ISSN: 0973-7510
E-ISSN: 2581-690X
Rotavirus is a significant global health concern, causing severe diarrhea in young children and imposing a substantial economic burden on healthcare systems. To tackle this and improve public health, the development of effective rotavirus vaccines as well as quality control testing of vaccines are crucial. This comprehensive review explores the historical context of rotavirus vaccine development, the various vaccine types and approaches in use today, and the critical selection of vaccine strains. It also delves into the formulation of rotavirus vaccines, the role of epidemiological studies in tracking disease prevalence and transmission patterns, and the essential quality control measures for vaccine production. Analytical methods for quality control, such as antigen characterization, potency assays, identity testing, sterility and safety testing, and stability studies, are discussed. Before being released onto the market, a quality control evaluation should be completed at national control laboratories, as this ensures the vaccine’s safety and quality. The review highlights the challenges associated with rotavirus detection, including sample handling, cross-reactivity, and specificity issues. Additionally, it addresses the impact of rotavirus vaccines on public health and emphasizes the importance of continued surveillance and monitoring as according to the latest WHO and national regulatory guidelines, these vaccines are closely monitored for their safety and effectiveness. The review concludes by outlining current challenges, innovations, and future perspectives in the field of rotavirus vaccination, underscoring the ongoing commitment to combat this pervasive and dangerous pathogen.
Surveillance, Safety, Pathogen, National Control Laboratories
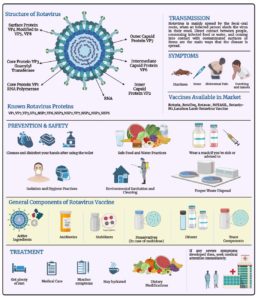
Rotavirus, a member of the Reoviridae family,1 causes a tough challenge on the world healthcare stage.2 This Double-stranded RNA virus, recognized by its wheel-like structure under the electron microscope, is a prominent cause of severe diarrhea, affecting people of all ages worldwide,3-5 as shown in Figure 1. However, it is among the youngest, the infants and young children, who suffer most of its impact, with severe bouts of diarrhea and vomiting that can lead to dehydration and, in some cases, life-threatening complications.6,7 It is a globally encountered virus, with almost every child receiving at least one infection before age.8 This rotavirus leads to nearly about ~125,000-200,000 fatalities each year, and around 50% of all associated fatalities come up in only four countries (India, Nigeria, Pakistan, and the Democratic Republic of the Congo).8-10 Rotavirus has several species, with different strains and subtypes identified such as Group A rotaviruses (RVA) is the most common and life-threatening.11,12 RVA infections show seasonal patterns in multiple regions, mainly increasing during the winter season in temperate climes and maintaining a continuous presence in tropical areas.3
While rotavirus infections have similar clinical symptoms like severe diarrhea, vomiting, fever, and cramps, their effect varies greatly depending on geographic location, healthcare availability, and vaccination status.13,14 The introduction of rotavirus vaccinations in various countries has proven beneficial in lowering the risk of associated disorders.8 Due to its serious clinical effects, particularly on newborns and young children, it is an major concern. Its worldwide prevalence and propensity for severe disease make rotavirus a major target of public health initiatives, including vaccination and diagnostic techniques, to decrease its effect and ensure the health of vulnerable populations8,13,15 Rotavirus detection is challenged by viral diversity, cross-reactivity, low viral load, sample degradation, limited diagnostic capacity in LMICs, and difficulties in distinguishing symptomatic infections from asymptomatic.16,17
To minimize this burden, comprehensive preventative strategies, including vaccination, should be focused on health policy. In India National Immunization Schedule (NIS) for Infants, Children, and Pregnant Women rotavirus vaccination is given at 6 weeks, 10 weeks and 14 weeks with a dose of 5 drops for Infants and children.18,19 Quality control of the rotavirus vaccine critical step for patient safety and the vaccine effectiveness.20 It signifies that each batch of that vaccine follows high regulatory criteria according to Government regulations.21,22 This strict surveillance assures that rotavirus vaccines remain efficacious in preventing severe infections, especially in susceptible groups like newborns and young children.23,24 In this thorough review, our purpose is to give in-depth data on rotavirus, including its structure and categorization to its clinical importance, formulation of rotavirus vaccines, quality control procedures, and QC analytical techniques in brief. We also discussed its epidemiology, vaccine, and control measures as well.
Using PICO (Population/Interventions/Comparisons/Outcomes) search methodology, present study focuses on rotavirus-infection, particularly of population in India and across the world. Rotavirus – vaccine research and implementation, as well as monitoring, preventative, and control measures, were all included in the scope of intervention. The study aimed on reviewing rotavirus incidence, morbidity, and death through comparing different types of vaccines and management strategies. As a narrative review, no formal protocol was registered, transparency was maintained through defined criteria and structured search methods. Multiple electronic databases, including PubMed, Scopus, Web of Science, Google Scholar, MDPI, Medline (OVID), Embase (OVID), and CINAHL (EBSCO Host), were used, with the language limitation set to English. Data search was included with variable time ranging from January 1990 to May 2023. The EndNote-20 reference manager was used to manage references and manage duplication.
We excluded studies that were not peer-reviewed, did not focus on rotavirus, or were published in languages other than English without available translations. This ensured that the review included only reliable, relevant, and accessible sources. The inclusion criteria comprised of research done in India and worldwide that reported on the incidence or fatality rates of rotavirus in all age categories and involved control, preventive, and monitoring measures. Key study characteristics, including author, area, year, study design, degree of evidence, sample size, interventions, outcomes, and major findings, were recorded on a comprehensive data collecting form that was created. A structured table of clinical trials containing NCT is also incorporated (Table 1). No., study title, study status, brief summary, conditions interventions, sponsors and collaborators, sex, age, phases study, and type study design was created by extracting data from ICTRP (International Clinical Trials Registry Platform) and NIH Clinical Trials official database. Levels of the research were assessed separately by coauthors, who resolved any conflicts in review or contact with the corresponding author. To enhance knowledge about vaccination coverage, recommendations on the quality of rotavirus vaccine from different manufacturers were taken including different sources like official papers, publications and from agencies like UNICEF, the World Health Organization (WHO), Indian Ministry of Health and Family Welfare and pharmacopeia’s of different nations taken into consideration
Table (1):
Clinical trials of different rotavirus vaccines with brief discussion
NCT. No. |
Study Title |
Study Status |
Brief Summary |
Conditions |
Interventions |
Sponsors and Collaborators |
Sex |
Age |
Phases |
Study Type |
Study Design |
Ref. |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
NCT01061658 |
Safety and Immunogenicity Study of the Tetravalent Rotavirus Vaccine |
UNKNOWN |
90 healthy Indian newborns participated in a Phase I/II trial which was double-blind, placebo-controlled, and assessed the immunogenicity and safety of the BRV-TV rotavirus vaccine. At 6-8, 10-12, and 14-16 weeks of age, the vaccination will be administered orally in three doses, each followed by 2.0 ml of antacid. |
Rotavirus Gastroenteritis |
BIOLOGICAL: BRV-TV Rotavirus Vaccine and Placebo |
Shantha Biotechnics Limited |
All |
Child |
Phase1, phase2 |
Interventional |
Randomized, parallel |
[38] |
NCT00938327 |
Reactogenicity and Safety of GlaxoSmithKline (GSK) Biologicals’ Rotarix™ (Human Rotavirus Vaccine) in Indian Infants |
COMPLETED |
In accordance with the Indian Prescribing Information, this post-marketing surveillance helped to assess the human rotavirus vaccine’s reactogenicity and safety information when given to healthy newborns. |
Infections, Rotavirus |
BIOLOGICAL: Rotarix™ |
GlaxoSmithKline |
All |
Child |
– |
Observational |
Observational model: | time perspective: p |
[39] |
NCT03474055 |
Study on Liquid Bovine Rotavirus Pentavalent Vaccine (LBRV-PV) to Evaluate Lot-to-Lot Consistency and to Compare Non-Inferiority With ROTASIIL (Lyophilized BRV-PV) in Healthy Infants in India |
COMPLETED |
This study analyses lot-to-lot consistency in LBRV-PV manufacture and compares the lyophilized ROTASIIL with liquid BRV-PV (LBRV-PV) in newborns. Three doses are given, scheduled four weeks apart, beginning at 6 to 8 weeks of age, to demonstrate non-inferiority in producing anti-rotavirus IgA antibodies. |
Rotavirus Gastroenteritis |
BIOLOGICAL: Liquid Rotavirus Vaccine |
Serum Institute of India Pvt. Ltd. |
All |
Child |
Phase2 phase3 |
Interventional |
Allocation: randomized | intervention model: parallel | masking: none | primary purpose: treatment |
[40] |
NCT02133690 |
A Clinical Trial to Study the Effect and Safety of Rotavirus Vaccine Against Severe Rotavirus Gastroenteritis in Healthy Indian Infants |
COMPLETED |
The purpose of this Phase 3 multicenter, randomised, double-blind, placebo-controlled trial was to evaluate the efficacy of the rotavirus vaccination. |
Rotavirus Gastroenteritis |
BIOLOGICAL: Live Attenuated Pentavalent (G1-G2-G3-G4-G9) Human X Bovine Reassortant Rotavirus Vaccine (BRV-PV) | OTHER: Placebo |
Serum Institute of India Pvt. Ltd. | PATH |
All |
Child |
Phase3 |
Interventional |
Allocation: randomized | intervention model: parallel | masking: quadruple (participant, care provider, investigator, outcomes assessor) | primary purpose: prevention |
[41] |
NCT02584816 |
Phase III Study on Rotavirus Vaccine to Evaluate Lot-to-lot Consistency and Interference With Routine UIP Immunization |
COMPLETED |
The aim of this Phase 3 open-label randomised trial is to assess the consistency of the manufacturing process of the bovine rotavirus pentavalent vaccine (BRV-PV) from lot to lot. |
Rotavirus Gastroenteritis |
BIOLOGICAL: BRV-PV (Lot A/B/C) or ROTARIX + DPT-HepB-Hib + OPV. |
Serum Institute of India Pvt. Ltd. | PATH |
All |
Child |
Phase3 |
Interventional |
Allocation: randomized | intervention model: parallel | masking: none | primary purpose: prevention |
[42] |
NCT01508533 |
Epidemiology of Rotavirus Infection in North India Community |
COMPLETED |
The goal of this study was to assist with the planning and execution of the phase III trials for the oral rotavirus vaccine 116E, as well as to do a disease burden study and support site preparation. |
Rotavirus Gastroenteritis |
– |
Society for Applied Studies | National Institute of Immunology, New Delhi | Ministry of Science and Technology, India |
All |
Child |
– |
Observational |
Observational model: | time perspective: p |
[43] |
NCT01305109 |
A Phase III Clinical Trial to Evaluate the Protective Efficacy of Three Doses of Oral Rotavirus Vaccine (ORV) 116E |
UNKNOWN |
A phase III randomised, double-blind, placebo-controlled study, the main goal of which is to compare the effectiveness of a placebo in India with three doses of ORV 116E, 10^5.0 FFU, against severe rotavirus gastroenteritis. |
Viral Gastroenteritis Due to Rotavirus |
Biological: ORV 116E | biological: placebo |
Bharat Biotech International Limited | Ministry of Science and Technology, India | PATH |
All |
Child |
Phase3 |
Interventional |
Allocation: randomized | intervention model: parallel | masking: quadruple (participant, care provider, investigator, outcomes assessor) | primary purpose: prevention |
[44] |
Epidemiological studies
The monitoring for finding the prevalence and rotavirus infection transmission trends is epidemiological research. These studies provide crucial information about the prevalence of related disorders in different age groups and geographical areas.25,26 Researchers can study the effects of vaccination campaigns, identify seasonal variations, and evaluate the efficacy of public health programs by closely monitoring the frequency of rotavirus infections.27 Epidemiological investigations require a fast and exact diagnosis. The use of molecular techniques has completely changed how rotavirus is researched. The classification and identification of rotavirus strains have undergone advancement using RT-PCR and enzyme immunoassays (EIAs). These novel diagnostic techniques enable the identification of specific genotypes, supplying information on the variety of rotaviruses in circulation and their potential impact on vaccine effectiveness. Serological tests have also developed into essential tools for estimating population immunity levels, providing essential data for assessing the effectiveness of vaccination campaigns.28 These advanced diagnostic methods have significantly improved our ability to watch rotavirus outbreaks and trends. Researchers can now quickly respond to new strains or shifts in genotype dominance by quickly detecting the genotypic profile of rotavirus. This improved method of diagnosis provides healthcare administrators with the real-time information they need to make accurate choices about immunization schedules, epidemic control, and budget allocation. Obtained data aids in mapping the transmission patterns of specific genotypes, allowing for the early diagnosis of outbreaks and the evaluation of strain alterations brought on by vaccination. In order to guarantee that vaccine formulations remain effective over time, the ability to recognize new strains early is important.25,28
Rotavirus vaccine development
Rotavirus vaccine development brings huge benefits to community health. For centuries, rotavirus infections led to severe morbidity and death, especially in infants globally.29 The launch of the first vaccine against rotavirus is RotaShield, in the late 1990s, signified a crucial advance, but due to concerns about safety, it was ultimately withdrawn from the market. This historical backdrop highlights the continuous significance of developing and refining rotavirus vaccine development.24,30 Today, multiple rotavirus vaccines are in use internationally, each using various vaccine types and techniques. These include live attenuated vaccines, such as Rotarix31 and RotaTeq,32 which imitate natural illness and give substantial immunity.33 Additionally, subunit vaccines like RotaVac31 and ROTASIIL34 include recombinant proteins to provide an immune responses.
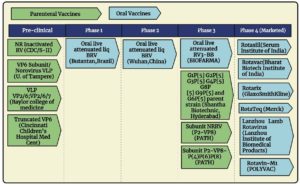
There were various vaccines are in developing stage as mentioned in Figure 2, several oral as well as parenteral vaccines are in different phases of clinical trials. The variety in vaccination types reflects persistent efforts for enhancing safety, effectiveness, and accessibility, particularly in locations with varied healthcare infrastructures. Selection of rotavirus strain is a critical part of vaccine development.35 The selection process of rotavirus strain includes checking the prevalence, pathogenicity, and antigenic characteristics of that specific strain. Multivalent vaccinations typically combine many strains to provide wide protection.34 This selection method is crucial because it affects the vaccine’s efficacy in preventing severe diarrhea due to rotavirus, which is one of the main cause of pediatric death and morbidity globally.30 Over the years, research and development (R&D) efforts resulted to the development of various rotavirus vaccines, each implementing unique ways to provide protection. The most renowned among these are the live-attenuated vaccines, which involve the monovalent Rotarix and the pentavalent RotaTeq vaccine.36 These vaccines are developed to replicate natural rotavirus infections without causing disease, also they trigger immune responses. Another new technique is the formulation of virus-like particle (VLP) vaccines, such as the Rotavac vaccine,37 which includes the structural components of rotavirus as well as API to induce immunity.
Formulation of rotavirus vaccines
The formulation of rotavirus vaccines aims to assure safety, stability, and effectiveness. For the formulation of vaccines, there are some crucial variables such as the selection of antigens, adjuvants, stabilizers, and preservatives as mentioned in Figure 1, the different components of rotavirus vaccine. Selecting the correct antigen is an essential stage in rotavirus vaccine development. There are several known viral proteins and non-structural proteins of rotavirus like VP1, VP2, VP3, VP4, NSP1, VP6, NSP3, NSP2, VP7, NSP4, NSP5, NSP645 and every protein has its function. Any host proteins that form viral particles as well as the by-products of a virus’s DNA are collectively referred to as viral proteins which is also known as VP. The roles of viruses are used to organize their proteins into groups, which consist of regulatory, accessory, nonstructural, and structural proteins. VP1 is the protein that acts through ssRNA binding, the RNA-dependent RNA polymerase, situated at the inner capsid’s five-fold axis, joins forces with VP3 to form a transcription complex. VP2 is the structural protein of the inner capsid, which is necessary for the replicase activity of VP1, and demonstrates non-sequence-specific RNA-binding activity.46 Likewise, protein VP3 has a Guanylyl transferase and methyltransferase are essential component of the virion transcription complex with VP1. Protein VP4 is an outer capsid spike protein, a cell-attachment protein, virulence determinant, haemagglutinin, and neutralization antigen specific to P-type, formed by VP4 trimers. VP4 is modified to form VP5 and VP8. Trypsin cleavage increases the infectiousness of the protein VP4.47 VP6 is a major virion protein which is a middle capsid structural protein that has a homotrimer shape. It is a subgroup antigen necessary for transcription. The last known viral protein in rotavirus is VP7 is the G-type neutralization antigen protein which is RER transmembrane calcium-binding protein, and outer capsid structural glycoprotein affects the immune system,47 as referred in Figure 1.
A protein that a virus generates but that is not an integral part of the viral particle is referred to as a nonstructural protein (NSP) in virology. In rotavirus 6 non-structural proteins were found till date, NSP1 functions with major sequence variation amongst strains, which is also connects the cytoskeleton with the other cytoskeleton. Although it reduces the host’s IFN-α response. The second NSP is NSP2 which binds to NSP5 and VP1, It is necessary for non-specific ssRNA binding, and contains NTPase and helicase, which are involved in viroplasm production and dsRNA synthesis.48 NSP3 is a translational regulator elongation factor elF4G1. It makes virus-like mRNA translation efficient, NSP3 functions as a counterpart of cellular poly(A)-binding protein by acting as a bridge between viral messenger RNA and the cellular translation mechanism. NSP4 glycoprotein also labelled as viral enterotoxin. It acts as receptor for double-layered particles budding across the ER membrane and keeps intracellular calcium levels and RNA replication.47 NSP5 protein binds ssRNA, which is important component of viroplasm which further responsible for viral replication, through hyperphosphorylation, O-linked glycosylation, homomultimer formation, and interaction with NSP2 and NSP6. NSP6 results as gene segment II is the second out-of-frame open-reading frame. It localizes to the viroplasm and participates in interactions with NSP5.49,50
The antigen mostly consists of viral proteins that activate an immune response without producing illness. Commonly utilized antigens include viral proteins like VP651 or the outer capsid proteins VP4, VP752 and VP8.52,53 These antigens are commonly created using recombinant DNA technologies or by producing weakened virus strains in cell cultures. The choice of antigen considerably determines the vaccine’s efficacy and the range of protection it delivers.54 While adjuvants play a vital role in boosting the immunological response to rotavirus vaccinations. By boosting the body’s immune system, adjuvants enhance the vaccine’s efficiency, particularly in conditions when the immunological response may be lower, such as in Infants. Antimicrobial agents like thimerosal, 2-PE and aluminum adjuvant (AH) are extensively used adjuvants in rotavirus vaccinations, however continuing research examines other adjuvants to better enhance immune responses.55 Rotavirus vaccine formulation must take stabilization into consideration. These vaccines need to keep their effectiveness and safety throughout their shelf life, even under different storage conditions. Carefully chosen stabilizers, such as sugars like sucrose, and trehalose, help to protect the vaccine’s integrity and retain its effectiveness. Additionally, lyophilization (freeze-drying) is a typical process used to increase the stability of rotavirus vaccines.56 Formulation of vaccine is a most important task that needs a fine balance between antigen selection, adjuvant inclusion, stability, and preservative concerns. By addressing these essential aspects, vaccine manufacturers can ensure that these vaccines are not only safe and stable but also highly successful in avoiding rotavirus-associated diseases, especially in susceptible pediatric groups.57
Marketed products
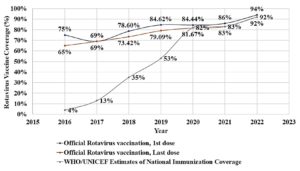
WHO-approved rotavirus vaccines highlight the need for further research and affordable options to ensure broader disease coverage. Statistical evaluation by WHO of rotavirus vaccination coverage in India as mentioned in Figure 3. The official WHO/UNICEF data of National immunization coverage was 4% in 2016 and in year 2022 it increased around 92%. Also, the first and last dose of rotavirus vaccination in 2022 was 94% and 92% respectively. Rotavac and ROTASIIL are only now routinely used in India and some other countries, but they will soon be available for use throughout the world (Table 2). As a result, the safety and effectiveness information from randomised controlled trials will be the main emphasis of this section. Rotavac is a live, monovalent G9P oral rotavirus vaccination that is naturally attenuated. In three sites in India, Nearly 6500 infants participated in a randomised, double-blind, placebo-controlled experiment to evaluate the safety and efficacy of this vaccination, with target schedule (6-7), at lowest 10, and at maximum 14 weeks of age, infants were randomised to receive either a placebo or the vaccine. They were then monitored for adverse events and gastroenteritis for up to two years of age. The usual immunisations were administered to the infants on the planned day (i.e., Same day of research vaccination). In the first year after available in market, Rotavac was expected to be 56% (95% CI: 37%-70%) effective against severe rotavirus gastroenteritis that required hospitalization or supervised rehydration.
Table (2):
Characteristics of Some Marketed Rotavirus Vaccine60-63
Vaccine |
Rotavac |
ROTASIIL |
Rotateq |
Rotarix |
Rotasure |
Rotavin-M1 |
|---|---|---|---|---|---|---|
Manufacturer |
Bharat Biologicals |
Serum Institute of India |
Merck |
GSK |
Abbott |
POLYVAC |
Status |
Worldwide Lic. |
Worldwide Lic. |
Worldwide Lic. |
Worldwide Lic. |
– |
Restricted Lic. |
Origin and Strain |
Single, attenuated human rotavirus strain (G9P[11]) |
5 human-bovine (UK) reassortant rotaviruses (G1, G2, G3, G4, G9) |
Bovine-human reassortant strain, pentavalent Bovine-human G1, G2, G3, G4, P1A(8) |
Human rotavirus strain, monovalent; Human G1 P(8) |
Vero Cell Derived |
Single, attenuated human rotavirus strain (G1P[8]) |
Valent |
1 |
5 |
5 |
1 |
– |
1 |
Route of administration |
Oral |
Oral |
Oral |
Oral |
Oral |
Oral |
Manufacturer Dose schedule |
Bharat Biotech 3-dose (with DTP 1, 2, and 3) |
Serum Institute of India 3-dose (with DTP 1, 2, and 3) |
1st dose: age 6-12 weeks; 2nd-3rd dose: 4-10 weeks interval; Full course must be completed by age 32 weeks |
1st dose: from age 6 weeks; 2nd dose: at least 4 weeks interval; Full course must be completed by age 24 weeks |
3-dose regimen, 4 weeks apart, beginning at 6 weeks of age. |
Polyvac; Center for Research and Production of Vaccines 2-dose (first dose from 6 weeks, second dose from 1-2 months; should be given before 6 months of age) |
Dose volume |
0.5 ml |
2.5 ml |
2 ml |
1.5 ml |
0.5 ml |
2.0 ml |
Doses per container |
5, 10 doses |
1, 2 doses |
3 doses |
2 doses |
Single dose |
Single dose |
Presentation |
Liquid in glass vial |
Lyophilized in glass vial; antacid diluent from separate vial used to reconstitute |
Liquid Single dose glass vial |
1-dose tube; 1-dose applicator; 5 x 1-dose tubes in Liquid form in vial |
Liquid vaccine in glass vial |
Liquid vaccine in glass vial |
Storage and Shelf Life |
Refrigerated (2-8 °C): 6 months Frozen (-20 °C): 5 y; Can be frozen-thawed 6 times without losing potency |
Refrigerated (2-8 °C): 30 months Diluent – Ambient temperature of refrigerated (2-8 °C): 60 months |
Refrigerated 2-8° C for 2years |
Refrigerated +2-8 ° C for 3 years |
N/a |
Frozen (-20-5 °C) |
Volume per course |
1-dose: 45 cm 3 5-dose:12.6 cmi 3 10-dose: 9.6 cmi³ |
1-dose: 53 cm 3 2-dose: 32 cm³ |
45.9 cmâ³/dose |
1-dose tube-34.2 cm3, 1-dose applicator- 170.6 cm³, 5 x 1-dose tubes-23.6 cm³ |
N/a |
N/a |
Vaccine vial monitor (VVM10) |
Yes (2,7) |
Yes (7,30,250+) |
N/a |
Yes (7) |
N/a |
No |
UNICEF price per course |
5- and 10-dose vials: 2.55 USD |
1-dose vial: 4.65 USD; 2- dose vial: 2.85 USD |
1 dose: 3.2 USD |
1 dose 2.19 USD |
N/a |
17.60 USD |
Co-administration |
N/a |
N/a |
DTP, hepb, Hib, IPV, PCV-7 |
DTP, hepb, Hib, IPV, PCV-7, menc |
N/a |
N/a |
Figure 3. Coverage of Rotavirus Vaccination in India from 2016-2022 as per WHO.
(Rotavirus vaccination coverage (Source: www.who.int))
ROTASIIL vaccine by Serum Institute of India have special qualities that, its a pentavalent live attenuated oral vaccine reassorting from bovine to humans, which is thermally stable during its lyophilized form, remaining stable at 2-8 °C for up to 18 months. This immunization was evaluated in double-blind, two randomised, placebo-controlled trials: one in Niger and one in India. In each research, infants were randomised at 6, 10, and 14 weeks of age to receive either a placebo or a vaccine. This was done together with any additional immunisations that were advised by the Expanded Program on Immunization (EPI) schedule. In India, Rotavac & ROTASIIL launch was based on phases. Currently, in 20 states, the Expanded Program on Immunization (EPI) schedule now includes Rotavac as of 2019. The government’s Ministry of Health and Family Welfare has acquired over 50 million doses already. Also, there are ongoing post-marketing studies to assess the vaccines’ efficacy and safety in relation to intussusception. Another vaccines like RotarixTM and RotaTeqTM are also shown effectiveness against rotavirus induced gastroenteritis and significantly decrease acute hospitalization related with RV, according to data from ongoing clinical trials and surveillance systems. The approved RV vaccines also have several issues also, including RotaShieldTM induced intussusception, which reduced parents’ acceptance of RV vaccinations and delayed RV research advancement.58,59
Quality control for rotavirus vaccines
QC involves inspection for quality while manufacturing as well as for release of product, making sure that vaccines follow strict safety, potency, and consistency standards before they are available to the general population. Any variation in the formulation of vaccines could have adverse effects, so vaccines that are given to large and vulnerable populations, particularly newborns and children have strict regulations as per the regulatory authorities of different nations. Rotavirus vaccine development uses various approaches to quality control. Starting points like the vaccination strains and the raw materials used in manufacture are selected carefully. To make sure they comply with predetermined standards, these items go through a detailed evaluation. To reduce the risk of contamination during production, the manufacturing process itself goes through inspection and compliance with Good Manufacturing Practices (GMP). It includes a complete inspection of the vaccine batches and finished product. The potency, purity, and consistency of each batch are evaluated. This includes both in vitro and in vivo tests to make sure it produces a proper immune response while maintaining safety criteria. As part of the quality control procedure, a number of tests are carried out on rotavirus vaccines to ensure their safety and effectiveness. These tests cover a range of vaccine-related topics, from the earliest phases of manufacture to public distribution. Common tests include identification checks to guarantee the strain of the vaccination is real, sterility checks to ensure no contaminants are present, and potency tests to determine the immunogenicity of the vaccine (Table 3).64
Table (3):
Rotavirus test available for final product as per different pharmacopoeia and WHO TRS
| Sr. No. | Test Name | IP | BP | EP | WHO |
|---|---|---|---|---|---|
| 1. | Visual Inspection | Yes | N/M* | N/M* | Yes |
| 2. | Sterility | Yes | Yes | Yes | Yes |
| 3. | Identification | Yes | Yes | Yes | Yes |
| 4. | Appearance | Yes | N/M* | N/M* | Yes |
| 5. | pH | Yes | N/M* | N/M* | Yes |
| 6. | Water/ Residual Moisture | Yes | Yes | Yes | Yes |
| 7. | Virus Concentration for assay based on | ||||
| I. Visualization of infected areas of cell monolayer. OR | Yes | Yes | Yes | Yes | |
| II. Comparison of capacity of vaccine to produce viral RNA. OR | Yes | Yes | Yes | Yes | |
| III. PCR | N/M* | N/M* | N/M* | Yes | |
| 8. | Thermal Stability/ Accelerated Stability | Yes | Yes | Yes | Yes |
| 9. | Labelling | Yes | Yes | Yes | Yes |
N/M*-Not Mentioned, Yes- Mentioned
These standard procedures provide specific attention to viral protein quantitation and genetic stability. Molecular methods like PCR are used to confirm the genetic stability of the vaccination strains, while enzyme-linked immunosorbent assays (ELISAs)65 are used to quantify important rotavirus antigens. Additionally, experiments on animals are carried out to assess the vaccine’s immunogenicity and safety in a living system. The granting of a Certificate of Analysis (CoA) for each vaccination batch is the most significant of these tests and evaluations. The CoA offers a thorough description of the quality control data, attesting to the fact that the vaccination satisfies all established standards for safety, potency, purity, and others. An entire set of quality control tests must be passed before a batch of vaccines is accepted for distribution and use.66 As per the requirements of Indian, European, and British pharmacopeias as well as WHO Technical Report Series (WHO TRS). Testing the vaccine’s potency, determining its composition, and making sure contaminants are absent are examples of quality control procedures. In the absence of strict quality control, vaccinations could be produced that are either unsafe or may not offer enough protection.
Antigen identification and quantification
In rotavirus vaccines, PCR-based methods are essential for antigen characterization and quantification. Comprehending the genetic composition of the virus is essential for the creation of a vaccine. The vaccine’s viral components may be precisely identified and quantified using PCR-based techniques, guaranteeing uniformity from batch to batch.16,67,68 In general, PCR (polymerase chain reaction) and ICT (immunochromatographic test) are both used to detect rotavirus. The rotavirus specificity and sensitivity values for the ICT were 100% and 74.5%, respectively.69 So overall compared to molecular techniques, ICT is less sensitive. Specifically, lower viral concentrations are unable to detect so it might be the cause of negative ICT findings.
Potency assays
Potency assays are essential for assessing the rotavirus vaccine’s ability to boost recipients’ immune systems. In these assays, the vaccine’s capacity to provide protection against an infection by rotavirus is frequently evaluated. Accurate efficiency tests guarantee that the vaccination will offer the anticipated level of immunity in real-life situations.70,71 As of right now, not well recognised in vitro or in vivo models available that permit to correlate of field efficacy with an immune response against severe gastroenteritis caused by rotavirus. The lack of a well-established model to evaluate vaccination efficacy becomes an important issue for determining vaccine potency and long-term stability as an entirely novel class of non-replicating rotavirus vaccines is developed. Due to the drawbacks of conventional techniques in detecting the potency of the multivalent rotavirus vaccine, a cell-based quantification RT-qPCR test (C-QPA) was developed, and its specificity, accuracy, and precision were verified.17 Using standards and samples of monovalent and trivalent rotavirus, the range of linear values and use in practice under various situations were studied to confirm the method’s adaptability in actual titer detection.17,70
Sterility and safety testing
To avoid contamination and guarantee the safety of vaccines, sterility and safety testing are essential. In these tests, harmful microbes are examined for, and it is verified that the vaccine has no trace of impurities that may harm patients.72 A satisfactory result indicates that no contaminating viable microorganisms have been found in the sample examined under the test conditions. Compliance with the sterility test alone cannot provide absolute assurance of freedom from microbial contamination. Greater assurance of sterility must come from reliable manufacturing procedures and compliance with good manufacturing practices. The test must be carried out under aseptic conditions designed to avoid accidental contamination of the product during testing. A grade laminar air-flow cabinet or an isolator is recommended for achieving these conditions. The test environment must be adapted to the way the tests are performed, and the working conditions should be monitored regularly. The test is designed to reveal the presence of microorganisms in the samples used in the test, and interpretation of the results assumes that all units of an article, the entire bulk product, or the contents of every container of the filled product in a lot or batch would have given the same results. Therefore, enough samples of units or containers should be examined to give a suitable result of the tests.73,74
Stability studies
Determining the shelf life and long-term efficacy of rotavirus vaccinations requires stability studies. Because vaccines might lose their effectiveness over time, stability tests are carried out to ascertain the product’s shelf life and ideal storage timings. For testing as per IP, as per requirements have to put three or more of the final lot’s containers at a higher temperature for a specified amount of time, according to the local national regulatory authority’s allowed parameters for that specific product. Then the determination of the viral concentration for both heated vaccine with the vaccine kept at the storage temperature suggested by the assay must done parallelly. During the exposure period, if the virus concentration in the heated containers does not drop below a predetermined threshold, then the stability condition passes. In the case of a multivalent vaccine, the loss of the virus can be determined from the overall virus concentration if there is no noticeable difference in the loss between serotypes.34,55,75
Vaccination and management techniques
Vaccinations have significantly improved public health by reducing the prevalence of rotavirus-related illnesses.76 Hospitalizations due to rotavirus-related diarrhea have significantly decreased after vaccines were introduced, according to multiple studies. Results shows that how successful these vaccinations are in the real world. Regional variations effects of vaccinations can differ from place to place and can be impacted by things like vaccination rates and the frequency of rotavirus strains in circulation. These results highlight how crucial rotavirus immunization is for both lowering healthcare expenses and saving lives.76,77 Ongoing Monitoring and Surveillance of rotavirus vaccines have shown great efficacy, ongoing monitoring and surveillance are necessary to track prevalence and vaccine effectiveness. The most significant issues to consider are adaptation and improvement and vaccine coverage. Constant surveillance aids in the detection of new rotavirus strains, which may require vaccine formulation modifications to preserve efficacy.
Difficulties, breakthroughs, and opportunities for the future
As rotaviruses continue to change, new strains start to appear. It is a continuous challenge to monitor and modify vaccinations to cover these new strains. To counter these new strains continuous research is being done to create vaccines that are more economical and effective, to enhance vaccine delivery strategies, and to expand vaccination coverage to underprivileged populations. The research and development of vaccines has grown significantly over the past 30 years. While numerous RV vaccine candidates are being developed, there are currently a few licensed RV vaccines are available in market. This is a safety concern as it can lead to the reassorting of novel or rare RV strains, potentially causing passive transmission in healthy children. Research on the time required for virus shedding and its impact on unvaccinated newborns’ risk of infection is needed. The effectiveness of RV vaccinations is generally good everywhere, but their efficacy is less in environments with limited resources, possibly due to increased rates of illness and fatality. Available RV vaccines have no significant impact on the safety profiles or protective immune responses of other vaccinations when administered together, highlighting the importance of keeping OPV and RV vaccinations separate in EPI schedules.25 Next-generation vaccines are being developed, such as a trivalent P24-VP8* nanoparticle vaccine, a novel nonreplicating RVV (NRRV), and a fully inactive RVV (IRV) strain (CDC-9) in the gnotobiotic piglet model. These vaccines aim to address the safety concerns and potential risks associated with RV vaccinations, particularly in underdeveloped areas with limited resources and financial problems.78 Besides immunization, encouraging cleanliness and hygiene behaviors might also help lower the rotavirus load. Future rotavirus control will depend on addressing these issues and embracing advancements in vaccination science, surveillance, and public health strategies.76,79
Overall, the review shows that the prevalence of rotavirus related diarrhea is worldwide. Recent studies suggest that the public, healthcare professionals, and policymakers are not yet fully aware about rotavirus disease. Additional efforts are required from the respective governments of the nation’s especially the developing and underdeveloped ones, to spread the information about the vaccinations against rotavirus leading to enhanced protection against the disease. Considering the remarkable outcomes of introducing rotavirus vaccines in developed regions, major investments in materials and human resources is still needed for future research and distribution of rotavirus vaccines and it should be stable and reasonably priced. In the coming years, the research and development of a globally effective rotavirus vaccines will be promising for lowering the morbidity and mortality of rotavirus associated gastroenteritis, the impact of the disease, and serious illnesses in children under the age of five. Enhancement of public knowledge of the infection due to rotavirus and promoting vaccination are essential components of preventive measures that focus on lowering the incidence rate.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
Not Applicable.
- Parashar UD, Bresee JS, Gentsch JR, Glass RI. Rotavirus. Emerg Infect Dis. 1998;4(4):561.
Crossref - Chen J, Grow S, Iturriza-Gomara M, Hausdorff WP, Fix A, Kirkwood CD. The challenges and opportunities of next-generation rotavirus vaccines: summary of an expert meeting with vaccine developers. Viruses. 2022;14(11):2565.
Crossref - Crawford SE, Ramani S, Tate JE, et al. Rotavirus infection. Nat Rev Dis Primers. 2017;3(1):1-16.
Crossref - Estes MK, Cohen J. Rotavirus gene structure and function. Microbiol Rev. 1989;53(4):410-449.
Crossref - Chamera S, Wycisk K, Czarnocki-Cieciura M, Nowotny M. Cryo-EM structure of rotavirus B NSP2 reveals its unique tertiary architecture. J Virol. 2024;98(3):e01660-23.
Crossref - Esona MD, Gautam R. Rotavirus. Clin Lab Med. 2015;35(2):363-391.
Crossref - Parashar UD, Burton A, Lanata C, et al. Global mortality associated with rotavirus disease among children in 2004. J Infect Dis. 2009;200(Suppl 1):S9-S15.
Crossref - Varghese T, Kang G, Steele AD. Understanding rotavirus vaccine efficacy and effectiveness in countries with high child mortality. Vaccines. 2022;10(3):346.
Crossref - Du Y, Chen C, Zhang X, et al. Global burden and trends of rotavirus infection-associated deaths from 1990 to 2019: an observational trend study. Virol J. 2022;19(1):166.
Crossref - Clark A, Mahmud S, Debellut F, et al. Estimating the global impact of rotavirus vaccines on child mortality. Int J Infect Dis. 2023;137:90-97.
Crossref - Gupta S, Chaudhary S, Bubber P, Ray P. Epidemiology and genetic diversity of group A rotavirus in acute diarrhea patients in pre-vaccination era in Himachal Pradesh, India. Vaccine. 2019;37(36):5350-5356.
Crossref - Carossino M, Vissani MA, Barrandeguy ME, Balasuriya UBR, Parreno V. Equine rotavirus A under the one health lens: potential impacts on public health. Viruses. 2024;16(1):130.
Crossref - Burke RM, Tate JE, Kirkwood CD, Steele AD, Parashar UD. Current and new rotavirus vaccines. Curr Opin Infect Dis. 2019;32(5):435.
Crossref - Hellysaz A, Hagbom M. Understanding the central nervous system symptoms of rotavirus: a qualitative review. Viruses. 2021;13(4):658.
Crossref - Cunliffe NA, Bresee JS, Gentsch JR, Glass RI, Hart CA. The expanding diversity of rotaviruses. Lancet. 2002;359(9307):640-642.
Crossref - Wang Y, Liu Y, Wang M, et al. Establishment of a cell based quantitative reverse transcription polymerase chain reaction (RT qPCR) assay for detection of multivalent rotavirus vaccine. J Med Virol. 2020;92(12):3157-3164.
Crossref - Wang Y, Liu Y, Bao H, et al. Application of the cell based RT qPCR assay (C QPA) for potency detection of the novel trivalent rotavirus vaccine in China. J Clin Lab Anal. 2023;37(23-24):e24989.
Crossref - Soares Weiser K, Bergman H, Henschke N, Pitan F, Cunliffe N. Vaccines for preventing rotavirus diarrhoea: vaccines in use. Cochrane Database Syst Rev. 2019(10):CD008521.
Crossref - Sadiq A, Bostan N, Khan J, Aziz A. Effect of rotavirus genetic diversity on vaccine impact. Rev Med Virol. 2022;32(1):e2259.
Crossref - Bencina G, Costantino C, Mameli C, et al. Real-world impact of rotavirus vaccination in European healthcare settings: A systematic literature review. Expert Rev Vaccines. 2022;21(8):1121-1136.
Crossref - Douglass T, Anderson A. Values, Politics and Power Relations in the Development and Regulation of Vaccines. Vaccines in Society. 2024:31-46.
Crossref - Glass RI, Tate JE, Jiang B, Parashar U. The rotavirus vaccine story: from discovery to the eventual control of rotavirus disease. J Infect Dis. 2021;224(Suppl 4):S331-S42.
Crossref - Geletu US, Usmael MA, Bari FD. Rotavirus in calves and its zoonotic importance. Vet Med Int. 2021;2021:6639701.
Crossref - Hung PJ, Chen CC. Diagnostic accuracy of rotavirus antigen tests in children: A systematic review and meta analysis. Trop Med Int Health. 2023;28(2):72-79.
Crossref - Raju B, Parikh R, Vetter V, Kolhapure S. Epidemiology of rotavirus gastroenteritis and need of high rotavirus vaccine coverage with early completion of vaccination schedule for protection against rotavirus diarrhea in India: A narrative review. Indian J Public Health. 2019;63(3):243-250.
Crossref - Akdag AI, Gupta S, Khan N, Upadhayay A, Ray P. Epidemiology and clinical features of rotavirus, adenovirus, and astrovirus infections and coinfections in children with acute gastroenteritis prior to rotavirus vaccine introduction in Meerut, North India. J Med Virol. 2020;92(8):1102-1109.
Crossref - Ghonaim AH, Hopo MG, Ghonaim NH, Jiang Y, He Q, Li W. The Epidemiology of Circulating Rotavirus Associated with Diarrhea in Egyptian Kids and Calves: A Review. Zoonoses. 2023;3(1).
Crossref - Kumar CG, Giri S, Chawla-Sarkar M, et al. Epidemiology of rotavirus diarrhea among children less than 5 years hospitalized with acute gastroenteritis prior to rotavirus vaccine introduction in India. Vaccine. 2020;38(51):8154-8160.
Crossref - Wang Y, Li J, Dai P, Liu P, Zhu F. Effectiveness of the oral human attenuated pentavalent rotavirus vaccine (RotaTeq™) postlicensure: a meta-analysis-2006-2020. Expert Rev Vaccines. 2021;20(4):437-448.
Crossref - Folorunso OS, Sebolai OM. Overview of the development, impacts, and challenges of live-attenuated oral rotavirus vaccines. Vaccines. 2020;8(3):341.
Crossref - O’Ryan M, Linhares AC. Update on Rotarix™: an oral human rotavirus vaccine. Expert review of vaccines. 2009;8(12):1627-41.
Crossref - Vesikari T. Rotavirus vaccination: a concise review. Clin Microbiol Infect. 2012;18(Supl 5):57-63.
Crossref - Skansberg A, Sauer M, Tan M, Santosham M, Jennings MC. Product review of the rotavirus vaccines ROTASIIL, ROTAVAC, and Rotavin-M1. Hum Vaccin Immunother. 2021;17(4):1223-1234.
Crossref - Naik SP, Zade JK, Sabale RN, et al. Stability of heat stable, live attenuated Rotavirus vaccine (ROTASIIL®). Vaccine. 2017;35(22):2962-2969.
Crossref - Omatola CA, Olaniran AO. Rotaviruses: From pathogenesis to disease control-A critical review. Viruses. 2022;14(5):875.
Crossref - Pereira P, Vetter V, Standaert B, Benninghoff B. Fifteen years of experience with the oral live-attenuated human rotavirus vaccine: reflections on lessons learned. Expert Rev Vaccines. 2020;19(8):755-769.
Crossref - Changotra H, Vij A. Rotavirus virus like particles (RV VLPs) vaccines: An update. Rev Med Virol. 2017;27(6):e1954.
Crossref - Mandeep S Dhingra M. Phase I/II, Randomized, Double-blind, Placebo-controlled, Dosage Selection (10e5.5 or 10e6.25 FFU of Each Constituent Serotype Per 0.5 mL) Study to Evaluate the Safety, Tolerability, and Immunogenicity of a 3-dose Series of Live Attenuated Tetravalent (G1-G4) Bovine-Human Reassortant Rotavirus Vaccine [BRV-TV] Administered to Healthy Indian Infants. India patent NCT01061658. 2010. https://adisinsight.springer.com/trials/700037969
- GlaxoSmithKline. Reactogenicity and Safety of GlaxoSmithKline (GSK) Biologicals’ Rotarix™ (Human Rotavirus Vaccine) in Indian Infants. India patent NCT00938327. 2018. https://clinicaltrials.gov/study/NCT00938327
- Ltd. SIoIP. Study on Liquid Bovine Rotavirus Pentavalent Vaccine (LBRV-PV) to Evaluate Lot-to-Lot Consistency and to Compare Non-Inferiority With ROTASIIL (Lyophilized BRV-PV) in Healthy Infants in India. India patent NCT03474055. 2018. https://clinicaltrials.gov/study/NCT03474055
- Ltd. SIoIP. A Clinical Trial to Study the Effect and Safety of Rotavirus Vaccine Against Severe Rotavirus Gastroenteritis in Healthy Indian Infants. India patent NCT02133690. 2018 2018-09-10. https://ctv.veeva.com/study/a-clinical-trial-to-study-the-effect-and-safety-of-rotavirus-vaccine-against-severe-rotavirus-gastro
- Ltd. SIoIP. Phase III Study on Rotavirus Vaccine to Evaluate Lot-to-lot Consistency and Interference With Routine UIP Immunization. India patent NCT02584816. 2018 2018-09-11. https://trial.medpath.com/clinical-trial/72bebd337a3600e2/nct02584816-bovine-rotavirus-vaccine-study-india
- N Bhandari. Disease Burden Study and Site Preparation for Planning and Implementation of the Phase III Trials for the Oral Rotavirus Vaccine 116E. India patent NCT01508533. 2012. https://clinicaltrials.gov/study/NCT01508533
- Limited BBI. A Phase III Clinical Trial to Evaluate the Protective Efficacy of Three Doses of Oral Rotavirus Vaccine (ORV) 116E (ROTAVAC). India patent NCT01305109. 2012 2012-06-12. https://ctv.veeva.com/study/a-phase-iii-clinical-trial-to-evaluate-the-protective-efficacy-of-three-doses-of-oral-rotavirus-vacc
- Caddy S, Papa G, Borodavka A, Desselberger U. Rotavirus research: 2014-2020. Virus Res. 2021;304:198499.
Crossref - Anderson ML, Esstman SM. In vitro particle-associated uridyltransferase activity of the rotavirus VP1 polymerase. Virology. 2022;577:24-31.
Crossref - Praveen M, Morales-Bayuelo A. Drug Designing against VP4, VP7 and NSP4 of Rotavirus Proteins-Insilico studies. Moroccan Journal of Chemistry. 2023;11(3):729-741.
Crossref - Martin D, Ouldali M, Menetrey J, Poncet D. Structural organisation of the rotavirus nonstructural protein NSP5. J Mol Biol. 2011;413(1):209-221.
Crossref - Aponte C, Mattion NM, Estes MK, Charpilienne A, Cohen J. Expression of two bovine rotavirus non-structural proteins (NSP 2, NSP 3) in the baculovirus system and production of monoclonal antibodies directed against the expressed proteins. Arch Virol. 1993;133(1-2):85-95.
Crossref - Donker NC, Kirkwood CD. Selection and evolutionary analysis in the nonstructural protein NSP2 of rotavirus A. Infect Genet Evol. 2012;12(7):1355-1361.
Crossref - Afchangi A, Jalilvand S, Mohajel N, Marashi SM, Shoja Z. Rotavirus VP6 as a potential vaccine candidate. Rev Med Virol. 2019;29(2):e2027.
Crossref - Kanai Y, Onishi M, Kawagishi T, et al. Reverse genetics approach for developing rotavirus vaccine candidates carrying VP4 and VP7 genes cloned from clinical isolates of human rotavirus. J Virol. 2020;95(2):10.1128/jvi. 01374-20.
Crossref - Lakatos K, McAdams D, White JA, Chen D. Formulation and preclinical studies with a trivalent rotavirus P2-VP8 subunit vaccine. Hum Vaccin Immunother. 2020;16(8):1957-1968.
Crossref - Uprety T, Wang D, Li F. Recent advances in rotavirus reverse genetics and its utilization in basic research and vaccine development. Arch Virol. 2021;166(9):2369-2386.
Crossref - Agarwal S, Hickey JM, McAdams D, et al. Effect of aluminum adjuvant and preservatives on structural integrity and physicochemical stability profiles of three recombinant subunit rotavirus vaccine antigens. J Pharm Sci. 2020;109(1):476-487.
Crossref - Pullagurla SR, Kumar P, Ogun O, et al. Modeling the long-term 2-8 C stability profiles of a live, rotavirus vaccine candidate (RV3-BB) in various liquid formulations via extrapolations of real-time and accelerated stability data. Biologicals. 2022;75:21-28.
Crossref - Cates JE, Tate JE, Parashar U. Rotavirus vaccines: progress and new developments. Expert Opin Biol Ther. 2022;22(3):423-432.
Crossref - Yung CF, Chong CY, Thoon KC. Age at first rotavirus vaccination and risk of intussusception in infants: a public health modeling analysis. Drug Safety. 2016;39(8):745-748.
Crossref - Barsoum Z. Paediatric rotavirus gastroenteritis: A prospective study of regional prevalent genotypes, genotype correlation with disease severity and viral co-infection in County Mayo, Ireland, in the year following rotavirus vaccine introduction in Ireland. Journal of Virological Methods. 2021;294.
Crossref - Rotavirus vaccine (RV) supply and demand update. 2022 ed; 2022:1-11. https://www.unicef.org/supply/reports/rotavirus-vaccine-rv-supply-and-demand-update
- WHO. Rotavirus: Vaccine Preventable Diseases Surveillance Standards. 4 September 2018 ed: World Health Organization. 2018:1-11. https://www.who.int/publications/m/item/vaccine-preventable-diseases-surveillance-standards-rotavirus
- Rotavirus vaccines: WHO position paper – July 2021. Weekly Epidemiological Record, 96(28): 301-219. https://www.who.int/publications/i/item/WHO-WER9628
- Vetter V, Gardner RC, Debrus S, Benninghoff B, Pereira P. Established and new rotavirus vaccines: a comprehensive review for healthcare professionals. Hum Vaccin Immunother. 2022;18(1):1870395.
Crossref - Nair NP, Reddy NS, Giri S, et al. Rotavirus vaccine impact assessment surveillance in India: protocol and methods. BMJ Open. 2019;9(4):e024840.
Crossref - Li C, Luo G, Zeng Y, et al. Establishment of sandwich ELISA for quality control in rotavirus vaccine production. Vaccines. 2022;10(2):243.
Crossref - Artaud C, Kara L, Launay O. Vaccine development: from preclinical studies to phase 1/2 clinical trials. Methods Mol Biol. 2019:2013:165-176.
Crossref - Joshi MS, Deore SG, Walimbe AM, Ranshing SS, Chitambar SD. Evaluation of different genomic regions of Rotavirus A for development of real time PCR. J Virol Methods. 2019;266:65-71.
Crossref - Gautam R, Esona MD, Mijatovic-Rustempasic S, Tam KI, Gentsch JR, Bowen MD. Real-time RT-PCR assays to differentiate wild-type group A rotavirus strains from Rotarix® and RotaTeq® vaccine strains in stool samples. Hum Vaccin Immunother. 2014;10(3):767-777.
Crossref - De Grazia S, Bonura F, Pepe A, et al. Performance analysis of two immunochromatographic assays for the diagnosis of rotavirus infection. J Virol Methods. 2017;243:50-54.
Crossref - McAdams D, Estrada M, Holland D, et al. Concordance of in vitro and in vivo measures of non-replicating rotavirus vaccine potency. Vaccine. 2022;40(34):5069-5078.
Crossref - Shin J, Lei D, Conrad C, Knezevic I, Wood D. International regulatory requirements for vaccine safety and potency testing: a WHO perspective&. Procedia in Vaccinology. 2011;5:164-170.
Crossref - Burnett E, Parashar U, Tate J. Rotavirus vaccines: effectiveness, safety, and future directions. Pediatr Drugs. 2018;20(3):223-233.
Crossref - Kanchan V, Zaman K, Aziz AB, et al. A randomized Phase I/II study to evaluate safety and reactogenicity of a heat-stable rotavirus vaccine in healthy adults followed by evaluation of the safety, reactogenicity, and immunogenicity in infants. Hum Vaccin Immunother. 2020;16(3):693-702.
Crossref - Groome MJ, Fairlie L, Morrison J, et al. Safety and immunogenicity of a parenteral trivalent P2-VP8 subunit rotavirus vaccine: a multisite, randomised, double-blind, placebo-controlled trial. Lancet Infect Dis. 2020;20(7):851-863.
Crossref - Lal M, Jarrahian C, Zhu C, et al. Stability of live attenuated rotavirus vaccine with selected preservatives and primary containers. Vaccine. 2016;34(22):2483-2489.
Crossref - Hallowell BD, Tate J, Parashar U. An overview of rotavirus vaccination programs in developing countries. Expert Rev Vaccin. 2020;19(6):529-537.
Crossref - Dennehy PH. Rotavirus infection: an update on management and prevention. Adv Pediatr. 2012;59(1):47-74.
Crossref - Xia M, Huang P, Jiang X, Tan M. A nanoparticle-based trivalent vaccine targeting the glycan binding VP8* domains of rotaviruses. Viruses. 2021;13(1):72.
Crossref - Banyai K, Laszlo B, Duque J, et al. Systematic review of regional and temporal trends in global rotavirus strain diversity in the pre rotavirus vaccine era: insights for understanding the impact of rotavirus vaccination programs. Vaccine. 2012;30(Suppl 1):A122-A130.
Crossref
© The Author(s) 2026. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.