and sheep, respectively. Rinaldi et al.,36 studied the prevalence of fascioliasis in Europe, particularly in three countries (Ireland, Italy, and Switzerland), and found infection rates of 61.6%, 7.9%, and 4%, respectively. All these results are agreed with our results. While a study in Nigeria evaluated animal fascioliasis by examining 400 cattle and 350 goats and revealed 197 (44.8%) cattle and 126 (36%) goats were positive for infection.9 In Bangladesh, 762 stool samples were collected from cattle and examined for fascioliasis, with 504 (66.14%) showing a positive result.35 The rate of the infection in these results is greater than our results.
The current study has several limitations. Notably, samples were derived from limited geographical areas, samples were preserved in different media, and Fasciola eggs were not included in the study. We recommend that future studies consider Fasciola eggs and different larval stages to obtain accurate species identification.
The importance of fascioliasis and its effect on human health both in Saudi Arabia and worldwide motivated this investigation of the prevalence of infection in sheep slaughtered for human consumption. It is necessary to implement more practical methods and preventative techniques to control infection with Fasciola spp. and limit the contamination of animals in pastures to reduce economic losses. Despite the accumulated knowledge on fascioliasis in recent years and technological advances, critical limitations remain. More research is required, especially with regard to histological changes of the parasite, treatment options, and increased resistance of the parasite in both animals and humans.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
ETHICS STATEMENT
Not applicable.
AVAILABILITY OF DATA
None.
- Zafar A, Khan MK, Sindhu D, et al. Seroprevalence of Fasciola hepatica in Small Ruminants of District Chakwal, Punjab, Pakistan. Pak Vet J. 2019;39(1):96-101.
Crossref - Mehmood K, Zhang H, Sabir AJ, et al. A review on epidemiology, global prevalence and economical losses of fasciolosis in ruminants. Microb Pathog. 2017;109:253-262.
Crossref - Calvani NED, Ichikawa-Seki M, Bush RD, Khounsy S, Slapeta J. Which species is in the faeces at a time of global livestock movements: single nucleotide polymorphism genotyping assays for the differentiation of Fasciola spp. Int J Parasitol. 2020;50(2):91-101.
Crossref - Howell AK, Malalana F, Beesley NJ, et al. Fasciola hepatica in UK horses. Equine Vet J. 2020;52(2):194-199.
Crossref - Tezer H, Yuksek SK, Parlakay AO, Gulhan B, Tavil B, Tunc B. Evaluation of cases with Fasciola hepatica infection: experience in 6 children. Asian Pac J Trop Dis. 2013;3(3):211-216.
Crossref - Ahmed E, Markvichitr K, Tumwasorn S, Koonawootrittriron S, Choothesa A, Jittapalapong S. Prevalence of Fasciola spp infections of sheep in the Middle awash River Basin, Ethiopia. Southeast Asian J Trop Med Public Health. 2007;38(1):51-57.
- Hassen Amer O, Ashankyty IM, Haouas NA. Prevalence of intestinal parasite infections among patients in local public hospitals of Hail, Northwestern Saudi Arabia. Asian Pac J Trop Med. 2016;9(1):44-48.
Crossref - Zhang JL, Si HF, Zhou XZ, Shang XF, Li B, Zhang JY. High prevalence of fasciolosis and evaluation of the efficacy of anthelmintics against Fasciola hepatica in buffaloes in Guangxi, China. Int J Parasitol Parasites Wildl. 2019;8:82-87.
Crossref - Abraham J, Jude I. Fascioliasis in cattle and goat slaughtered at Calabar abattoirs. J Biol Agric Healthc. 2014;4(18):34-40.
- Magaji A, Ibrahim K, Salihu M, Saulawa M, Mohammed A, Musawa A. Prevalence of fascioliasis in cattle slaughtered in Sokoto metropolitan abattoir, Sokoto, Nigeria, 2014. J Adv Epidemiol. 2014;2014:247258.
Crossref - Gonzalez LC, Esteban JG, Bargues MD, et al. Hyperendemic human fascioliasis in Andean valleys: an altitudinal transect analysis in children of Cajamarca province, Peru. Acta Trop. 2011;120(1-2):119-129.
Crossref - Ghanimatdan M, Chalechale A, Rezaei F, Rokni MB, Shahrokhi SR. Bioclimatic Analysis and Spatial Distribution of Livestock Fascioliasis in Iran. Iran J Parasitol. 2019;14(1):41-51.
Crossref - Seid U, Melese M. Review on Prevalence, Distrbution and Economic Significance of Liver Fluke in Ethiopia. ARC J Anim Vet Sci. 2018;4(2):38-48.
Crossref - Mas-Coma S, Bargues MD, Valero MA. Human fascioliasis infection sources, their diversity, incidence factors, analytical methods and prevention measures [published correction appears in Parasitology. 2020 Apr;147(5):601]. Parasitology. 2018;145(13):1665-1699.
Crossref - Chen JX, Chen MX, Ai L, et al. An Outbreak of Human Fascioliasis gigantica in Southwest China. PLoS One. 2013;8(8):e71520.
Crossref - Mas-Coma S, Bargues MD, Valero MA. Fascioliasis and other plant-borne trematode zoonoses. Int J Parasitol. 2005;35(11-12):1255-1278.
Crossref - Mas-Coma S, Valero MA, Bargues MD. Fascioliasis. Adv Exp Med Biol. 2014;766:77-114.
Crossref - Tikuye S. Study on Prevalence of Ruminant Fasciolosis and its Associated Risk Factors in Kombolcha, North East Ethiopia. J Vet Sci Technol. 2017;8(4):1000461.
Crossref - Nyindo M, Lukambagire AH. Fascioliasis: An Ongoing Zoonotic Trematode Infection. Biomed Res Int. 2015;2015:786195.
Crossref - Keiser J, Morson G. Fasciola hepatica: tegumental alterations in adult flukes following in vitro and in vivo administration of artesunate and artemether. Exp Parasitol. 2008;118(2):228-237.
Crossref - Sy I, Margardt L, Ngbede EO, et al. Identification of adult Fasciola spp. using matrix-assisted laser/desorption ionization time-of-flight (MALDI-TOF) mass spectrometry. Microorganisms. 2021;9(1):82.
Crossref - Farag F. Human fascioliasis in some countries of the Eastern Mediterranean Region. East Mediterr Health J. 1998;4(1):156-160.
Crossref - Magzoub M, Kasim AA. The prevalence of fascioliasis in Saudi Arabia. Trop Anim Health Prod. 1978;10(4):205-206.
Crossref - Al-Megrin AW. Parasitological and Immunological studies for Diagnosis of Fascioliasis among slaughtered sheep in Riyadh Region, Ph.D. Thesis, Girls’ College of Education in Riyadh. 2003.
- Degheidy NS, Sharaf EM, Al-Malki JS. The incidence of Fasciolasis and some types of bacteria incurred causing lack meat and livers quality of slaughtered animals at Al-Taif, KSA. J Assiut Vet Med. 2013;59(139):136-141.
Crossref - Elshraway NT, Mahmoud WG. Prevalence of fascioliasis (liver flukes) infection in cattle and buffaloes slaughtered at the municipal abattoir of El-Kharga, Egypt. Vet World. 2017;10(8):914-917.
Crossref - Ezatpour B, Hasanvand A, Azami M, Anbari K, Ahmadpour F. Prevalence of liver fluke infections in slaughtered animals in Lorestan, Iran. J Parasit Dis. 2015;39(4):725-729.
Crossref - Takang EE, LeBreton M, Ayuk CE, MacLeod ET. A socio-economic study of Fasciola infections in cattle and sheep at the Etoudi slaughterhouse, Yaoundé, Cameroon. J Helminthol. 2019;94:e92.
Crossref - Ghandour A, Tahir M, Shalaby I. A comparative study of the prevalence of some parasites in animals slaughtered in Jeddah abattoir. JKA. 1989;1:87-94.
Crossref - Hezam K, Morshed AF, Hassan AN, et al. Prevalence of parasitic helminthes among slaughtered animals in slaughterhouses in Taiz, Yemen. Int J Curr Microbiol Appl Sci. 2016;5(8):80-88.
Crossref - Babiker AE, Elmansory Y, Elsadig AA, Majid A. Prevalence of fasciolosis in cattle and sheep slaughtered for human consumption in the White Nile state, Sudan: a retrospective. J Vet Med Anim Prod. 2013;2(2):105-119.
- Hussein ANA, Khalifa RM. Fascioliasis prevalences among animals and human in Upper Egypt. J King Saud Univ Sci. 2010;22(1):15-19.
Crossref - Koyee Q M, Mustafa SO, Ahmed HA. Prevalence of Some Parasitic Helminthes Among Slaughtered Ruminants (Sheep, Goats and Cattles) in Hawler Slaughter House During 2010. Paper presented at the Hawler, Kurdistan Region, Iraq. 4th International Scientific Conference Salahaddin University, Erbil-Iraq. 2011.
- Hussain KL, Zghair ZR. Prevalence of fasciolosis in ruminant in Karbala city. J Entomol Zool. 2017;5(5):364-369.
- Karim MR, Mahmud MS, Giasuddin M. Epidemiological study of bovine fasciolosis: prevalence and risk factor assessment at Shahjadpur Upazila of Bangladesh. J Immunol Infect. 2015;3(3):25-29.
Crossref - Rinaldi L, Biggeri A, Musella V, et al. Sheep and Fasciola hepatica in Europe: the GLOWORM experience. Geospat Health. 2015;9(2):309-317.
Crossref
© The Author(s) 2022. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.