Bacterial exopolysaccharides (EPS) are high-molecular-weight polymers secreted into the extracellular environment, exhibiting diverse structures and functions. Association of EPS with pathogenesis has been extensively investigated, but now their role as multifunctional platforms with transformative potential across health, industry, and environmental sustainability are emerging. This review summarizes recent advances in EPS research, emphasizing their biosynthesis pathways, structural diversity, and functional properties. Special focus is given to bacterial sources, optimization strategies for large-scale production, and emerging applications in biomedicine, environmental remediation, and food industry. Advances in genetic engineering and fermentation technology have enhanced EPS yield and tailored functionalities, expanding their industrial relevance. Challenges such as production cost, downstream processing, and structural characterization are critically discussed. Future perspectives highlight the integration of omics tools, synthetic biology, and sustainable bioprocessing to unlock the full potential of bacterial EPS as versatile, eco-friendly biomaterials.
Bacterial Exopolysaccharides, Biosynthesis, Structural Diversity, Fermentation Technology, Genetic Engineering, Biotechnological Applications, Sustainable Bioprocessing
Microorganisms such as bacteria, actinomycetes, yeasts, moulds, and viruses are prolific producers of exopolysaccharides (EPS) among which bacteria are the most abundant in nature and account for 90%-95% of microorganisms. Their capacity for extreme adaptation has made them the most successful organisms on Earth despite several harsh environmental conditions. During their period of growth, they produce several biologically active compounds. Exopolysaccharide, or EPS, is one such bioactive compound. EPS can be defined as polymers with high molecular weights released into the environment by microorganisms. It was Sutherland who first described exopolysaccharides as high molecular weight carbohydrate polymers that are produced by marine bacteria. The exopolysaccharides are produced in bacteria either intracellularly or extracellularly. EPS are composed of sugar residues, which can be seen as a capsule or slime layer without any association with the host cell. EPS is also discovered to be extremely important in immune-stimulatory functions anticancer functions and blood cholesterol-lowering effects.1-8 EPS produced by bacteria consists of several functional groups including hydroxyl, carbonyl, acetate etc. This enables modification in EPS and new properties can be imparted to EPS.9 Bacterial EPS has a variety of biological roles. They protect cells against extreme temperature, salinity, aridity, UV-rays, unfavourable pH levels, phagocytosis, osmotic stress, and chemical agents (oxidants, heavy metals, and antibiotics).10-14 Polysaccharides are secreted by the majority of marine bacteria and aid in their survival.10 The cryoprotective effect was found in Arctic bacteria.11 The bioethanol-producing bacteria Zymomonas mobilis can flourish in conditions with up to 16% alcohol, and their released EPS also exhibits this biological activity. EPS is the primary component of the biofilm matrix in both Gram-positive and Gram-negative bacteria, and it is crucial for bacterial adhesion, aggregation, and biofilm formation.15-17 Extracellular polymeric substances constitute the biofilm matrix and aid in promote horizontal gene transfer, prevent bacteria from drying out, and provide protection against external agents like antibiotics.
Despite the extensive research on microbial exopolysaccharides, the existing information is disjointed, with biological functions, structural variations, and ecological significance being examined separately. This review characterizes bacterial EPS as versatile biomolecules by aggregating information regarding their role in biofilm development, resilience to environmental stresses, and interactions with host systems. The review places particular focus on aspects of EPS functionality that remain largely uninvestigated, including their potential relevance in health-related fields and their role in surviving extreme climatic conditions. By adopting a comprehensive perspective, this study offers a more thorough and cohesive understanding of bacterial EPS, highlighting their growing importance as flexible bioactive polymers.
Types of exopolysaccharides
EPS has two different extracellular secretion states: capsular polysaccharides which remain firmly attached to the microbial cell wall to form a capsule and polysaccharides in the slime form that are partially or totally released into the environment.18,19 EPS can be structurally classified into:
Homopolysaccharides (HoPS)
Consisting of the same kind of monosaccharide, for example curdlan.
Heteropolysaccharides (HePS)
Made up of several monosaccharides.20 Common sugars such as glucose, fructose, and galactose are found in heteropolysaccharides, along with uronic acids, amino sugars and less common monosaccharides like rhamnose, mannose, fucose, and xylose.21,22
The structural description of EPS usually includes repeating units, linkage positions, glycosidic bond conformation, molecular weight range, monosaccharide content, shape, and spatial structures.23
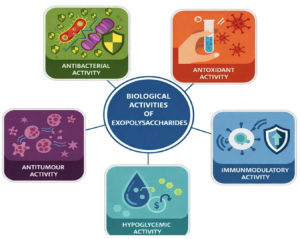
Biological activities of exopolysaccharides
It is often hypothesized that EPS with immunomodulatory function are all triple-helical structures, and indicates there is a greater chance of antioxidant action in EPS with a porous structure. The structure-activity relationships of these EPS are revealed by analyses of their molecular weight, conformation, biological activity, and monosaccharide content. The microstructure and surface morphology of EPS are determined by their content and structure, which in turn affects their biological activity.24 Bacterial exopolysaccharides have the following biological roles, which are impacted by their structure.
A comprehensive summary of the reported biological activities of bacterial exopolysaccharides along with their sources is provided in Table 1, and an overall schematic representation of the structure–activity relationship and associated biological roles is illustrated in Figure 1.
Table (1):
Summary of Biological activities of Exopolysaccharides
No. |
Exopolysaccharide (EPS) producing organisms |
Source of the organism |
Key structural features of EPS |
Biological activity |
Ref. |
|---|---|---|---|---|---|
1 |
Aspergillus spp. DHE6 |
Soil sample |
Polysaccharide backbone with reactive hydroxyl and ether groups |
Antibacterial activity |
[27] |
2 |
Lactobacillus gasseri FR4 Streptococcus thermophilus GST-6 Lactococcus garvieae C47 |
Fermented food |
Heteropolysaccharide EPS with glucose and rhamnose |
Antibacterial activity |
[28,29,30] |
3 |
Pediococcus pentosaceus SSC-12 |
Silage |
Heteropolysaccharide EPS with glucose and rhamnose |
Antibacterial activity |
[31] |
4 |
Lactobacillus spp. |
Fermented food |
Dextran EPS, negatively charged (sulfate/anionic groups) |
Antifungal, anti-biofilm |
[33] |
5 |
Porphyridium cruentum |
Marine red microalga, free-living in marine waters |
Branched EPS with Glucose, galactose, rhamnose as the repeating units |
Antioxidant activity |
[37] |
6 |
Bacillus sp. |
Sichuan pickles (fermented food) |
Branched EPS with Glucose, galactose, rhamnose as the repeating units |
[36] |
|
7 |
S-Enterobacter ludwigi |
Soil |
Branched EPS with Glucose, galactose, rhamnose as the repeating units |
[37] |
|
8 |
Alteromonas australica |
Marine environment |
AC-EPS (produced under acidic pH) AL-EPS (produced under alkaline pH) consisting of different proportions of Mannose, Galactose and D-glucuronic acid |
Antioxidant activity |
[40] |
9 |
Auricularia auricula-judae |
Decaying hardwood trees |
Predominantly glucose with β-1,3-linked straight-chain glucan Linear glucan backbone without extensive branching |
Antitumor activity |
[43] |
10 |
Lactobacillus plantarum 70810 |
Fermented food |
Sulfated exopolysaccharides with non-sulfated EPS variants |
Antitumor activity |
[49] |
11 |
Enterococcus hirae WEHI01 |
Fermented food |
Homopolysaccharide Composed solely of galactose |
Immunomodulatory, macrophage-activating activity |
[52] |
12 |
Lactobacillus paracasei VL8 |
Fermented food |
High-molecular weight sulfated heteropolysaccharide EPS, sulfated sugar backbone enhances bioactivity, composed of glucose and galactose |
Immunomodulatory activity |
[54] |
13 |
Lactobacillus plantarum KX041 |
Fermented food |
Triple helical conformation High molecular weight likely contributes to bioactivity |
Immunomodulatory activity |
[56] |
14 |
Sparassis crispa (Birch mushroom) |
Forest mushroom |
Glycosidic linkages crucial for biological activity |
Hypoglycemic activity |
[57] |
15 |
Cordyceps militaris (medicinal fungus) |
Forest mushroom |
Backbone dominated by galactose 1→4 linkage |
Hypoglycemic activity |
[60] |
Antibacterial activity
A variety of functional groups, including hydroxyl, phosphate, and carbonyl groups, are present in EPS. These functional groups have antibacterial activity via interacting with bacterial cell membranes to a certain degree.25,26 The EPS produced by novel Aspergillus spp. DHE6 mostly contains functional groups such as -OH, -CH, -C=C, and C-O-C. It also possesses high antibacterial activity against dangerous human diseases, including Pseudomonas aeruginosa, Bacillus subtilis, Bacillus aureus, and Bacillus pertussis.27
Research has demonstrated that EPS composed of glucose and rhamnose are more likely to exhibit antibacterial activity when generated from Lactobacillus gasseri FR4, Streptococcus thermophilus GST-6, and Lactococcus garvieae C47.28-30 Similarly, Pediococcus pentosaceus SSC-12 EPS, which is primarily composed of glucose and rhamnose, has a potent antibacterial effect.31 Another study found that Lactobacillus-produced dextran prevents Candida albicans from forming biofilms.32 Through their sulfate groups, the negative charges in EPS can interact with bacteria more effectively, demonstrating antifungal action.33
Antioxidant activity
The primary characteristic of its antioxidant action is thought to be the exopolysaccharides’ ability to donate hydrogen, however the underlying process is unclear.34 A number of variables, including the type of glycosidic link, branching patterns, and monosaccharide composition, contribute to EPS’s antioxidant activity. The antioxidant activity of EPS is significantly determined and influenced by its monosaccharide content and composition ratio. For instance, EPS made up of glucose-repeating units has poor hydroxyl radical scavenging activity and potent scavenging action against superoxide and 2,2-diphenyl-1-picrylhydrazyl- hydrate (DPPH), which is comparable to that of ascorbic acid standard.35,36 The primary monosaccharides in EPS, galactose, glucose, and rhamnose, have antioxidant properties. The EPS investigations on Porphyridium cruentum, Bacillus sp. S-1, and Enterobacter ludwigii demonstrated this conclusion.37,38 All of these imply that EPS’s antioxidant activity may be impacted by the type of glycosidic bond and branching arrangement.39
EPS with a high degree of branching has been shown to have significant antioxidant action as well. From Tetragenococcus halophilus SNTH-8, THPS-1 and THPS-2, two highly branched polysaccharides with exceptional emulsifying and antioxidant qualities, were separated and purified by Li et al.40 The biological activity of EPS generation can also be influenced by the culture conditions, such as pH. For instance, Alteromonas australica QD’s EPS produced at varying pH levels. The findings showed that identical monosaccharides with varying amounts of mannose, galactose, and D-glucuronic acid were present in acidic pH EPS (AC-EPS) and alkaline pH EPS (AL-EPS). It has been discovered that AL-EPS exhibits strong antioxidant activity.41
Antitumor activity
EPS structural investigations indicate that the anticancer activities of EPS depend on β-1,3-glycosidic linkages on glucose chains and β-1,6-glycosidic bonds on branched chains.42 As an illustration, consider β-(1,3)-D-glucan with (1→6) branched chains. Porphyra mushrooms were used to extract a variety of polysaccharides, which contains the anticancer active fraction.43 The anticancer polysaccharide obtained from Auricularia auricula-judae was also made up of β-1,3-bound straight-chain glucan.44 To a certain degree, the anticancer activity of EPS is determined by the flexibility of the polysaccharide backbone.45 Hydrogen bonds and the electrostatic repulsion of substituents within the polysaccharide molecule combine to provide flexibility. High flexibility makes it easier for the polysaccharide and immune system to interact, which boosts EPS‘s anticancer effect.46,47 Additionally, polysaccharide branching has been shown to impair intermolecular binding and reduce intramolecular interactions, which can impact anticancer efficacy.48,49 EPS with branching degrees between 0.2 and 0.33 exhibit stronger anticancer activity.50
Sulfated polysaccharides exhibited more anticancer activity than non-sulfated polysaccharides. The growth of tumor cells was suppressed more quickly by EPS from Lactobacillus plantarum 70810 (such as r-EPS1 and r-EPS2) than by r-EPS1. The sulfate group and β-glycosidic bond composition of r-EPS are thought to be intimately linked to its potent anticancer action.49
Immunomodulatory activity
The immunomodulatory effect of EPS is directly correlated with the presence of galactose, according to results based on structural characteristics and immunomodulatory activity.51 In 1988, it was revealed that rabbits’ antibodies against galactose reacted with polysaccharide antigens.51 With a molecular weight of 2.59 × 103 Da, the EPS generated by the probiotic Enterococcus hirae WEHI01 is solely made up of galactose and efficiently enhances macrophage-mediated immune responses.52
Analysis of the structure-activity relationship revealed a significant correlation between the molecular weight and EPS’s immunomodulatory effect. Higher molecular weight EPS may suppress the immune system. Immune responses are more effectively elicited by acidic heteropolysaccharides.53 For instance, glucose and galactose make up the majority of Lactobacillus paracasei VL8’s high-molecular weight sulfated heteropolysaccharides, which exhibit potent immunomodulatory properties.54 EPS with a triple-helical shape may have immunomodulatory action. For instance, it has been discovered that Aureobasidium pullulans CGMCC 23063, which has a triple-helical conformation linked to chains and round spheres, contains EPS.55 Lactobacillus plantarum KX041 produces a new crude EPS with a triple helical shape that exhibits strong immunological activity, encouraging Raw264.7 phagocytosis and proliferation.56
Hypoglycemic activity
EPS’s molecular weight, branching structure, and high-order structure are all strongly correlated with its hypoglycemic action.57,58 Birch mushroom polysaccharides were employed by to mimic intestinal digestion. The digested polymer (UIOPS-1I) has a decreased molecular mass and significantly greater inhibitory action against glucosidase.57 An essential component of EPS’s hypoglycemic action is glycosidic linkages. The majority of EPS with hypoglycemic action were discovered to include 1→3, 1→4, and 1→6 glycosidic linkages.59 For instance, the galactose 1→4 linkage that dominates the EPS backbone of Cordyceps militaris efficiently suppresses glucosidase activity and restores glucose tolerance in mice.60
Biosynthesis of exopolysaccharides
Biosynthesis of EPS in bacteria takes place by the following mechanisms:
- Wzx/Wzy dependent pathway
- Synthase dependent pathway
- ABC transporter-dependent pathway14,61
- Extracellular biosynthesis by sucrase protein62
Typically, the synthase-based and extracellular synthesis pathway produces homopolysaccharides. The ABC transporter-dependent pathway and the Wzx/Wzy-dependent pathway are used to synthesise heteropolysaccharides. Both Gram-positive and Gram-negative bacteria produce extracellular polymeric substances (EPS).63 The Gram-positive bacteria which produce EPS include Bacillus, Leuconostoc, Lactobacillus, Paenibacillus, Streptococcus spp., Bifidobacterium, Rhodococcus genera.14,64 EPS producing Gram-negative bacteria include: Agrobacterium, Acetobacter, Gluconobacter, Gluconoacetobacter, Kozakia, Rhizobium, Alcaligenes, Pseudomonas, Enterobacter, Vibrio, Erwinia, Xanthomonas, Klebsiella genera. The different pathways for the synthesis of exopolysaccharides are detailed below:
Wzx/Wzy – Dependent pathway
This mechanism comprises three stages65:
Synthesis of nucleotides
In this pathway, sugar molecules are transported into cells actively and converted to several monomeric units. After that, it is moved and connected to an internal membrane-based undecaprenyl phosphate (Und-P) anchor (C55 lipid carrier).66
Assemblage of the repeating units
At this point, repeating units are created by glycosyltransferases (GTs). Wzx flippase then translocates these units across the cytoplasmic membrane.66,67
Polymerization and export
In this final phase, the oligosaccharides undergo a variety of enzymatic modifications, such as methylation and acetylation, before the Wzy protein polymerizes them into polysaccharides. ABC transporters subsequently release the formed polysaccharides to the cell surface.68
ABC transporter-dependent pathway
This mechanism, which is present in Myxococcus xanthus, forms an Und-PP-sugar by transporting the sugar molecules to an Und-P molecule at the inner membrane. Certain GTs found on the inner side of the cytoplasmic membrane synthesize the polysaccharides.69 A tripartite efflux pump then translocates the generated polysaccharide. This route primarily synthesizes capsular polysaccharides.70-72
Synthase- dependent pathway
In this mechanism, the assembly of UDP-glucose is carried out by a bacterial cellulose synthesis (bcs) A located at the inner membrane.72
Extracellular biosynthesis
In this biosynthesis mechanism, carried out by the sucrase protein, sucrose is converted to its monomeric units outside the cell.73 The monosaccharide molecules are subsequently polymerized by the glycosyltransferases to produce fructan (levan) and dextran.18 The synthesised exopolysaccharides are secreted to the external environment.74
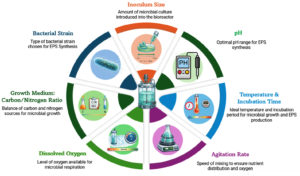
Factors affecting exopolysaccharide production
The industrial applications of bacterial exopolysaccharides are very limited due to their low production. The primary cause of this is the low productivity of bacterial strains that have not yet been used in industry.75 Several factors influence EPS production in bacteria, including nutritional, environmental, and genetic parameters. A detailed summary of these factors is presented in Table 2, and a schematic overview is illustrated in Figure 2.
Table (2):
Summary of Factors Regulating Exopolysaccharide Production
No. |
Factor |
Typical Optimal Range |
Effect on EPS Yield |
Ref. |
|---|---|---|---|---|
1 |
Carbon source |
Sucrose > glucose |
~9-fold increase |
[76] |
2 |
Nitrogen Source |
Tryptone > inorganic nitrogen source |
~7-fold |
[77] |
3 |
pH |
5.5–7 |
~2-fold increase |
[79] |
4 |
Temperature |
18–25 °C |
Stress enhances yields up to 30 times |
[80] |
5 |
Incubation Period |
48–96 h |
Peak before degradation |
[82] |
6 |
Agitation speed |
150 rpm |
Increased EPS yield vs static condition |
[78] |
7 |
Inoculum size |
5%-6% |
Higher yields |
[84] |
8 |
Aeration |
Controlled aeration |
~5-fold increase |
[85] |
9 |
Bacterial strain |
Species specific |
~10-fold difference |
[78] |
Factors affecting EPS production in bacteria include:
Growth medium composition (Carbon and Nitrogen sources)
The carbon source used in the production medium affects the yield of exopolysaccharides (EPS). In most of the EPS yielding organisms, sucrose is found to provide a better yield than glucose. A 9-fold increase in EPS yield was observed from ~4.55-41.25 g/l in Paenibacillus sp. when sucrose was increased from 1.5%-10% under same conditions of pH and temperature.76 The EPS output is also dependent on the source of nitrogen. As demonstrated in the example of Bacillus sp., where the use of tryptone as the nitrogen source increased the EPS production from 143 mg/l to 1071 mg/l.77
pH Level
The influence of pH is highly strain-dependent. Maximum yield of EPS is obtained at a slightly acidic to neutral pH which normally lies in the range of 5-7. A double fold increase in EPS yield was obtained for Lactic acid bacteria, from 491 mg/l to 1029 mg/l of EPS with an optimum pH of ~5.5.79 For Paenibacillus sp., optimal EPS yield was obtained near a pH of 6.5-7.2 and the yield was declined at pH 8.5.78,79
Temperature
Yields can be multiplied by selecting a temperature that causes stress or changes metabolism toward EPS. EPS synthesis frequently reaches its peak at temperatures that are just below those ideal for biomass growth. The highest yield of EPS was obtained for L. rhamnosus, L. plantarum, and L. lactis at temperatures around 18-25 °C.80 EPS production was raised about 30-fold higher when a stressful condition of low temperatures of -2 °C and 10 °C was provided for Colwellia psychrerythraea, an extremophile.81
Incubation period
EPS yield is time-dependent. It typically reaches its maximum accumulation between 48 and 96 hours, after which enzyme breakdown may take place. In certain Lactic strains, optimum EPS was attained after about 72 hours.82
Agitation speed
Proper agitation is required for sufficient oxygen and nutrient transfer which can improve EPS production. An increase in agitation from 120-150 rpm resulted in a significant curdlan production from Paenibacillus while at 180 rpm, yield of EPS decreased due to the development of shear stress.78 A high agitation of 100-150 rpm resulted in a high EPS yield of 8.12 g/L compared to static culture conditions in the case of Agrobacterium tumefaciens.83
Inoculum size
A larger size of inoculum frequently results in a higher EPS yield as more cells start EPS production earlier. In comparison to lesser inoculum sizes, Bacillus subtilis produced the maximum EPS (3010 µg/mL) when the inoculum was increased from 5%-6%.84
Oxygen availability
Many bacteria need oxygen for the precursor synthesis and higher rate of aeration increases EPS production. The addition of whey protein hydrolysate under controlled pH resulted in a significant fivefold increase of EPS in Streptococcus thermophilus. This is due to the improvement in both oxygen and nutrients.85,86
Specific bacterial strain
Yields can be significantly altered by strain selection alone. For instance, under ideal circumstances, Paenibacillus TKU023 produced up to ~41 g/L EPS, while other strains only produced ~3-4 g/L.78
Among these, the carbon source and C/N ratio are the most critical elements influencing EPS yield and characteristics. The producers of levan (up to 100 g/L), dextran (up to 66 g/L), curdlan (up to 48 g/L), and xanthan (up to 40 g/L) have the highest EPS output.75 Consequently, more study is required to establish a highly effective EPS production system. The main ways to reduce costs are via isolating new, highly productive strains, using cheap culture media, and using genetic engineering techniques to create more productive strains. Glucose and sucrose are the primary carbon sources utilized to produce bacterial EPS. Since the cost of culture media makes up about 30% of the overall cost of the fermentation process, a variety of media based on industrial and agricultural waste are now available.75
Genetic engineering strategies to improve the exopolysaccharide production
In addition to the above-mentioned factors recent genetic strategies have significantly advanced the microbial exopolysaccharide production. This is possible by modulating the associated gene clusters and regulatory networks. CRISPR/Cas-based genome editing has gained special attention and emerged as a strong tool to elevate the EPS yield by targeting the overexpression of key enzymes like glycosyltransferase, modifying transcriptional regulators, and the deletion of competing pathways involved in the synthesis of polysaccharides. The key genetic engineering strategies to improve the EPS production are87:
Increasing the precursor Supply
The synthesis of heteropolysaccharides depends on nucleotide sugars such as UDP- glucose, dTDP-glucose for EPS synthesis. The mechanisms for this process involve the following:
Overexpression of important enzymes
Enhancement of the important enzymes like UDP-glucose pyrophosphorylase and phosphoglucomutase (PGM) in Streptococcus thermophilus has resulted in a significant yield of EPS.87
Carbon flux balance
Precursors can be spared and EPS generation can be increased by altering pyruvate metabolism in Lactobacillus species to decrease lactic acid buildup.87
Optimizing Substrate Utilization:
Extending substrate range
It can be more economical and increase yields to engineer bacteria to use inexpensive agro-industrial waste or alternative carbon sources like glycerol or particular sugars (fructose, sucrose).
Improving sugar transport
EPS generation can be directly impacted by increasing the uptake of desirable carbon sources through the overexpression of particular sugar transporter genes or the engineering of their regulators.86
Genetic engineering techniques
The EPS biosynthesis genes and their regulatory networks can be precisely manipulated. Current methods consist of Liu et al.87:
Manipulation of specific genes
- Gene overexpression: When specific genes within the EPS operon like epsA and epsE in S. thermophilus are overexpressed EPS yields have increased.
- Gene deletion/knockout: By removing competing metabolic pathways, more resources can be directed toward the synthesis of EPS by preventing the creation of undesirable byproducts.
Advanced genetic techniques
With the use of CRISPR-Cas9 system technology, EPS properties can be customized and production efficiency can be raised by accurate genome editing. It has been utilized to discover important genes and optimize expression levels for optimal production by methodically controlling entire eps gene clusters.
- Synthetic Biology: More control over the final polymer’s structure and characteristics can be achieved by developing completely new, synthetic EPS pathways in non-native hosts like Escherichia coli.87
- c-di-GMP signaling: The secondary messenger cyclic di-GMP (c-di-GMP) plays a critical role in controlling EPS generation. By modifying the diguanylate cyclases and phosphodiesterases that control intracellular c-di-GMP levels EPS production can be significantly enhanced.88
Applications of exopolysaccharides
Exopolysaccharides produced by microorganisms have found widespread use in various industries including food, environmental, pharmaceutical, and cosmetic sectors (Table 3).
Table (3):
Summary of Exopolysaccharide producing Organism and their Industrial Applications
No. |
Area of application |
Exopolysaccharide (EPS)producing organism |
Structural Features of EPS |
Property of EPS |
Application |
Ref. |
|---|---|---|---|---|---|---|
1 |
Food Industry |
Latilactobacillus curvatus, Leuconostoc mesenteroides, Leuconostoc citreum, Limosilactobacillus fermentum, Lactobacillus kefiranofaciens, Lacticaseibacillus casei, Latilactobacillus sakei, Limosilactobacillus reuteri, Pediococcus pentosaceus, Leuconostoc pseudomesenteroides, Lentilactobacillus parabuchneri |
Dextran (homopolysaccharide) Primarily composed of α-(1→6)-linked glucose, with varying degrees of branching |
High molecular weight Highly hydrophilic Branched glucan structure Strong water-binding capacity |
Confectionery products Bakery products (soft crumb structure) Ice cream (as stabilizer and adjuvant) Fermented dairy products |
[11,63,74,93] |
2 |
Environmental Bioremediation |
Comamonas denitrificans, Brachymonas denitrificans, Acinetobacter calcoaceticus, Acinetobacter lwoffii, Aeromonas hydrophila |
High molecular weight and abundant presence of negatively charged groups |
EPS-mediated biofilm matrix formation High affinity for nutrient adsorption and retention Enhanced nitrogen and phosphorus removal |
Biofilter-based wastewater treatment systems |
[108, 109] |
3 |
Health |
Lactobacillus paracasei NFBC 338 |
β-glucan (β-glycan) exopolysaccharide |
Contributes to structural stability and stress resistance Protection against acidic pH, bile salts, and digestive enzymes |
Oral probiotic formulations |
[113] |
4 |
Health |
Streptococcus thermophilus CRL 1190 |
High molecular weight polysaccharide |
Gastroprotective activity |
Formulations against gastritis |
[114] |
5 |
Health |
Bifidobacterium spp., Lactobacillus spp., Streptococcus spp. |
High molecular weight polysaccharide |
Mucoadhesive and protective properties Anti-ulcer activity |
Probiotic and functional food formulations |
[114] |
6 |
Health |
Streptococcus mutans |
consist mainly of glucans synthesized from dietary sucrose |
Sticky, insoluble glucan matrix Strong surface-adhesive properties |
Dental plaque formation |
[116] |
7 |
Health |
Lactobacillus helveticus KLDS1.8701 |
High molecular weight polysaccharide |
Immunomodulatory EPS |
Probiotic formulations |
[117,118] |
8 |
Cosmetics |
Leuconostoc mesenteroides, Streptococcus mutans |
Dextran Glucose-based α-glucan |
Highly hydrophilic and film-forming Excellent water-binding capacity |
Skin creams and lotions Moisturizers and cosmetic formulations |
[54] |
9 |
Cosmetics |
Xanthomonas spp. |
High molecular weight polysaccharide Stable over a wide range of pH and temperatures |
Strong gelling and thickening ability Gelling, emulsifying, and foaming agent |
Skin creams, lotions, gels, and foams Cosmetic emulsions and cleansers |
[121,122] |
10 |
Cosmetics |
Streptococcus spp. |
Anionic high-molecular weight polysaccharide |
Hydration and lubrication |
Ophthalmic solutions Dermal fillers |
[123,124] |
11 |
Cosmetics |
Vibrio spp. |
Bioactive EPS stimulating HA synthesis |
Skin hydration and anti-aging |
Anti-aging creams Serums Moisturizers |
[126] |
Food industry
Microbial exopolysaccharides are widely used in the food industry.89 The unique properties of exopolysaccharides secreted by Lactic acid bacteria (LAB) have gained special attention in this industry.90 The species coming under Pediococcus, Lactiplantibillus, Lacticaseibacillus, Latilactobacillus, Leuconostoc, Fructilactobacillus, Weisella, and Streptococcus species are capable of synthesising EPS.63,91 However, the species coming under Weisellia are not recognized as safe by the Food and Drug Administration Department, U.S.92 Dextran is a common exopolysaccharide and is produced by several LAB species which include Latilactobacillus curvatus (formerly Lactobacillus curvatus), Leuconostoc mesenteroides, Limosilactobacillus fermentum (formerly Lactobacillus fermentum), Leuconostoc citreum, Lactobacillus kefiranofaciens, Lacticaseibacillus casei (formerly Lactobacillus casei), Latilactobacillus sakei (formerly Lactobacillus sakei), Limosilactobacillus reuteri (formerly Lactobacillus reuteri), Pediococcus pentosaceus and Leuconostoc pseudomesenteroides Lentilactobacillus parabuchneri (formerly Lactobacillus parabuchneri).11,63,74,93 They are widely used in the food sector to increase the viscosity and moisture retention of confections, prevent crystallization, and improve the softness and structure of crumbs. They are also good emulsifiers, stabilisers and an adjuvant in ice cream manufacturing units.
Environmental bioremediation
The rapid growth of industrialisation has polluted the ecosystem in addition to its other long-lasting effects.94 The discharge of untreated wastewater into the land and water bodies destroys the ecosystem’s biodiversity. The discharge of wastewater containing excess nitrogen and phosphorous to water bodies enhances the growth of algae thus decreasing the dissolved oxygen and this in turn reduces the natural fish population.95,96 The decrease in water quality leads to the scarcity of safe drinking water. The possible methods for treating the wastewater are mixing, and disinfection using chlorine, sedimentation and filtration. But they do more harm than their benefits. They are not only expensive but also release toxic secondary pollutants that can harm life due to their persistent nature.97-99 Therefore, we switched to biological treatment methods which included the usage of activated sludge. However, the disadvantage of this method is excess sludge production and lesser aggregate generation.100,101 Later Zheng et al in 2008 discovered that the exopolysaccharides produced by microbes can be used for wastewater treatment which is much more economical, efficient and user-friendly.102
EPS immobilised in agar beads or as biofilms can be used for nutrient removal from wastewater.103 Several bioreactors including airlift suspensions, fluidized beds, anaerobic sludge blankets, packed beds, and trickled beds use biofilm-based coating for filtration.104-106 The filter is composed of exopolysaccharides of a consortium of microorganisms that can filter the wastewater that is being treated.103 The microbial community thus degrade the organic matter.107 Anderson in 2009 discovered that usage of Comamonas denitrificans and Brachymonas denitrificans immobilized on biofilters removed the nitrogen content in the treated water.108 Similar activity was discovered in Acinetobacter calcoaceticus, Acinetobacter iwoffi, and Aeromonas hydrophila for the removal of phosphorous.108 EPS possess negative functional groups such as hydroxyl, carboxyl, sulphate and phosphate which bind metal ions Pb2+, Cd2+, Cu2+, Zn2+ present in contaminated environments. This reduces the toxicity and availability of the metals thus prevents their entry into the food chain.109 Extracellular polymeric substances (EPS) play a vital role in maintaining the structural strength and stability of biofilms, which are clusters of microorganisms surrounded by a matrix that they produce. In these biofilms, microorganisms can work together in a synergistic manner, resulting in an enhanced breakdown of complex pollutants.103 Moreover, EPS improve the sorption of pollutants like heavy metals and organic compounds present in the biofilm matrix. The microbial consortium breaks down these heavy metals as a result of the sequestration, which also reduces their bioavailability and distribution.110,111
Health
Extracellular polysaccharides produced by microbes help them to thrive in adverse conditions including protection from the attack of other organisms. Bacteria that produced enormous amounts of EPS were found to be self-protected from the attack of bacteriophages and nisin.112 Stack et al discovered that β-glycan produced by Lactobacillus paracasei NFBC 338 can survive under severe gastrointestinal stress.113 EPS produced by S. thermophilus CRL 1190 was purified and found to be effective against gastritis.114 Similarly, EPS from Bifidobacteria, Lactobacilli, and Streptococci strains were found to have certain antiulcer effects.114 The cell that had EPS bound to it by the bacteria Lactobacillus plantarum exhibited an enhanced ability to bind to mutagens like heterocyclic amines and thus inactivated it.115 Extracellular polysaccharides produced by Streptococcus mutans have a pivotal role in adhering the bacterial cell to the surface of the tooth. This aids in dental plaque formation, thus facilitating protection via bacterial colonization.116 By raising anti-inflammatory cytokines like IL-10 and reducing pro-inflammatory cytokines like IL-1β and IL-6, exopolysaccharides produced by L. helveticus KLDS1.8701 shown anti-colitic properties. The EPS released by L. helveticus LZ-R-5 and L. kiferi WXD029 shown strong immunomodulatory action by promoting the growth of RAW264.7 macrophages and generating a number of cytokines, including nitric oxide, TNF-α, IL-6, and IL-10. It was also found that these LAB-EPS can boost acid phosphatase activity and encourage macrophage phagocytosis, making it a potentially useful immunomodulatory agent.117,118
Cosmetics
Biocompatibility, hydrophilic and non-toxic nature -these attributes of exopolysaccharides are very well exploited by cosmetic industries, especially when used for skin formulations, which helps in maintaining a hydrated environment.119 Dextran produced from Leuconostoc mesenteroides and Streptococcus mutans is a good smoothening and brightening agent for skin.54 It makes the skin firm, promotes radiance, and reduces pimple formation. Dextran is also well known for its anti-inflammatory property, which improves blood flow and the synthesis of nitric oxide (NO) in human epidermal cells.120 Xanthan produced from Xanthomonas assist in gelling and therefore is used in moisturising and smoothening skin. Xanthan is also used as an emulsifier and foaming agent in the formulation of skin.121,122 Hyaluronan or Hyaluronic acid (HA), an anionic high-molecular-weight polysaccharide produced from Streptococcus sp. was first discovered in the vitreous humour of the eye.123,124 Because of its ability to contain water, it is often utilized in cosmetics.125 Exopolysaccharides produced by Vibrio sp. enhance the endogenous synthesis of Hyaluronic acid in fibroblasts and this is extensively used in the cosmetic industry for hydration and antiaging. The discovery has been awarded patent in the year 2021 (EP2827837B1) by European Patent Office.126
Ecological importance of bacterial exopolysaccharides
The bacterial exopolysaccharides provide several benefits to the surrounding environment which include:
Role in microbial survival
Exopolysaccharides act as a shield and protect the host cell against several harsh and extreme conditions like UV radiation, osmotic stress and desiccation. It also prevents the entry of toxins and antimicrobial compounds.127
Biofilm formation
EPS are the major constituent of biofilms which helps microbes adhere to the surfaces and also enables a cell-cell communication and genetic transfer. This also serves as protection for the microbial consortia against pathogen attack.127
Role in soil and aquatic ecosystem
EPS bind to the soil particles thus enhances the structure and aggregation of soil. This improves the aeration, and nutrient cycling. In aquatic environments EPS helps in marine snow and floc formation that is essential to sedimentation and carbon cycling.127
Nutrient cycling
Under nutrient starving conditions, EPS act as a carbon and energy reserve for microbes which contribute significantly to the biogeochemical cycle by mobilizing nutrients like iron, phosphorous and trace elements.128
Symbiotic and plant-microbe interactions
EPS associated with the plant rhizosphere aid in root colonization and protect the beneficial microbes in adverse conditions. For example, exopolysaccharides produced from Rhizobium help in nitrogen fixation.129
Detoxification and bioremediation
EPS help in sequestration of toxic metals like Lead (Pb), Copper (Cu), Cadmium (Cd), etc. and other organic pollutants and this reduces their bioavailability. This phenomenon in turn aids in the process of bioremediation and contribute to the survival of microbes in the polluted environments.130
Safety concerns in large-scale production of exopolysaccharides
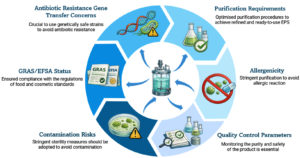
The mass production of microbial exopolysaccharides (EPS) presents considerable industrial and biomedical opportunities. Nevertheless, it also poses crucial safety issues (Figure 3). Numerous microbes that produce EPS are opportunistic pathogens and contain virulence factors that may pose risks during cultivation. The presence of harmful microorganisms or their byproducts in production systems can harm the product safety and impact both human and environmental health. Furthermore, the use of large bioreactors may result in the unintentional release of genetically modified microbial strains into the ecosystem. Another issue is the utilization of growth media that may contain components derived from animals, which could carry pathogens or introduce allergens. Therefore, strict compliance with biosafety regulations and ongoing monitoring are vital to reduce potential risks during the industrial production of EPS.131
From a regulatory point of view, EPS meant for use in food or biomedicine must adhere to globally accepted safety regulations. Strains used in food-related applications should ideally have the Generally Recognized as Safe (GRAS) status of US Food and Drug Administration. A comparable safety approval is provided by the Qualified Presumption of Safety (QPS) framework developed by the European Food Safety Authority. GRAS or QPS designation of a microbial strain guarantees that it has a track record of safe use or is backed by thorough toxicological, genomic and metabolic evaluations.132 Before being used in industry, microorganisms without this kind of regulatory approval must undergo thorough safety assessments. To guarantee constant product quality, purity and traceability industrial EPS production must also follow Good Manufacturing Practices (GMP). Practising GMP guidelines are supportive to get rid of leftover cells, recombinant DNA endotoxins or allergenic contaminants. GMP emphasis on controlled fermentation conditions and verified sterilization techniques. It allows Methodical documentation and stringent downstream purification processes.133 When using genetically modified microorganisms (GMMs) for increased EPS production, biosafety issues are especially important. To stop the unintentional release of engineered strains into the environment, biosafety guidelines follow a strictly monitored biological and physical containment.134 Ensuring environmental safety requires adherence to national and international GMO regulations as well as risk assessment and post-production surveillance. For microbial EPS to be commercialized safely and sustainably, regulatory oversight must be integrated at the stages of strain selection, bioprocess design and product validation.
Strategies to overcome biosafety challenges
To tackle these biosafety issues, various strategies can be employed. One such method is choosing non-pathogenic or less pathogenic host organisms for EPS production, such as Lactobacillus, Bacillus subtilis, or other microorganisms classified as Generally Recognized as Safe (GRAS). These host strains lower the risk of pathogenicity while still achieving high EPS yield. Another method involves genetic modification, where the metabolic pathways in safe microbial hosts are enhanced to efficiently produce the desired EPS, reducing reliance on harmful strains. Additionally, using closed bioreactor systems, implementing strict sterilization procedures, and conducting regular microbial testing help ensure containment and prevent contamination. Developments in synthetic biology enable the creation of microbial strains equipped with safety mechanisms, including autotrophy or kill switches, which can inhibit survival in uncontrolled environments. Together, these strategies facilitate the secure, scalable, and sustainable generation of EPS for use in industrial and biomedical fields.135
Challenges and future perspectives
One of the important challenges faced by exopolysaccharides is their commercialization, since there is insufficient specific knowledge about the structure-function relationship particularly in relation to their biological functions.136 Since different bacteria produce different EPS structures, the biological effects would be different. Therefore, detailed knowledge is essential before its use. Some pathogenic strains that produce EPS are another hurdle in the commercialization of EPS. For example, Pseudomonas aeruginosa which produces Alginate, has been genetically engineered to be a non-pathogenic one for EPS production. Therefore, more studies are required before its production and commercialization.137 Another problem faced is the small quantity of EPS produced during extractions. An effective method should be designed so that an appreciable quantity of EPS can be produced commercially.62 Modification of EPS by addition or deletion of functional groups, or changing single monosaccharides is also of great concern in producing EPS with improved functions.23
Microbial exopolysaccharides are a diverse and exciting class of biopolymers with important applications in industry, biomedicine, and the environment. They are useful for applications in food, medicines, agriculture, and environmental remediation because of their structural variety, biocompatibility, biodegradability, and functional qualities like emulsification, flocculation, antioxidant, and immunomodulatory activities. The potential for producing EPS on a large scale and at a reasonable cost has been further increased by developments in metabolic engineering, molecular characterization, and process optimization. However, to fully realize their economic and therapeutic potential, more thorough understanding of structure–function interactions and sustainable production methods are required. Ongoing research is expected to promote the wider use of exopolysaccharides as sustainable substitutes for synthetic polymers in the near future.
ACKNOWLEDGMENTS
The authors extend their gratitude to School of Biosciences, Mar Athanasios College for Advanced Studies Tiruvalla (MACFAST), Pathanamthitta, Kerala, India, for providing the necessary resources to conduct this review. The authors also thank CSIR-HRDG for the funding.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
This study was funded by the CSIR-HRDG, Council of Scientific & Industrial Research-Human Resource Development Group (08/1324(16515)/2023-EMR-I).
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
This article does not contain any studies on human participants or animals performed by any of the authors.
- Vinderola G, Perdigon G, Duarte J, Farnworth E, Matar C. Effects of the oral administration of the exopolysaccharide produced by Lactobacillus kefiranofaciens on the gut mucosal immunity. Cytokine. 2006;36(5-6):254-260.
Crossref - Hidalgo-Cantabrana C, Lopez P, Gueimonde M, et al. Immune modulation capability of exopolysaccharides synthesised by lactic acid bacteria and bifidobacteria. Probiotics & Antimicro Prot. 2012;4(4):227-237.
Crossref - Matsuzaki C, Kamishima K, Matsumoto K, et al. Immunomodulating activity of exopolysaccharide-producing Leuconostoc mesenteroides strain NTM048 from green peas. J Appl Microbiol. 2014;116(4):980-989.
Crossref - Kitazawa H, Harata T, Uemura J, Saito T, Kaneko T, Itoh T. Phosphate group requirement for mitogenic activation of lymphocytes by an extracellular phosphopolysaccharide from Lactobacillus delbrueckii ssp. bulgaricus. Int J Food Microbiol. 1998;40(3):169-175.
Crossref - Nishimura J. Exopolysaccharides produced from Lactobacillus delbrueckii subsp. bulgaricus. Adv Microbiol. 2014;4(14):1017-102.
Crossref - Nakajima H, Suzuki Y, Hirota T. Cholesterol lowering activity of ropy fermented milk. J Food Sci. 1992;57(6):1327-1329.
Crossref - Maeda H, Zhu X, Omura K, Suzuki S, Kitamura S. Effects of an exopolysaccharide (kefiran) on lipids, blood pressure, blood glucose, and constipation. Biofactors. 2004;22(1-4):197-200.
Crossref - Ryan PM, Ross RP, Fitzgerald GF, Caplice NM, Stanton C. Sugar-coated: exopolysaccharide producing lactic acid bacteria for food and human health applications. Food Funct. 2015;6:679-693.
Crossref - Aditya T, Allain JP, Jaramillo C, Restrepo AM. Surface Modification of Bacterial Cellulose for Biomedical Applications. Int J Mol Sci. 2022;23(2):610.
Crossref - Casillo A, Lanzetta R, Parrilli M, Corsaro MM. Exopolysaccharides from marine and marine extremophilic bacteria: Structures, properties, ecological roles and applications. Mar Drugs. 2018;16(2):69.
Crossref - Wang J, Salem DR, Sani RK. Extremophilic exopolysaccharides: A review and new perspectives on engineering strategies and applications. Carbohydr Polym. 2019;205:8-26.
Crossref - Isfahani FM, Tahmourespour A, Hoodaji M, Ataabadi M, Mohammadi A. Characterizing the new bacterial isolates of high yielding exopolysaccharides under hypersaline conditions. J Clean Prod. 2018;185:922-928.
Crossref - Knowles EJ, Castenholz RW. Effect of exogenous extracellular polysaccharides on the desiccation and freezing tolerance of rock-inhabiting phototrophic microorganisms. FEMS Microbiol Ecol. 2008;66(2):261-270.
Crossref - Kaur N, Dey P. Bacterial Exopolysaccharides as Emerging Bioactive Macromolecules: From Fundamentals to Applications. Res Microbiol. 2022;29(174):104024.
Crossref - Kuschmierz L, Meyer M, Brasen C, Wingender J, Schmitz OJ, Siebers B. Exopolysaccharide composition and size in Sulfolobus acidocaldarius biofilms. Front Microbiol. 2022;13:982745.
Crossref - Secor PR, Michaels LA, Bublitz DC, Jennings LK, Singh PK. The Depletion Mechanism Actuates Bacterial Aggregation by Exopolysaccharides and Determines Species Distribution & Composition in Bacterial Aggregates. Front Cell Infect Microbiol. 2022;12:86973632.
Crossref - Balducci E, Papi F, Capialbi DE, Del Bino L. Polysaccharides’ Structures and Functions in Biofilm Architecture of Antimicrobial Resistant (AMR) Pathogens. Int J Mol Sci. 2023;24(4):4030.
Crossref - Lynch KM, Zannini E, Coffey A, Arendt EK. Lactic Acid Bacteria Exopolysaccharides in Foods and Beverages: Isolation, Properties, Characterization, and Health Benefits. Annu Rev Food Sci Technol. 2018;9:155-176.
Crossref - Barcelos MCS, Vespermann KAC, Pelissari FM, Molina G. Current status of biotechnological production and applications of microbial exopolysaccharides. Crit Rev Food Sci Nutr. 2020;60(9):1475-1495.
Crossref - Zhang R, Edgar KJ. Properties, chemistry, and applications of the bioactive polysaccharide curdlan. Biomacromol. 2014;15(4):1079-1096.
Crossref - Jiang Y, Yang Z. A functional and genetic overview of exopolysaccharides produced by Lactobacillus plantarum. J Funct Foods. 2018;47:229-240.
Crossref - Oerlemans MMP, Akkerman R, Ferrari M, Walvoort MTC, De Vos P. Benefits of bacteria-derived exopolysaccharides on gastrointestinal microbiota, immunity and health. J Funct Foods. 2021;76:104289.
Crossref - Kanamarlapudi SLRK, Muddada S. Characterization of exopolysaccharide produced by Streptococcus thermophilus CC30. Biomed Res Int. 2017;2017: e4201809.
Crossref - Zhou Y, Cui Y, Qu X. Exopolysaccharides of lactic acid bacteria: Structure, bioactivity and associations: a review. Carbohydr Polym. 2019;207:317-332.
Crossref - Rajoka MSR, Wu Y, Mehwish HM, Bansal M, Zhao L. Lactobacillus exopolysaccharides: new perspectives on engineering strategies, physicochemical functions, and immunomodulatory effects on host health. Trends Food Sci Technol. 2020;103:36-48.
Crossref - Goh YF, Roder HL, Chan SH, et al. Associational resistance to predation by protists in a mixed species biofilm. Appl Environ Microbiol. 2023;89(2): e01741-22.
Crossref - El-Ghonemy DH. Antioxidant and antimicrobial activities of exopolysaccharides produced by a novel Aspergillus sp. DHE6 under optimized submerged fermentation conditions. Biocatal Agric Biotechnol. 2021;36:102150.
Crossref - Zhang J, Cao Y, Wang J, et al. Physicochemical characteristics and bioactivities of the exopolysaccharide and its sulphated polymer from Streptococcus thermophilus GST-6. Carbohydr Polym. 2016;146:368-375.
Crossref - Rani RP, Anandharaj M, David RA. Characterization of a novel exopolysaccharide produced by Lactobacillus gasseri FR4 and demonstration of its in vitro biological properties. Int J Biol Macromol. 2018;109:772-783.
Crossref - Ayyash M, Abu-Jdayil B, Itsaranuwat P, et al. Exopolysaccharide produced by the potential probiotic Lactococcus garvieae C47: structural characteristics, rheological properties, bioactivities and impact on fermented camel milk. Food Chem. 2020;333:127418.
Crossref - Fan Y, Li X, Tian R, Tang R, Zhang J. Characterization and biological activity of a novel exopolysaccharide produced by Pediococcus pentosaceus SSC-12 from silage. Microorganisms. 2022;10(1):18.
Crossref - Matsubara VH, Wang Y, Bandara HMHN, Mayer MPA, Samaranayake LP. Probiotic lactobacilli inhibit early stages of Candida albicans biofilm development by reducing their growth, cell adhesion, and filamentation. Appl Microbiol Biotechnol. 2016;100(14):6415-6426.
Crossref - Nehal F, Sahnoun M, Smaoui S, Jaouadi B, Bejar S, Mohammed S. Characterization, high production and antimicrobial activity of exopolysaccharides from Lactococcus lactis F-mou. Microb Pathog. 2019;132:10-19.
Crossref - Andrew M, Jayaraman G. Structural features of microbial exopolysaccharides in relation to their antioxidant activity. Carbohydr Res. 2020;487:107881.
Crossref - Abdhul K, Ganesh M, Shanmughapriya S, Kanagavel M, Anbarasu K, Natarajaseenivasan K. Antioxidant activity of exopolysaccharide from probiotic strain Enterococcus faecium (BDU7) from Ngari. Int J Biol Macromol. 2014;70:450-454.
Crossref - Hu X, Pang X, Wang PG, Chen M. Isolation and characterization of an antioxidant exopolysaccharide produced by Bacillus sp. S-1 from Sichuan Pickles. Carbohydr Polym. 2019;204:9-16.
Crossref - Paikra SK, Panda J, Sahoo G, Mishra M. Characterization of exopolysaccharide derived from Enterobacter ludwigii and its possible role as an emulsifier. 3 Biotech. 2022;12(9):212.
Crossref - Tang W, Dong M, Wang W, et al. Structural characterization and antioxidant property of released exopolysaccharides from Lactobacillus delbrueckii ssp. bulgaricus SRFM-1. Carbohydr Polym. 2017;173:654-664.
Crossref - Yang X, Wu J, An F, et al. Structure characterization, antioxidant and emulsifying capacities of exopolysaccharide derived from Tetragenococcus halophilus SNTH-8. Int J Biol Macromol. 2022;208:288-298.
Crossref - Li F, Hu X, Sun X, et al. Effect of fermentation pH on the structure, rheological properties, and antioxidant activities of exopolysaccharides produced by Alteromonas australica QD. Glycoconjugate J. 2022;39(6):773-787.
Crossref - Wang W, Ju Y, Liu N, Shi S, Hao L. Structural characteristics of microbial exopolysaccharides in association with their biological activities: a review. Chem Biol Techno Agric. 2023;10(1):137.
Crossref - Ramamurthy D, Nundalall T, Cingo S, et al. Recent advances in immunotherapies against infectious diseases. Immunother Adv. 2021;1(1):ITaa007.
Crossref - Misaki A, Kakuta M, Sasaki T, Tanaka M, Miyaji H. Studies on interrelation of structure and antitumor effects of polysaccharides: antitumor action of periodate-modified, branched (1→3)-β-D-glucan of Auricularia auricula-judae, and other polysaccharides containing (1→3)-glycosidic linkages. Carbohydr Res. 1981;92(1):115-129.
Crossref - Demleitner S, Kraus J, Franz G. Synthesis and antitumour activity of derivatives of curdlan and lichenan branched at C-6. Carbohyd Res. 1992;226(2):239-246.
Crossref - Ji X, Peng B, Ding H, Cui B, Nie H, Yan Y. Purification, structure and biological activity of pumpkin polysaccharides: a review. Food Rev Int. 2023;39(1):307-319.
Crossref - Öner ET, Hernández L, Combie J. Review of Levan polysaccharide: From a century of past experiences to future prospects. Biotechnol Adv. 2016;34(5):827-844.
Crossref - Kasaai MR. A comparative study of molecular structure, solution properties and food application for three branched polysaccharides: amylopectin, glycogen, and dextran. Curr Trends Polym Sci. 2012;16:49-63.
- Bohn JA, BeMiller JN. (1→3)-β-D-glucans as biological response modifiers: a review of structure-functional activity relationships. Carbohydr Polym. 1995;28(1):3-14.
Crossref - Wang K, Li W, Rui X, Chen X, Jiang M, Dong M. Structural characterization and bioactivity of released exopolysaccharides from Lactobacillus plantarum 70810. Int J Biol Macromol. 2014;67:71-78.
Crossref - Wang N, Shan Z, Jia X, et al. Galf-containing polysaccharides from medicinal molds: sources, structures and bioactive properties. Trends Food Sci Technol. 2023;131:244-263.
Crossref - Notermans S, Veeneman GH, Van Zuylen CWEM, Hoogerhout P, Van Boom JH. (1→5)-linked b-D-galactofuranosides are immunodominant in extracellular polysaccharides of Penicillium and Aspergillus species. Mol Immunol. 1988;25(10):975-979.
Crossref - Jia K, Wei M, He Y, Wang Y, Wei H, Tao X. Characterization of novel exopolysaccharides from Enterococcus hirae WEHI01 and its immunomodulatory activity. Foods. 2022;11(21):3538.
Crossref - Yildiz H, Karatas N. Microbial exopolysaccharides: resources and bioactive properties. Process Biochem. 2018;72:41-46.
Crossref - Liu Y, Mao K, Zhang N, et al. Structural characterization and immunomodulatory effects of extracellular polysaccharide from Lactobacillus paracasei VL8 obtained by gradient ethanol precipitation. J Food Sci. 2022;87(5):2034-2047.
Crossref - Liao Y, Gao M, Wang Y, Liu X, Zhong C, Jia S. Structural characterization and immunomodulatory activity of exopolysaccharide from Aureobasidium pullulans CGMCC 23063. Carbohydr Polym. 2022;288:119366.
Crossref - Xu Y, Cui Y, Wang X, et al. Purification, characterization and bioactivity of exopolysaccharides produced by Lactobacillus plantarum KX041. Int J Biol Macromol. 2019;128:480-492.
Crossref - Wang C, Li W, Chen Z, et al. Effects of simulated gastrointestinal digestion in vitro on the chemical properties, antioxidant activity, α-amylase and α-glucosidase inhibitory activity of polysaccharides from Inonotus obliquus. Food Res Int. 2018;103:280-288.
Crossref - Yang Y, Liu J, Tan Y, Wang S, Chen H, Zhou A. Progress in understanding the structure-activity relationship and hypoglycemic mechanism of polysaccharides. Food Science. 2021;42(23):355-363.
Crossref - Mansel BW, Ryan TM, Chen H-L, Lundin L, Williams MAK. Polysaccharide conformations measured by solution state X-ray scattering. Chem Phys Lett. 2020;739:136951.
Crossref - Yu X. Study on structure, synthesis-related enzymes and hypoglycemic activity of exopolysaccharide from Cordyceps militaris mycelium [thesis]. Tianjin University of Science & Technology. 2019.
- Mohd Nadzir M, Nurhayati RW, Idris FN, Nguyen MH. Biomedical applications of bacterial exopolysaccharides: a review. Polymers. 2021;13(4):530.
Crossref - Schmid J, Sieber V, Rehm B. Bacterial exopolysaccharides: biosynthesis pathways and engineering strategies. Front Microbiol. 2015;6:496.
Crossref - Angelin J, Kavitha M. Exopolysaccharides from probiotic bacteria and their health potential. Int J Biol Macromol. 2020;162:853-865.
Crossref - Diaz-Cornejo S, Otero MC, Banerjee A, Gordillo-Fuenzalida F. Biological properties of exopolysaccharides produced by Bacillus spp. Microbiol Res. 2023;268:127276.
Crossref - Schmid J. Recent insights in microbial exopolysaccharide biosynthesis and engineering strategies. Curr Opin Biotechnol. 2018;53:130-136.
Crossref - Rehm BHA. Bacterial polymers: biosynthesis, modifications and applications. Nat Rev Genet. 2010;8(8):578-592.
Crossref - Islam ST, Lam JS. Synthesis of bacterial polysaccharides via the Wzy-dependent pathway. Can J Microbiol. 2014;60(11):697-716.
Crossref - Rana S, Upadhyay LSB. Microbial exopolysaccharides: synthesis pathways, types and their commercial applications. Int J Biol Macromol. 2020;157:577-583.
Crossref - Perez-Burgos M, Garcia-Romero I, Jung J, Schander E, Valvano MA, Sogaard-Andersen L. Characterization of the exopolysaccharide biosynthesis pathway in Myxococcus xanthus. J Bacteriol. 2020;202(19):e00335-20.
Crossref - Cuthbertson L, Kos V, Whitfield C. ABC transporters involved in export of cell surface glycoconjugates. Microbiol Mol Biol Rev. 2010;74(3): 341-362.
Crossref - Huszczynski SM, Hao Y, Lam JS, Khursigaraa CM. Identification of the Pseudomonas aeruginosa O17 and O15 O-specific antigen biosynthesis loci reveals an ABC transporter-dependent synthesis pathway and mechanisms of genetic diversity. J Bacteriol. 2020;202(19):e00347-20.
Crossref - Krasteva PV, Bernal-Bayard J, Travier L, et al. Insights into the structure and assembly of a bacterial cellulose secretion system. Nat Commun. 2017;8(1):2065.
Crossref - Ua-Arak T, Jakob F, Vogel RF. Fermentation pH modulates the size distributions and functional properties of Gluconobacter albidus TMW 2.1191 levan. Front Microbiol. 2017;8:807.
Crossref - Yimaz T, Simseko. Potential health benefits of ropy exopolysaccharides produced by Lactobacillus plantarum. Molecules. 2020;25(14):3293.
Crossref - Netrusov AI, Liyaskina EV, Kurgaeva IV, Liyaskina AU, Yang G, Revin VV. Exopolysaccharides producing bacteria: a review. Microorganisms. 2023;11(6):1541.
Crossref - Liyaskina EV, Rakova NA, Kitykina AA, et al. Production and characterization of the exopolysaccharide from strain Paenibacillus polymyxa 2020. PLoS ONE. 2020;16(7):e0253482.
Crossref - Efremenko E, Senko O, Maslova O, Stepanov N, Aslanli A, Lyagin I. Biocatalysts in synthesis of microbial polysaccharides: properties and development trends. Catalysts. 2022;12(11):1377.
Crossref - Liang T-W, Wang S-L. Recent advances in exopolysaccharides from Paenibacillus spp.: production, isolation, structure, and bioactivities. Marine Drugs. 2015;13(4):1847-1863.
Crossref - Ergene E, Avci A. Effects of Cultural Conditions on Exopolysaccharide Production by Bacillus sp. ZBP4. J Agr Sci-Tarim Bili. 2018;24(3):386-393
- Sorensen HM, Rochfort KD, Maye S, et al. Exopolysaccharides of lactic acid bacteria: Production, purification and health benefits towards functional food. Nutrients. 2022;14(14):2938.
Crossref - Radchenkova N, Yildiz SY. Advanced optimization of bioprocess parameters for exopolysaccharides synthesis in extremophiles. Processes. 2025;13(3):822.
Crossref - Elmansy EA, Elkady EM, Asker MS, Abdallah NA, Khalil BE, Amer SK. Improved production of Lactiplantibacillus plantarum RO30 exopolysaccharide (REPS) by optimization of process parameters through statistical experimental designs. BMC Microbiol. 2023;23(1):361.
Crossref - Moselhy MA, Lotfy FS, Makboul HE, Ali AS. Antimicrobial and antioxidant characteristics of exopolysaccharides produced by Agrobacterium tumefaciens CS5 and T1. Egypt J Chem. 2020;63(11):4423-4438
- Al-Abbasi R. Quantification of exopolysaccharide produced by Bacillus subtilis and the effect of different factors on its production. Rafidain J Sci. 2018;27(1):82–91.
Crossref - Vaningelgem F, Zamfir M, Adriany T, De Vuyst L. Fermentation conditions affecting the bacterial growth and exopolysaccharide production by Streptococcus thermophilus ST 111 in milk-based medium. J Appl Microbiol. 2004;97(6):1257–1273.
Crossref - Yadav MK, Song JH, Vasquez R, Lee JS, Kim IH, Kang DK. Methods for detection, extraction, purification, and characterization of exopolysaccharides of lactic acid bacteria—A systematic review. Foods. 2024;13(22):3687.
Crossref - Liu L, Zhang X, Yin L, Zhang H, Li J, Ma Y. Advances and challenges in bioproduction of microbial exopolysaccharides: Synthesis mechanisms, engineering strategies, and future perspectives. Carbohydr Polym. 2025;367:124010.
Crossref - Zhang J, Hu T, Sun Q. A review of structure–activity relationship, enzymatic and genetic modulation of exopolysaccharides from lactic acid bacteria. Food Sci Hum Wellness. 2025;14(8):9250368.
Crossref - Revin VV, Liyaskina EV, Parchaykina MV, et al. Bacterial cellulose-based polymer nanocomposites: a review. Polymers. 2022;14(21):4670.
Crossref - Korcz E, Kerenyi Z, Varga L. Dietary fibers, prebiotics, and exopolysaccharides produced by lactic acid bacteria: potential health benefits with special regard to cholesterol-lowering effects. Food Funct. 2018;9(6):3057-3068.
Crossref - Korcz E, Varga L. Exopolysaccharides from lactic acid bacteria: techno-functional application in the food industry. Trends Food Sci Technol. 2021;110:375-384.
Crossref - Zheng J, Wittouck S, Salvetti E, et al. A taxonomic note on the genus Lactobacillus: description of 23 novel genera, emended description of the genus Lactobacillus Beijerinck 1901, and union of Lactobacillaceae and Leuconostocaceae. Int J Syst Evol Microbiol. 2020;70(4):2782-2858.
Crossref - Xu D, Hu Y, Wu FF, Jin YM, Xu XM, Ganzle MG. Comparison of the functionality of exopolysaccharides produced by sourdough lactic acid bacteria in bread and steamed bread. J Agric Food Chem. 2020;68: 8907-8914.
Crossref - Banerjee A, Sarkar S, Govil T, et al. Extremophilic exopolysaccharides: biotechnologies and wastewater remediation. Front Microbiol. 2021;12:721365.
Crossref - Aslan S, Kapdan IK. Batch kinetics of nitrogen and phosphorus removal. Ecol Eng. 2006;28(1):64-70.
Crossref - Singh G, Thomas PB. Nutrient removal from membrane bioreactor permeates using microalgae and in a microalgae membrane photoreactor. Bioresour Technol. 2012;117:80-85.
Crossref - Sarkar S, Banerjee A, Halder U, Biswas R, Bandopadhyay R. Degradation of synthetic azo dyes of textile industry: a sustainable approach using microbial enzymes. Water Conserv Sci Eng. 2017;2(2):121-131.
Crossref - Crini G, Lichtfouse E. Advantages and disadvantages of techniques used for wastewater treatment. Environ Chem Lett. 2019;17:145-155.
Crossref - Syafiuddin A, Fulazzaky MA. Decolorization kinetics and mass transfer mechanisms of Remazol Brilliant Blue R dye mediated by different fungi. Biotechnol Rep. 2021;29:e00573.
Crossref - Lettinga G, De Man A, Van der Last ARM, et al. Anaerobic treatment of domestic sewage and wastewater. Water Sci Technol. 1993;27(9):67-73.
Crossref - LaPara TM, Alleman JE. Thermophilic aerobic biological wastewater treatment. Water Res. 1999;33(4):895-908.
Crossref - Zheng Y, Ye Z-L, Fang X-L, Li Y-H, Cai W-M. Production and characteristics of a bioflocculant produced by Bacillus sp. F19. Bioresour Technol. 2008;99(16):7686-7691.
Crossref - Guarin TC, Pagilla KR. Microbial community in biofilters for water reuse applications: a critical review. Sci Total Environ. 2021;773:145655.
Crossref - Mendez R, Lema JM. Biofilm Reactors Technology in Wastewater Treatment. In: Melo LF, Bott TR, Fletcher M, Capdeville B. (eds) Biofilms — Science and Technology. NATO ASI Series, vol 223. Springer, Dordrecht.
Crossref - Nicolella C, Van Loosdrecht MCM, Heijnen JJ. Wastewater treatment with particulate biofilm reactors. J Biotechnol. 2000;80(1):1-33.
Crossref - Qureshi N, Singh V. Process economics of renewable biorefineries. In: Qureshi N, Hodge D, Vertès A, eds. Biorefineries: Integrated Biochemical Processes for Liquid Biofuels. Amsterdam, Netherlands: Elsevier; 2014:295-322.
Crossref - Hammes F, Velten S, Egli T, Juhna T. Biotreatment of drinking water. In: Moo-Young M, ed. Comprehensive Biotechnology. 2nd ed. Vol 6. Amsterdam, Netherlands: Elsevier; 2011:517-530.
Crossref - Andersson S. Characterization of bacterial biofilms for wastewater treatment [dissertation]. Kungliga Tekniska Hagskolan. 2009.
- Cui D, Tan C, Deng H, et al. Biosorption mechanism of aqueous Pb²⁺, Cd²⁺, and Ni²⁺ ions on extracellular polymeric substances (EPS). Archaea. 2020; 2020:8891543.
Crossref - Cao Z, Yan W, Ding M, Yuan Y. Construction of microbial consortia for microbial degradation of complex compounds. Front Bioeng Biotechnol. 2022;10:1051233.
Crossref - Qattan SYA. Harnessing bacterial consortia for effective bioremediation: targeted removal of heavy metals, hydrocarbons, and persistent pollutants. Environ Sci Eur. 2025;37(1).
Crossref - Nagaoka M, Hashimoto S, Watanabe T, Yokokura T, Mori Y. Anti-ulcer effects of lactic acid bacteria and their cell wall polysaccharides. Biol Pharm Bull. 1994;17(8):1012-1017.
Crossref - Stack HM, Kearney N, Stanton C, Fitzgerald GF, Ross RP. Association of β-glucan endogenous production with increased stress tolerance of intestinal lactobacilli. Appl Environ Microbiol. 2009;76(2):500-507.
Crossref - Salazar N, Gueimonde M, Hernandez-Barranco AM, Ruas-Madiedo P, de los Reyes-Gavilan CG. Exopolysaccharides produced by intestinal Bifidobacterium strains act as fermentable substrates for human intestinal bacteria. Appl Environ Microbiol. 2008;74(15):4737-4745.
Crossref - Tsuda H, Hara K, Miyamoto T. Binding of mutagens to exopolysaccharide produced by Lactobacillus plantarum mutant strain 301102S. J Dairy Sci. 2008;91(8):2960-2966.
Crossref - Klein MI, Duarte S, Xiao J, Mitra S, Foster TH, Koo H. Structural and molecular basis of the role of starch and sucrose in Streptococcus mutans biofilm development. Appl Environ Microbiol. 2009;75(3):837-841.
Crossref - You X, Li Z, Ma K, et al. Structural characterization and immunomodulatory activity of an exopolysaccharide produced by Lactobacillus helveticus LZ-R-5. Carbohydr Polym. 2020;235:115977.
Crossref - Xiu L, Sheng S, Hu Z, et al. Exopolysaccharides from Lactobacillus kiferi as adjuvant enhanced the immuno-protective against Staphylococcus aureus infection. Int J Biol Macromol. 2020;161:10-23.
Crossref - Gupta PL, Rajput M, Oza T, Trivedi U, Sanghvi G. Eminence of microbial products in cosmetic industry. Nat Prod Bioprospect. 2019;9(4):267-278.
Crossref - Kato I. The application of sodium dextran sulfate to the field of cosmetics. Int J Cosmetic Sci. 2007;29(1): 68-68.
Crossref - Bilanovic D, Starosvetsky J, Armon RH. Preparation of biodegradable xanthan-glycerol hydrogel, foam, film, aerogel and xerogel at room temperature. Carbohydr Polym. 2016;148:243-250:
Crossref - Kumar A, Rao K, Han SS. Application of xanthan gum as polysaccharide in tissue engineering: a review. Carbohydr Polym. 2017;180:128-144.
Crossref - Meyer K, Palmer JW. The polysaccharide of the vitreous humor. J Biol Chem. 1934;107(3):629-634.
Crossref - Maclennan AP. The production of capsules, hyaluronic acid and hyaluronidase by Group A and Group C streptococci. Microbiology. 1956;14(1):134-142.
Crossref - Allemann I, Baumann L. Hyaluronic acid gel (Juvederm™) preparations in the treatment of facial wrinkles and folds. Clin Interv Aging. 2008;3(4):629-634.
Crossref - Guerin M, Boulenguer P, Rinaudo M. Exopolysaccharide for the treatment and/or care of the skin, mucous membranes and/or nails. European patent EP 2827837 B1; 2020.
- Costa OYA, Raaijmakers JM, Kuramae EE. Microbial extracellular polymeric substances: ecological function and impact on soil aggregation. Front Microbiol. 2018;9:1636.
Crossref - Duan Z, Tan X, Shi L, Zeng Q, Ali I, Zhu R. Phosphorus accumulation in extracellular polymeric substances (EPS) of colony-forming cyanobacteria challenges imbalanced nutrient reduction strategies in eutrophic lakes. Environ Sci Technol. 2023;57(4):1600-1612.
Crossref - Acosta-Jurado S, Fuentes-Romero F, Ruiz-Sainz JE, Janczarek M, Vinardell JM. Rhizobial exopolysaccharides: genetic regulation of their synthesis and relevance in symbiosis with legumes. Int J Mol Sci. 2021;22(12):6233.
Crossref - Kondakindi VR, Pabbati R, Erukulla P, Maddela NR, Prasad R. Bioremediation of heavy metals-contaminated sites by microbial extracellular polymeric substances – A critical view. Environ Chem Ecotoxicol. 2024;6:408-421.
Crossref - Ates O. Systems biology of microbial exopolysaccharides production. Front Bioeng Biotechnol. 2015;3:200.
Crossref - EFSA BIOHAZ Panel, Ricci A, Allende A, et al. Scientific opinion on the update of the list of QPS-recommended biological agents intentionally added to food or feed as notified to EFSA†. EFSA Journal. 2017;15(3):4664.
Crossref - U.S. Food and Drug Administration. Q7 Good Manufacturing Practice Guidance for Active Pharmaceutical Ingredients: Guidance for Industry. Published September 2016. Updated April 14, 2020. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/q7-good-manufacturing-practice-guidance-active-pharmaceutical-ingredients-guidance-industry
- Wright O, Stan GB, Ellis T. Building-in biosafety for synthetic biology. Microbiology. 2013;159(7):1221–1235.
Crossref - Ravagnan G, Schmid J. Promising non-model microbial cell factories obtained by genome reduction. Front Bioeng Biotechnol. 2024;12:1427248.
Crossref - Hussain A, Zia KM, Tabasum S, et al. Blends and composites of exopolysaccharides; properties and applications: A review. Int J Biol Macromol. 2017;94(Part A):10-27.
Crossref - Valentine ME, Kirby BD, Withers TR, et al. Generation of a highly attenuated strain of Pseudomonas aeruginosa for commercial production of alginate. Microb Biotechnol. 2020;13(1):162-175.
Crossref
© The Author(s) 2026. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.