ISSN: 0973-7510
E-ISSN: 2581-690X
Tuberculosis (TB) caused by acid-fast bacilli, specifically Mycobacterium tuberculosis, is indeed one of the most serious infectious diseases globally. Early detection, identification, and treatment of active tuberculosis reduce the risk of infection. The key challenge in controlling the burden of illness is the absence of a reliable diagnostic method. The primary objective of this research is to evaluate the role of Truenat in comparison with the Ziehl-Neelsen technique and fluorescent microscopy in patients suspected of having Koch’s bacillus, and to determine the prevalence of rifampicin resistance among the positive cases. A prospective cross-sectional study was carried out in our laboratory for one year. Overall, 229 samples were tested using all three methods, 60% were from males and 40% from females, with the most severely affected individuals aged 21-45 years (60.6%). The positivity of Ziehl-Neelsen stain was 22.3% (n = 51), fluorescent stain was 24.0% (n = 55), and Truenat was 26.6% (n = 61), respectively. Rifampicin resistance was identified in one positive case. The conventional methods were compared to the molecular technique, considering it to be the gold standard for assessing sensitivity, specificity, positive predictive value, and negative predictive value. The recorded value was quantitatively analysed, with a P-value below 0.05 being statistically significant. Truenat has demonstrated itself to be a more effective method when compared to conventional techniques. Among the microscopy techniques, the fluorescence stain is found to be more accurate and rapid than the Ziehl-Neelsen stain.
Acid-fast Bacilli, Fluorescent Stain, Mycobacterium tuberculosis, Truenat, Ziehl-Neelsen Stain
Tuberculosis disease is found to affect all parts of the world, where the illness has become more complicated due to drug resistance, raising worries for global health. Early detection of this infection is difficult in healthcare settings.1 The World Health Organisation (WHO) reports that three million people currently die from tuberculosis.2 It is the most prevalent infectious disease that results in death worldwide, following the human immunodeficiency virus (HIV).3 Several investigations can be carried out if Mycobacterium tuberculosis is suspected, which include medical examination, X-rays of the chest, conventional staining, cultures for MTB, immunological techniques, and molecular techniques.4,5 Smear microscopy is performed using Ziehl-Neelsen (ZN) or fluorescent staining for the detection of Mycobacteria. It’s often the only method of diagnosis available for evaluating patients with TB symptoms in underdeveloped nations. Conventional microscopy is quick and inexpensive in large patient populations with limited resources.4,5 They are also beneficial in evaluating the therapeutic response and finding out whether or not the patient has been cured of the disease at the end of their treatment.6 However, the method’s biggest drawback is its poor sensitivity, particularly for individuals who are co-infected with HIV.7
Culture in LJ medium is the standard reference for the bacteriological detection of tuberculosis. Although it is more sensitive than microscopy, it requires four to eight weeks to yield results because tubercle bacilli are slow-growing organisms.8,9 In contrast, microscopic inspection provides an immediate result. Therefore, the most promising diagnostic approach to solving this issue is the use of molecular tools like PCR.10
Molbio Diagnostics, based in Bangalore, India, has created a novel chip-based molecular-level detection device called Truenat, which consists of the Trueprep AUTO, TruenatTM MTB, and MTB-rifampicin (RIF) kits. The Truenat assay uses real-time polymerase chain reaction (RT-PCR) to quickly extract deoxyribonucleic acid (DNA) from the samples in less than an hour to identify tuberculosis (TB) along with rifampicin resistance (RIF).11
TB is curable and can be treated with antibiotics, but multidrug-resistant MTB continues to be a threat to society. Quick identification of patients and the starting of medication are one of the pillars of the TB control programme for preventing transmission, relapse, and the development of drug resistance. Considering the constraints of smear microscopy, the Indian molecular methodology can be employed to overcome the hurdles encountered with it for the identification of Mycobacterium tuberculosis along with drug resistance. Hence, present research was carried out in a resource limited setting to determine the performance of the Truenat in the identification of tuberculosis in comparison to conventional microscopy methods regarding sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV). The identification of rifampicin drug resistance among the isolated positive cases was also another objective.
A prospective observational analysis was carried out in the central laboratory of our tertiary care hospital from February 2023 to January 2024. The institutional ethical committee gave its approval (Reg. No. ECR/1487/Inst/TN/2020) (MAPIMS/IEC/52/2023) before the study was conducted.
Inclusion and exclusion criteria
Patients of all ages and sexes who presented with symptoms of unexplained fever with evening rise of temperature, cough with expectoration for two or more weeks, unexplained weight loss, and loss of appetite, suspected of having Mycobacterium tuberculosis, were included. Extrapulmonary tuberculosis cases were also included. Those already started with anti-tubercular treatment and known cases of carcinoma of the lung were excluded from the study. Clinical samples of sputum, bronchoalveolar lavage, pleural fluid, and endotracheal secretions were collected from the suspected cases (outpatients and inpatients). Data collection was carried out using a proforma and handled with absolute confidence. Before this, informed and written consent from participants was obtained for adults. Only informed consent was provided for underage patients.
Sample collection and processing
The TB suspects were instructed to collect one spot plus an additional specimen the next day and to place them into a wide-mouth, clean, heat- and leak-resistant sputum bottle. Based on the location, method, and degree of contamination, extrapulmonary specimens were split into two categories: Aseptically collected specimens, which were typically sterile and devoid of other pathogens, such as pleural fluid, were obtained by pleural tapping. Specimens that were contaminated by common flora, such as bronchial washings by bronchoscopy and endotracheal secretion.
Pulmonary and extrapulmonary samples were subjected to the following processing. Smear preparation was carried out in a biosafety cabinet. Each sample was placed on two glass slides; after it had been smeared and dried, it was heat-fixed by slowly moving the slide over the flame. Acid-fast bacteria were detected in each of these smears by using ZN and Auramine O stain microscopy. The grading of the bacteriological stains was provided by NTEP guidelines after being evaluated under an oil immersion and fluorescence microscope.12 In addition, Truenat testing was used for every sample to accurately detect M. tuberculosis along with rifampicin resistance. By using all three procedures, a total of 229 non-repetitive samples that met the above criteria were examined.
Ziehl-Neelsen
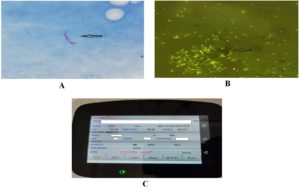
The primary stain used on the heat-fixed smears was 0.1% carbol fuchsin. The smeared slide was heated for five to seven minutes at frequent intervals without allowing it to dry, and then was washed. Subsequently, it was treated with a 25% sulphuric acid solution to decolourise it until it lost its colour for two to three minutes, after which it was washed. The last stage was counterstaining with 0.1% Loffler’s methylene blue for 30 seconds, followed by tap water washing, drying, and examining under a microscope with an objective lens. Mycobacterium tuberculosis bacilli appeared as pink, long, slender beads when stained with the ZN method in a blue background, as shown in Figure 1A.10
Fluorescent stain (Auramine O)
The freshly made Auramine O was used to stain the heat-fixed smear for five to seven minutes. Following that, it was thoroughly cleaned with tap water, and then 0.5% acid alcohol was used as a decolourizer for two minutes. It was again washed to get rid of the acid. The slide was stained again with 0.1% potassium permanganate as a counterstain for two minutes, then cleaned, allowed to dry, and examined with a 40x lens of an LED microscope to look for tubercle bacilli, which were visible as brilliant yellow on a dark background, as seen in Figure 1B.10
Figure 1. Identification of Mycobacterium tuberculosis by smear microscopy and molecular method. (A: Acid-fast bacilli in Ziehl-Neelsen stain; B: Tubercle bacilli in fluorescent stain; C: Tuberculosis detected in Truenat)
Truenat
A 0.5 ml sample was mixed with the liquefaction buffer of two drops for five minutes at room temperature and left to liquefy. Using the transfer pipette, 0.5 mL of the above specimen was then transferred to a lysis buffer container, and the liquefaction buffer was added further. The cap was fixed tightly and mixed thoroughly. The lysis buffer container was then incubated at room temperature for three minutes. To extract MTB DNA using Trueprep® AUTO v2, the full contents of the lysis buffer container were eventually shifted to the cartridge’s sample chamber by utilising a transfer pipette. The Truenat MTB target gene was ribonucleoside diphosphate reductase large subunit. The deoxyribonucleic acid extracts were loaded into the TruenatTM MTB chip and then into the Truelab® Quattro Real Time Quantitative micro PCR analyzer by Molbio Diagnostics. These findings were evaluated as MTB-detected, as seen in Figure 1C, MTB-not-detected, or indeterminate. An additional quantity of the same extract of DNA was added to the rifampicin (RIF-MTB) chip in case the TB test yielded positive results for identifying drug resistance. Truenat software version for reproducibility was 2.10.839.PCR. This chip-based method has optical and thermal calibration conducted once year with a cutoff range of 3.39 to 3.41. Preventive maintenance for Truelab® Quattro and Trueprep® AUTO v2 is carried out every three months by evaluating their functional efficiency using blank extraction, positive control, negative control along with known samples. Every sample is accompanied by internal quality control that are linked to the cartridge. A graph that shows amplification in positive controls and a flat line in negative controls and targets indicates its validity.
Statistical analysis
The data was recorded in a master chart using Microsoft Excel 2021 (Microsoft Corporation, Redmond, WA). SPSS software version 20.0 (IBM Corp., Armonk, NY) was used to statistically analyse the data. Frequencies and percentages were calculated for qualitative data and tabulated. The sensitivity, specificity, positive predictive value, negative predictive value, and diagnostic accuracy results for the detection of Mycobacterium tuberculosis were calculated by smear microscopy with Truenat and were compared with each other. A chi-square analysis was calculated for categorical values. A p-value of less than 0.05 was considered significant.
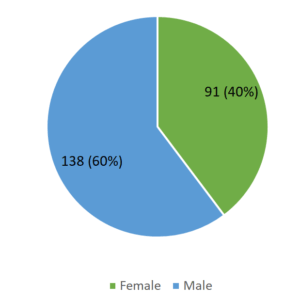
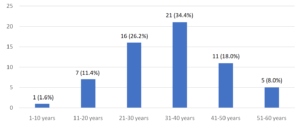
Our study involved 229 specimens, which were obtained from patients suspected of having TB; 138 were males and 91 were females, as seen in Figure 2. Truenat reported 61 positive cases overall. Among which the majority of them were in the age group of 31-40 years, i.e., 21 cases (34.4%), followed by 16 patients (26.2%) between 21-30 years, and 11 cases (18.0%) within 41-50 years, as indicated in Figure 3. According to the samples that were received in our central laboratory, the most commonly obtained were sputum samples (180), followed by endotracheal secretions (29), while the least commonly collected were pleural fluid (14) and bronchoalveolar lavage specimens (6). Every sample was subjected to all three techniques. The cases identified to be positive for ZN and FM stain were 51 and 55, whereas 61 cases were detected by Truenat. The Ziehl-Neelsen and fluorescent stains had 23.3% and 24.0% positive smears for MTB. In contrast, Truenat had the highest positive results at 26.6%. Among the 61 positive cases of Truenat, ZN, and FM stain, there were 49 and 52 MTB cases, classified as true positives; however, out of 168 patients who tested negative with Truenat, 166 and 165 cases were negative by Ziehl-Neelsen and fluorescent stain and were considered to be true negatives. When taking into consideration the overall MTB cases that were positive and negative by Ziehl-Neelsen and fluorescent stain, 2 cases were falsely found to be positive and 12 cases that were found to be falsely negative by Ziehl-Neelsen stain; similarly, fluorescent stain had 3 cases that were falsely positive and 9 cases that were falsely negative. Table 1 shows the sensitivity, specificity, diagnostic accuracy, positive predictive value, and negative predictive value of Ziehl-Neelsen and fluorescent stain that were analysed using Truenat as a standard. The P-value came out to be <0.001 by the Chi-Square Test, which was significant. Among the total positives in 61 Truenat cases, only one sample was found to be rifampicin-resistant. Since the false positive cases from smear microscopy did not satisfy our study’s objective, they were not subjected to further screening for non-tuberculosis bacteria.
Table (1):
Evaluation of smear microscopy against Truenat
Statistics |
ZN stain vs. Truenat |
Fluorescent stain vs. Truenat |
|---|---|---|
Sensitivity |
80.3% |
85.2% |
Specificity |
98.8% |
98.2% |
Positive predictive value |
96.1% |
94.5% |
Negative predictive value |
93.3% |
94.8% |
Disease prevalence |
22.3% |
24.0% |
Diagnostic accuracy |
93.9% |
94.6% |
Chi-square value (χ2) |
164.25 |
173.44 |
P-value |
less than 0.001 |
Our nation possesses an extended tradition of TB investigation along with its development initiatives. The existence of tuberculosis bacteria in the specimen is regarded as an indication of the infective age. Even though there are numerous highly advanced techniques for identifying tuberculosis bacteria and treating the disease, it remains a serious public health issue with unfavourable social and economic outcomes throughout the country. As a result, the laboratory diagnosis is essential in identifying TB cases.1
Our study showed that men (60.3%) were significantly higher when compared to females (39.7%) who gave their sample specimens for testing for MTB. Neelu et al. study indicated a prevalence higher in male (52.3%) than females (47.7%), and the study by Sanjay et al. also showed a similar prevalence of 65.16% in males and 34.83% in females.13,14 Patients between 21 and 45 years of age were identified as being the most infected with tuberculosis in the present study. The report was consistent with other studies, which showed 21-40 years of age as the most affected.15,16 This finding supports the notion that the prevalence of MTB is more common among the working population. Men are found to be more likely to develop tuberculosis than women because of their unhealthy habits, such as drinking alcohol or smoking. Female patients are fewer due to factors such as dependence, low education, and ignorance in underdeveloped countries.
Among the 229 samples analysed in this study, ZN stain and Auramine O (AO) stain yielded overall positive results for 51 and 55 patients, with smear positive rates of 22.3% and 24.0%, respectively. This was compared to various data obtained in different studies, which indicated that FM had a higher percentage than ZN stain.17-20 The sensitivity and specificity of FM were higher (85.2% and 98.2%) than ZN stain (80.3% and 98.8%) in our study, which was similar to research conducted by Dzodanu et al., Makeshkumar et al., and Bhansal et al.21-23
Diagnostic accuracy and disease prevalence of FM staining are 94.6% and 24.0%, respectively; ZN stain is 93.9% and 22.3%, denoting that AO stain has greater significance in terms of identification of the infection. This was demonstrated by a P-value of <0.005 being significant. Based on these findings, smears stained with auramine are a more sensitive way to demonstrate AFB in specimens than samples stained with ZN. The diagnostic value of fluorescent stain is significantly higher when compared with Ziehl-Neelsen’s stain, particularly in cases where there is an inadequate bacillary count in the samples. Thus, the Auramine O stain technique is superior and can be suggested for labs that handle a huge quantity of samples, since it is time-efficient.24
However, there are several disadvantages of fluorescent microscopy, such as low specificity, impurities in auramine, artefacts that create fluorescence, and incorrect smear diagnosis leading to false positives. Technical errors such as inadequate slide preparation, improper staining methods, and inaccurate observations may be the cause of the false negative cases. Its limitations in detecting rifampicin resistance are its main weakness. A molecular method like Truenat aids in the identification of pathogens, along with drug resistance. This will help to prevent unnecessary medical intervention, adverse effects from medication, and stress for the patient and their family.
According to research by Padmaja et al., Kumar et al., and Taddese et al., using the molecular technique resulted in a higher number of positive cases than using the standard microscopic method, which had a comparatively small number of cases.25-27 This is comparable to our study, which yielded 61 positive cases in Truenat overall. Rifampicin resistance was identified in only one sample out of 61 positive Mycobacterium tuberculosis cases by Truenat, thus resulting in positivity, which is less than the rates found in studies by Arora et al.28 The variation may be explained by differences in the mutations that cause drug resistance in various geographic areas.
Molecular techniques have certain setbacks, though they are recognised as highly effective tests for tuberculosis. Such include test failures brought on by variations in targeted genes, DNA disturbances, and reliance on bacillary amounts. Despite these drawbacks, Truenat is a better method compared to conventional microscopic methods for TB diagnosis. It produces clear results and has high sensitivity. Among the microscopic methods, fluorescence staining is the better technique, as represented in Table 2. The lack of continuous patient monitoring limited the provision of clinical follow-up. Furthermore, the study sample came from a single healthcare facility, which limited the applicability of the results to the whole population. To establish conclusive findings, more research with diverse populations from various regions of the nation is required.
Table (2):
Merits and demerits of ZN stain, fluorescent stain, and Truenat29
Techniques |
Merits |
Demerits |
|---|---|---|
Ziehl-Neelsen stain (method) |
It has a low cost and can analyse more samples. |
Ideally, 105 bacteria in each millilitre of salivary specimen are necessary for detection, and their accuracy varies in terms of detection. |
Fluorescent stain (microscopy) |
Excellent for high-load settings (>25 smears per day), less time-consuming, and slides can be screened at a lower magnification than ZN microscopy. |
Expensive in comparison to ZN staining, there is a need for a dark room, the health risk of UV light exposure, and the mercury lamp lighting systems have a short life span. |
Truenat |
There is limited instruction for laboratory workers on how to understand a drug’s sensitivity and detect TB, along with drug resistance. |
Annual calibration is mandatory. |
Patients suspected to have MTB based on symptoms or radiological findings had their sample specimens examined by smear microscopy staining. The conventional method of investigation carried out by ZN staining was inadequate to diagnose many of the suspected cases who failed to receive treatment. Overall, our data demonstrated that fluorescent microscopy outperforms the ZN technique in the detection of bacteria. In recent days, molecular techniques like PCR have become even more precise and sensitive than microscopic techniques. Employing Truenat in our study, smear-negative cases were also detected, along with rifampicin resistance in a resource-limited setting. This paved the way for early diagnosis, accurate detection, and the right medication choice. As a result, it led to not excluding any suspected case of infection from effective treatment, control of the infection, and prevention of its further spread in society. Thereby reducing morbidity and mortality. Considering the advantages of Truenat, as it is easily portable, battery operated and requires little infrastructure, it can be implemented practically in areas with limited resources, and thus it can take the role of conventional microscopy techniques.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
This study was approved by the Institutional Ethics Committee (Human Studies), Melmaruvathur Adhiparasakthi Institute of Medical Sciences and Research, Chennai (Reg. No. ECR/1487/Inst/TN/2020) (MAPIMS/IEC/52/2023).
INFORMED CONSENT
Written informed consent was obtained from the participants before enrolling in the study.
- Gilmour B, Alene KA. Ending tuberculosis: Challenges and opportunities. Front Tuberc. 2024;2:1487518.
Crosseref - Shrivas A, Singh S. Tuberculosis Diagnosis and Management: Recent Advances. J Glob Infect Dis. 2025;17(1):3-9.
Crossref - Derseh NM, Agimas MC, Aragaw FM, et al. Incidence rate of mortality and its predictors among tuberculosis and human immunodeficiency virus coinfected patients on antiretroviral therapy in Ethiopia: systematic review and meta-analysis. Front Med (Lausanne). 2024;11:1333525.
Crossref - Tamura G, Llano G, Aristizabal A, Valencia J, Sua L, Fernandez L. Machine-learning methods for detecting tuberculosis in Ziehl-Neelsen stained slides: A systematic literature review. Intell Syst Appl. 2024;22:200365.
Crossref - Masali HT, Takpere A, Shahapur P. A comparative Study of Ziehl-Neelsen Stain and Fluorescent Stain Microscopy in the Diagnosis of Pulmonary Tuberculosis. J Pure Appl Microbiol. 2021;15(4):2027-2033.
Crossref - Gelalcha AG, Kebede A, Mamo H. Light-emitting diode fluorescent microscopy and Xpert MTB/RIF® assay for diagnosis of pulmonary tuberculosis among patients attending Ambo hospital, west-central Ethiopia. BMC Infect Dis. 2017;17(1):613.
Crossref - Workineh M, Maru M, Seman I, et al. Agreement between Direct Fluorescent Microscopy and Ziehl-Neelsen Concentration Techniques in Detection of Pulmonary Tuberculosis in Northwest Ethiopia. Ethiop J Health Sci. 2017;27(5):459-464.
Crossref - Acharya R, Vyas M. Comparative Study between FM Staining and ZN Staining in Diagnosing Sputum Smear Positive PTB at S.P. Medical College and Hospital. Int J Med Res Prof. 2018; 4(4):134-37.
Crossref - Nandagopal B, Sankar S, Lingesan K, Appu KC, Sridharan G, Gopinathan AK. Evaluation of a nested PCR targeting IS6110 of Mycobacterium tuberculosis for detection of the organism in the leukocyte fraction of blood samples. Indian J Med Microbiol. 2010;28(3):227-232.
Crossref - Birhman N, Payal N, Khandait M, Bhardwaj M. Comparative study on conventional diagnostic methods with GeneXpert and TrueNat Mycobacterium tuberculosis (MTB) assay of tuberculosis disease. MGM J Med Sci. 2024;11(3):508-513.
Crossref - Meaza A, Tesfaye E, Mohamed Z, et al. Diagnostic accuracy of Truenat Tuberculosis and RifampicinResistance assays in Addis Ababa, Ethiopia. PLoS ONE. 16(12).
Crossref - Patel J, Dave P, Satyanarayana S, Kumar AM, Shah A, Ananthakrishnan R, Ratnu A. Pretreatment sputum smear grade and smear positivity during follow-up of TB patients in Ahmedabad, India. Public Health Action. 2013;3(4):308-310.
Crossref - Neelu SP, Terin J, Dutta H, Kalyani M. Comparison of Ziehl-Neelsen, Kinyoun’s and Fluorescent Staining for detection of Mycobacterium tuberculosis in sputum samples before and after petroff’s concentration technique. Asian J Pharm Clin Res. 2018;11(4):110-113.
Crossref - Sanjay MG, Radha PM, Babu NP, Sushant M, Jitesh A. Genotypic diagnosis of extrapulmonary tuberculosis-CBNAAT a novel tool. Med Pulse Int J Med. 2017;4(2):79-82.
Crossref - Jadhav DS, Deshmukh VV, Mahato NH, Valand AG. Comparative Study of Ziehl-Neelsen Stain versus Fluorescent Microscopy in Diagnosis of Tuberculous Lymphadenitis on FNAC at A Tertiary Care Centre. Trop J Path Micro. 2019;5(4):217-222.
Crossref - Lawrence, Debbarma M, Baveja CP, Kumar S, Khanna A, Sapriina J. Comparative evaluation of flourescent staining with ziehl-neelsen and kinyoun staining in the diagnosis of clinically suspected cases of pulmonary tuberculosis. Int J Contemp Med Res. 2016;3(7):1970-1974.
- Raju PV, Reddy CR. To compare the efficacy of fluorescent microscopy versus ZN stain microscopy in detecting the pulmonary tuberculosis. Int J Contemp Med Res. 2020;7(12):L1-L5.
- Githui W, Kitui F, Juma ES, Obwana DO, Mwai J, Kwamanga D. A comparative study on the reliability of the fluorescence microscopy and Ziehl- Neelsen method in the diagnosis of Pulmonary tuberculosis. East Afr Med J. 1993;70(5):263-266.
- Ulukanligil M, Aslan G, Tasci S. A comparative study on the different staining methods and number of specimens for the detection of acid fast bacilli. Mem Inst Oswaldo Cruz. 2000;95(6):855-858.
Crossref - Prashanthi K, Kumari AR. Efficacy of flurochrome stain in the diagnosis of Pulmonary Tuberculosis co- infected with HIV. Indian J Med Microbiol. 2005;23(3);179-185.
Crossref - Dzodanu EG, Afrifa J, Acheampong DO, Dadzie I. Diagnostic Yield of Fluorescence and Ziehl-Neelsen Staining Techniques in the Diagnosis of Pulmonary Tuberculosis: A Comparative Study in a District Health Facility. Tuberc Res Treat. 2019;2019:4091937.
Crossref - Makeshkumar V, Madhavan R, Narayanan S. Prevalence of drug resistance in Mycobacterium tuberculosis in a teaching hospital of Kanchipuram District, Tamil Nadu, South India. Am J Microbiol Res. 2014;2(1):35-40.
- Bansal R, Sharma PK, Jaryal SC, Gupta PK, Kumar D. Comparison of sensitivity and specificity of ZN and fluorescent stain microscopy with culture as gold standard. J Tuberc Res. 2017;5(02):118-128.
Crossref - Tarhan G, Ordulu L, Gumuslu F, Ceyhan I, Cesur S. Comparison of auramine-rhodamine and Erlich-Ziehl-Neelsen staining methods for the diagnosis of tuberculosis. Mikrobiyol Bul. 2003;37(2-3):131-136.
- Padmaja GV, Srujana K, Sadhana C. Comparison of Ziehl-Neelsen’s stain, fluorescent stain with CBNAAT of sputum for the diagnosis of pulmonary tuberculosis. J NTR Univ Health Sci. 2019; 8(4):238-243.
Crossref - Kumar P, Bhardwaj P. Diagnosis of pulmonary tuberculosis with cartridge based nucleic acid amplification test and light emitting diode fluorescent microscopy: a comparative study. Int J Adv Med. 2019;6(5):1580-1583.
- Taddese BD, Desalegn DM, Misganaw AS, et al. Comparison of GeneXpert against Light-Emitting Diode Fluorescent Microscopy for the Diagnosis of Pulmonary Tuberculosis in Addis Ababa, Ethiopia. J Microb Biochem Technol. 2018;10:12-15.
Crossref - Arora D, Jindal N, Bansal R, Arora S. Rapid Detection of Mycobacterium tuberculosis in Sputum Samples by Cepheid Xpert Assay: A Clinical Study. J Clin Diagn Res. 2015;9(5):DC03-DCO5.
Crossref - Forbes BA, Sahm DF, Weissfeld AS, eds. Bailey & Scotts Diagnostic Microbiology 12th edition, Mosby Elsevier. 2007.
© The Author(s) 2026. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.