ISSN: 0973-7510
E-ISSN: 2581-690X
Dengue fever is a viral disease transmitted by mosquitoes that continues to pose a serious public health threat worldwide, infecting millions annually. Nearly 40% of the global population remains at risk. Thus, early and precise diagnosis is crucial for monitoring the spread of the disease and ensuring appropriate medical care. Here, we report serotyping of infecting DENVs and natural anti-dengue IgG antibodies in total of 207 DENV-infected serum specimens, of which 95 and 112 dengue suspects’ sera collected from Dhaka Medical College Hospital and Chittagong Medical College Hospital, respectively. 35 out of 95 samples from Dhaka were NS1 positive, whereas 17 samples were tested positive by RT-PCR. It was noted that all 17 samples were single DENV serotype, where DENV4 constituted the highest prevalence (35%), followed by DENV2 (29%), DENV1 (24%) and DENV3 (12%). On the other hand, 42 out of 112 NS1 positive samples collected from Chittagong Medical College Hospital were DENV positive by RT-PCR. Among them, the single DENV serotype was 76%, and multiple DENV serotypes were 24% of which DENV4 was also the most prevalent DENV serotype. Investigation of natural anti-dengue IgG antibodies in DENV-infected sera indicated that natural DENV infections resulted both serotype-specific and sero-cross-reactive anti-dengue antibodies. The correlation of infecting DENV serotype and natural anti-dengue IgG and IgM antibody response between Dhaka and Chittagong has been reported in this study. Our analysis revealed that dengue-infected samples from Chittagong presented a more critical situation compared to those from Dhaka, as 24% of the samples demonstrated concurrent infection with two or more DENV serotypes, whereas the Dhaka cases were associated with infection by a single serotype.
DENV Serotypes, DENV Sero-specificity, Primary DENV Infection, Secondary DENV Infection, Concurrent Multiple DENV Infection, Anti-DENV ED3 Antibodies, IgG and IgM Antibody
Dengue, a serious recurrent public health problem especially in developing countries, is caused by four antigenically distinct dengue viruses (DENV) serotypes, DENV1-4. Infection by an individual DENV raises long-lasting immunity against the infecting serotype. However, subsequent infections with different serotypes have long been reported to be associated with severe dengue hemorrhagic fever (DHF) and dengue shock syndrome (DSS) and this is due to a few minute conformational differences on the Envelope Protein Domain 3 (ED3). DENV is a RNA virus whose genome encodes 10 proteins.1 The envelope protein (E-protein) is the outer layer of the virus. It is composed of three domains (ED1~ED3),2-4 where ED2 is involved in membrane fusion, and 3ED3 is the major target of neutralizing antibodies.5 Antibodies against ED3 alone could block dengue infection in
in vitro cell culture studies and in natural dengue infections,6-9 and they could generate long lasting immune response.10-12 Interestingly, ED3 of four DENV serotypes have very similar sequences and structures.2 However, the DENV-specific ED3 could alone determine DENV sero-specificity13-16 and therefore, ED3s are worth considering for investigating DENV infection from natural immune response against DENV infection which may be of interest in future dengue disease management. Endy and co-workers17 stated that DENV disease is a consequence of a complicated interplay of viral residences and host immune responses restricting the efficacy of several DENV vaccine candidates. In addition, the scientific community is still lacking an animal model system for investigating dengue vaccine candidates.18 Thus here comparison between serotyping of infecting DENVs and natural anti-dengue IgG antibodies in 95 DENV-infected samples obtained from Dhaka medical college hospital and that of 112 NS1 positive dengue suspects’ sera collected from Chittagong Medical College, Bangladesh after the dengue outbreak in Chittagong 2019. Serotyping in both cases DENV4 was the most prevalent DENV serotype. From the observations, it is noted that if a patient is infected with DENV1, highly 1ED3-2ED3-3ED3 cross-reacting anti-ED3 IgM & IgG antibodies are generated in Chittagong DENV-infected samples; which is very similar in dengue samples of Dhaka. Since no specific dengue treatment is available and/or potent vaccines are yet to come, so this study involves our efforts to observe natural infections etiology, and find any formulation form this type of correlation.
Collection of DENV-infected human sera
This study was performed with a total of 207 DENV-infected serum specimens of which 95 samples were obtained from Dhaka Medical College Hospital and 112 DENV-infected samples collected from Chittagong Medical College Hospital. Ethical approval has been received from Chittagong University (AERB-FBSCU-20221031-(1)) with written consent of the participants. All patients’ data were totally anonymous and kept confidential. Among the participants, 78.57% were male and 21.42% were female, aged between 19 and 50 years. Of these, 40.47% lived in urban areas and 59.52% in rural regions. Serum samples were collected from hospitalized individuals who were confirmed to have dengue (DENV NS1 positive; July-August 2019) and receiving treatment. The clinical signs and symptoms of dengue infections may include fever, vomiting, myalgia, musculoskeletal pain, rash, retro-orbital discomfort, headache, nausea, diarrhea, ache-pain, jaundice and photophobia. Prior to use, all serum samples were heat-inactivated and stored at -80 °C in 100 µL aliquots.
DENV serotyping
Using an RNA extraction Promega Corporation kit, 100 µL of serum samples were first used to separate and purify total RNAs (Promega Corporation). The cDNAs were created by using of primers specific to DENV.19 In more detail, the reaction mixture (which included RNA extract (7.8 µL), Oligo dT (1 µL), random primer (1 µL), and conserved primer for DENV (0.8 µL) were incubated at 70 °C for 5 minutes, then rapid freezing was performed on ice. After adding the PCR master mix (9.4 L), a thermal cycler (Thermo Fisher Scientific, USA; 25 °C for 5 min, 42 °C for 60 min, and 70 °C for 15 min) was used to conduct the PCR. At last, infection-causing DENV serotypes were determined using DENV serotype-specific primers (DENV1 = 5′-CCC-CGT-AACACT-TTG-ATC-GCT-CCA-TT-3′; DENV2 = 5′-CGC-CACAAG-GGC-CAT-GAA-CAG-3′, DENV3 = 5′-GCA-CAT-GTTGAT-TCC-AGA-GGC-TGT-C-3′; DENV4 = 5′-GTT-TCCAAT-CCC-ATT-CCT-GAA-TGT-GGT-GT-3′) utilizing the standard cyber- green PCR procedure. (cDNA (4 µL); DENV-specific and DENV conserved primers 0.5 µL + 0.5 µL); and master mix (10 µL); H20 (5 µL). The PCR methodology utilized the following procedure: initial denaturation at 96 °C for 2 minutes, followed by denaturation at 96 °C for 15 seconds, annealing at 55 °C for 33 seconds, and extension at 72 °C for 1 minute. Steps 2 through 4 were repeated 45 times with fluorescence data being recorded at the extension step. Furthermore, infecting DENV serotyping determined by Subergreen-based RT-PCR protocol was confirmed by using one-step qRT- PCR serotyping kit (Fast Track Dengue Diagnostics), and the qPCR data was collected using the qTower instrument (Analytik Jena).
Immunological assay
Detection of anti-ED3 antibodies in dengue-infected sera in humans
The presence of anti-DENV ED3 IgG and IgM antibodies (collectively referred to as anti-ED3 antibodies) in human serum samples from naturally infected individuals was evaluated using an ELISA assay performed in 96-well microtiter plates (Nunc), following previously established methods.20,21 Plates were coated overnight at room temperature with 2.5 µg/mL of purified ED3 proteins in phosphate-buffered saline (PBS, pH 7.4; 100 µL per well). After removing any unbound protein with PBS washes, the wells were blocked with 1% BSA in PBS for 45 minutes at 37 °C. Following an additional PBS wash, dengue-positive sera were added at an initial dilution of 1:50 in 0.1% BSA/PBS, then serially diluted threefold and incubated at 37 °C for 2 hours. After thoroughly washing three times with PBS-0.05% Tween-20 and once with PBS, unbound antibodies were eliminated. After blotting the microtiter plates, 100 µL/well of anti-human IgG-HRP conjugate (1:4000 dilution) and anti-human IgM-HRP conjugate (1:10,000 dilution) were added to 0.1% BSA in PBS-0.05% Tween-20, and the plates were incubated for 90 minutes at 37 °C. Again three PBS-0.05% Tween-20 washes and one PBS wash were used to eliminate the any unbound conjugates. A microplate reader (Dynatech Microplate Reader) was used to evaluate the color intensity after 20 minutes of incubation at 450 nm (OD450 nm) after the substrate OPD (o-phenylenediamine) was added at a concentration of 0.4 mg/ml supplemented with 4 mM H2O2 (100 µL/well). Using a threshold of OD450 nm = 0.1 and 0.4 above the background values for IgM and IgG, respectively, antibody titers were computed from the power fitting of OD450 nm versus the reciprocal of the antisera dilution.21
DENV seroprevalence in Dhaka
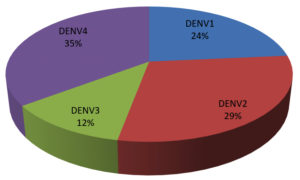
Patients presenting with clinical manifestations consistent with dengue infection and confirmed by expert physicians were included as subjects with naturally acquired dengue infection in this study. All 95 subjects included in this study were screened by NS1-based dengue diagnosis and among them 35 samples were NS1 positive (DENV-infected) and 60 were NS1 negative (dengue suspects). However Dengue serotyping study was carried out on 35 NS1 positive samples and 17 DENV positive samples (tested by RT-PCR). All of them were single DENV serotype where DENV4 constituted the highest prevalent (35%) followed by DENV2 (29%), DENV1 (24%) and DENV3 (12%) (Figure 1). Serotyping of rest of the 60 samples were performed by IgG antibody titer.
Figure 1. DENV Seroprevalence in Dhaka, Bangladesh: Serotyping was done by RT-PCR. Among 35 samples tested 17 were positive for DENV serotypes. All of them were single DENV serotype where DENV4 constituted the highest prevalent 35%
DENV seroprevalence in Chittagong
According to RT-PCR DENV serotyping, 42 out of 112 samples tested positive for DENV, with 32 (76%) having a single DENV serotype and 10 (24%) having two or more DENV serotypes concurrently.22 DENV4 was the most common serotype among single DENV serotype infections, followed by DENV2, DENV1, and DENV3 serotypes which were in contrast to previous reports from Bangladesh.23 Among the 42 DENV positive patients, single-serotype infections were detected as follows: DENV-1 (7 patients), DENV-2 (8 patients), DENV-3 (4 patients), and DENV-4 (13 patients). Concurrent infections were also observed, including DENV-1/3 (5 patients), DENV-1/4 (3 patients), DENV-2/3 (1 patient), and DENV-1/3/4 (1 patient).
Correlation between antibodies of Natural Infections in Dhaka and Chittagong DENV-infected patients
Single DENV infections
Among 112 human samples from Chittagong, 42 DENV positive samples were investigated for antibody by ELISA IgG and IgM. These types of antibodies were compared with Dhaka DENV-infected samples by ELISA IgG. From the observations, it was noted that patients infected with DENV1 produced high levels of cross-reactive anti-ED3 IgM and IgG antibodies (1ED3-2ED3-3ED3) in the Chittagong samples (Figures 2a and 2b) which was very similar to the antibody response observed in Dhaka DENV-infected samples (Figure 2c). In DENV2-infected Chittagong samples, IgM antibodies showed cross-reactivity with 1ED3 and 4ED3 (Figure 2a), whereas IgG antibodies from both Chittagong and Dhaka samples showed cross-reactivity with 1ED3-2ED3-3ED3-4ED3 (Figures 2b and 2c). DENV3 infection produced more specific 3ED3-targeted IgG antibodies in both Chittagong and Dhaka samples (Figures 2b and 2c). It is very similar in mice infection. IgG responses in DENV4-infected samples from Chittagong and Dhaka showed markedly low reactivity against 4ED3 (Figures 2b and 2c). In DENV-4-infected samples, IgM antibody responses were highly sero-cross-reactive with 1ED3-2ED3-3ED3-4ED3 (Figure 2a). Although a single DENV infection induces cross-reactive antibodies, it may be more beneficial for the human body in providing protection against the virus.
Figure 2. Correlation between Dhaka and Chittagong natural single DENV infections: (a) Anti-ED3 antibody titers of IgM of Chittagong samples (b) Anti-ED3 antibody titers of IgG of Chittagong samples (c) Anti-ED3 antibody titers of IgG of Dhaka samples in natural infection which shows that DENV1 infection generated highly 1ED3-2ED3-3ED3 cross-reacting anti-ED3 IgM & IgG antibodies in Chittagong samples and which is very similar in Dhaka samples
Multiple DENV infections
In multiple DENV serotype infections, RT-PCR–based IgM and IgG antibody titers from Chittagong samples of DENV1,4 and DENV1,3 infections showed sero-cross-reactive anti-ED3 antibodies against 1ED3-2ED3-3ED3-4ED3 (Figures 3a and b) which is very similar in mice study.24 On the other hand, in Dhaka samples, IgG antibody-based serotyping of 60 cases showed that DENV1, DENV3, and DENV4 infections were highly specific to 1ED3, 3ED3, and 4ED3, respectively, whereas DENV2 serotyping was not detected based on antibody titers (Figure 3c). But these samples (Figure 3c) were not DENV positive identified by RT-PCR serotyping. However, during the acute stage of infection, RT-PCR is regarded as the gold standard in DENV serotyping for verifying the presence of viral RNA in the infected sera.25 It may, however, have a significant false negative prediction rate in the very early and very late stages of infection.26-28 As a result, RT-PCR by itself is insufficient for diagnosing dengue. So it is clear that natural infection in both areas was mostly sero-cross reactive and both scenery was almost same.
Figure 3. Correlation between Dhaka and Chittagong natural multiple DENV infections: (a) Anti-ED3 antibody titers of IgM in natural infection of Chittagong samples (b) Anti-ED3 antibody titers of IgG in natural infection of Chittagong samples. (c) Serotyping of rest of the samples were performed by IgG antibody titer of Dhaka samples shows natural study was mostly cross-reactive
The observations from this study suggest that DENV serotype-specific ED3s could serve as a useful model for investigating DENV infections in detail. ED3s may be applied for both the detection and investigation of natural and artificial dengue infections. It has been noted that in mice study, antibodies are mostly serotype-specific (personal communication) whereas in natural dengue infection, antibodies are mostly cross reactive. This observation may be due to the fact that the study on mice was in fixed/controlled environment whereas we have no idea whether humans have been affected by any other dengue virus before. In this study we analyzed 207 DENV-infected samples of Dhaka and Chittagong area. We have come to the conclusion that DENV1 is mostly 1ED3-2ED3-3ED3 sero-cross reactive in natural infection which is similar to mice study. So this concept helps for the development of dengue model system in future.
ACKNOWLEDGMENTS
The authors acknowledge Professor Yutaka Kuroda of Tokyo University of Agriculture and Technology, Japan, for DENV-ED3 variants, and Chittagong Medical College Hospital and Dhaka Medical College Hospital for their generalised support, providing anti-dengue sera.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
MMI conceptualized the study. MS collected patients sera. MMI funding acquisition. MGR performed experiments. MGR and MMI wrote the manuscript. AS supervised the study and revised the manuscript. All authors read and approved the final manuscript for publication.
FUNDING
The research was funded through the revenue budget of the University of Chittagong, Bangladesh.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
This study was approved by the Institutional Ethical Committee, University of Chittagong, Bangladesh, under ethical approval number AERB-FBSCU-20221031-(1).
INFORMED CONSENT
Written informed consent was obtained from the participants before enrolling in the study.
- Mondotte JA, Lozach P-Y, Amara A, Gamarnik AV. Essential Role of Dengue Virus Envelope Protein N Glycosylation at Asparagine-67 during Viral Propagation. J Virol. 2007;81(13):7136-7148.
Crossref - Modis Y, Ogata S, Clements D, Harrison SC. Structure of the dengue virus envelope protein after membrane fusion. Nature. 2004;427(2004):313-319.
Crossref - Modis Y, Ogata S, Clements D, Harrison SC. A ligand-binding pocket in the dengue virus envelope glycoprotein. Proc Natl Acad Sci U S A. 2003;100(12):6986-6991.
Crossref - Zhang X, Jia R, Shen H, Wang M, Yin Z, Cheng A. Structures and functions of the envelope glycoprotein in flavivirus infections. Viruses. 2017;9(1):338.
Crossref - Gromowski GD, Barrett ADT. Characterization of an antigenic site that contains a dominant, type-specific neutralization determinant on the envelope protein domain III (ED3) of dengue 2 virus. Virology. 2007;366(2):349-360.
Crossref - Gromowski GD, Barrett ND, Barrett ADT. Characterization of Dengue Virus Complex-Specific Neutralizing Epitopes on Envelope Protein Domain III of Dengue 2 Virus. J Virol. 2008;82(17):8828-8837.
Crossref - Matsui K, Gromowski GD, Li L, Schuh AJ, Lee JC, Barrett ADT. Characterization of dengue complex-reactive epitopes on dengue 3 virus envelope protein domain III. Virology. 2009;384(1):16-20.
Crossref - Crill WD, Roehrig JT. Monoclonal Antibodies That Bind to Domain III of Dengue Virus E Glycoprotein Are the Most Efficient Blockers of Virus Adsorption to Vero Cells. J Virol. 2001;75(16):7769-7773.
Crossref - Mune M, Rodriguez R, Ramirez R, et al. Carboxy-terminally truncated Dengue 4 virus envelope glycoprotein expressed in Pichia pastoris induced neutralizing antibodies and resistance to Dengue 4 virus challenge in mice. Arch Virol. 2003;148(11):2267-2273.
Crossref - Apt D, Raviprakash K, Brinkman A, et al. Tetravalent neutralizing antibody response against four dengue serotypes by a single chimeric dengue envelope antigen. Vaccine. 2006;24(3):335-344.
Crossref - Etemad B, Batra G, Raut R, et al. An envelope domain III-based chimeric antigen produced in Pichia pastoris elicits neutralizing antibodies against all four dengue virus serotypes. Am J Trop Med Hyg. 2008;79(3):353-363.
Crossref - Elahi M, Islam MM, Noguchi K, Yohda M, Toh H, Kuroda Y. Computational prediction and experimental characterization of a “size switch type repacking” during the evolution of dengue envelope protein domain III (ED3). Biochim Biophys Acta. 2014;1844(3):585-592.
Crossref - Gubler DJ. The global emergence/resurgence of arboviral diseases as public health problems. Arch Med Res. 2002;33(4):330-342.
Crossref - Wahala WMPB, Kraus AA, Haymore LB, Accavitti-Loper MA, de Silva AM. Dengue virus neutralization by human immune sera: Role of envelope protein domain III-reactive antibody. Virology. 2009;392(1): 103-113.
Crossref - Brandler S, Lucas-Hourani M, Moris A, et al. Pediatric measles vaccine expressing a dengue antigen induces durable serotype-specific neutralizing antibodies to dengue virus. PLoS Negl Trop Dis. 2007;1(3):e96.
Crossref - Hussain M, Idrees M, Afzal S. Development of Global Consensus of Dengue Virus Envelope Glycoprotein for Epitopes Based Vaccine Design. Curr Comput Aided-Drug Des. 2015;11(1):84-97.
Crossref - Sabchareon A, Wallace D, Sirivichayakul C. Protective efficacy of the recombinant, live-attenuated, CYD tetravalent dengue vaccine in Thai schoolchildren: a randomised, controlled phase 2b trial. Lancet. 2012;380(9853):1559-67.
Crossref - Holmes EC, Twiddy SS. The origin, emergence and evolutionary genetics of dengue virus. Infect Genet Evol. 2003;3(1):19-28.
Crossref - Yong YK, Thayan R, Chong HT, Tan CT, Sekaran SD. Rapid detection and serotyping of dengue virus by multiplex RT-PCR and real-time SYBR green RT- PCR. Singapore Med J. 2007;48(7):662-668.
- Kulkarni MR, Islam MM, Numoto N, et al. Structural and biophysical analysis of sero-specific immune responses using epitope grafted Dengue ED3 mutants. Biochim Biophys Acta. 2015;1854(10 Pt A):1438-1443.
Crossref - Islam MM, Miura S, Hasan MN, Rahman N, Kuroda Y. Anti-Dengue ED3 Long-Term Immune Response with T-Cell Memory Generated Using Solubility Controlling Peptide Tags. Front Immunol. 2020;11:333.
Crossref - Roy MG, Uddin K, Islam D, Singh A, Islam MM. All four dengue virus serotypes co-circulate in concurrent dengue infections in a single dengue session in Chittagong, Bangladesh. Bioresearch Commun. 2022;8(1):1042-1048.
Crossref - Shirin T, Muraduzzaman AKM, Alam AN, et al. Largest dengue outbreak of the decade with high fatality may be due to reemergence of DEN-3 serotype in Dhaka, Bangladesh, necessitating immediate public health attention. New Microb New Infect. 2019;29:100511.
Crossref - Islam MD, Sharmin T, Tipo IH, et al. The Immunogenicity of DENV1-4 ED3s Strongly Differ despite Their Almost Identical Three-Dimensional Structures and High Sequence Similarities. Int J Mol Sci. 2023;24(3):2393.
Crossref - Bhatnagar J, Blau DM, Shieh WJ, et al. Molecular detection and typing of dengue viruses from archived tissues of fatal cases by RT-PCR and sequencing: Diagnostic and epidemiologic implications. Am J Trop Med Hyg. 2012;86(2):335-340.
Crossref - Anwar A, Wan G, Chua KB, August JT, Too HP. Evaluation of Pre-Analytical Variables in the Quantification of Dengue Virus by Real-Time Polymerase Chain Reaction. J Mol Diagnostics. 2009;11(6):537-542.
Crossref - Fleige S, Pfaffl MW. RNA integrity and the effect on the real-time qRT-PCR performance. Mol Aspects Med. 2006;27(2-3)126-139.
Crossref - Koo C, Kaur S, Teh ZY, et al. Genetic variability in probe binding regions explains false negative results of a molecular assay for the detection of dengue Virus. Vector-Borne Zoonotic Dis. 2016;16(7)489-495.
Crossref
© The Author(s) 2026. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.